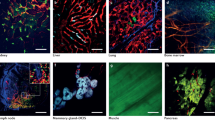
Biomechanical interactions of cancer cells with the microvasculature were studied using high resolution intravital videomicroscopy. We compared initial arrest of murine B16F10 melanoma and D2A1 mammary carcinoma cells fluorescently labelled with calcein-AM, in low pressure (liver) vs high pressure (cremaster muscle) microvascular beds. Cells were arrested due to size restriction at the inflow side of the microcirculation, penetrating further and becoming more deformed in muscle than liver [median length to width ratios of 3.3 vs 1.3 for D2A1 cells, and 2.5 vs 1.2 for B16F10, at 1 min post-injection (p.i.)]. During the next 2 h many cells became stretched, giving maximum length to width ratios of 68 vs 22.1 (D2A1) and 28 vs 5.6 (B16F10) in muscle vs liver. Ethidium bromide exclusion demonstrated that over 97% of the cells maintained membrane integrity for > 2 h p.i. (In contrast, when an acridine orange labelling procedure was used, membrane disruption of B16F10 cells occurred within 15 min p.i.) Our experiments do not indicate the ultimate fate of the cancer cells, but if cell lysis occurs it must be on a time scale of hours rather than minutes. We report a process of ‘clasmatosis’ in cancer cells arrested in the microcirculation: large membrane-enclosed fragments (>3 µm in diameter) became ‘pinched off’ from arrested cells, in both liver and muscle, often within minutes or even seconds of arrest. The significance of this process is not yet understood. In this study intravital videomicroscopy has thus provided a valuable clarification of the interactions of cancer cells with vessel walls during metastasis.
Similar content being viewed by others
References
Weiss L, 1990, Metastatic inefficiency. Advances in Cancer Research, 54, 159–211.
Fidler IJ, 1970, Metastasis: quantitative analysis of distribution and fate of tumor emboli labeled with 125I-5-iodo-2′-deoxyuridine. Journal of the National Cancer Institute, 45, 773–782.
Liotta LA, Vembu D, Saini RK and Boone C, 1978, In vivo monitoring of the death rate of artificial murine pulmonary micrometastases. Cancer Research, 38, 1231–1236.
Mayhew E and Glaves D, 1984, Quantitation of tumorigenic disseminating and arrested cancer cells. British Journal of Cancer, 50, 159–166.
Paget S, 1889, The distribution of secondary growths in cancer of the breast. Lancet, 1, 571–573.
Hart IR, 1982, “Seed and soil” revisited: mechanisms of site-specific metastasis. Cancer Metastasis Reviews, 1, 5–16.
Nicolson GL, 1988, Organ specificity of tumor metastasis: role of preferential adhesion, invasion and growth of malignant cells at specific secondary sites. Cancer Metastasis Reviews, 7, 143–188.
Hanna N, 1982, Role of natural killer cells in control of cancer metastasis. Cancer Metastasis Reviews, 1, 45–64.
Key M, 1983, Macrophages in cancer metastasis and their relevance to metastatic growth. Cancer Metastasis Reviews, 2, 75–88.
Weiss L, 1987, The hemodynamic destruction of circulating cancer cells. Biorheology, 24, 105–115.
Weiss L and Schmid-Schönbein GW, 1989, Biomechanical interactions of cancer cells with the microvasculature during metastasis. Cell Biophysics, 14, 187–215.
Weiss L. The biomechanics of cancer cell traffic, arrest, and intravascular destruction. In: Orr FW, Buchanan MR and Weiss L, eds. Microcirculation in Cancer Metastasis, pp. 131–144. Boca Raton: CRC Press, 1991.
Wood S, 1958, Pathogenesis of metastasis formation observed in vivo in the rabbit ear chamber. Archives of Pathology, 66, 550–558.
Zeidman I, 1961, The fate of circulating tumor cells. I. Passage of cells through capillaries. Cancer Research, 21, 38–39.
Blomqvist G, Skolnik G, Braide M, Bjursten L-M, Blixt A and Bagge U, 1988, Differences in lodgement of tumor cells in muscle and liver. Clinical & Experimental Metastasis, 6, 285–289.
Weiss L, Nannmark U, Johansson BR and Bagge U, 1992, Lethal deformation of cancer cells in the microcirculation: a potential rate regulator of hematogenous metastasis. International Journal of Cancer, 50, 103–107.
Chambers AF, Schmidt EE, MacDonald IC, Morris VL and Groom AC, 1992, Early steps in hematogenous metastasis of 1316171 melanoma cells in chick embryos studied by high-resolution intravital videomicroscopy. Journal of the National Cancer Institute, 84, 797–803.
MacDonald IC, Schmidt EE, Morris VL, Chambers AF and Groom AC, 1992, Intravital videomicroscopy of the chorioallanotoic microcirculation: a model system for studying metastasis. Microvascular Research, 44, 185–199.
Fidler IJ, 1973, Selection of successive tumour lines for metastasis. Nature New Biology, 242, 148–149.
Fidler IJ, 1975, Biological behavior of malignant melanoma cells correlated to their survival in vivo. Cancer Research, 35, 218–224.
Miller FR, McEachern D and Miller BE, 1989, Growth regulation of mouse mammary tumor cells in collagen gel cultures by diffusible factors produced by normal mammary gland epithelium and stromal fibroblasts. Cancer Research, 49, 6091–6097.
Morris VL, Tuck AB, Wilson SM, Percy D and Chambers AF, 1993, Tumor progression and metastasis in murine D2 hyperplastic alveolar nodule mammary tumor cell lines. Clinical & Experimental Metastasis, 11, 103–112.
Chambers AF and Wilson S, 1988, Use of NeoR B16F1 murine melanoma cells to assess clonality of experimental metastases in the immune-deficient chick embryo. Clinical & Experimental Metastasis, 6, 171–182.
MacPhee PJ, Schmidt EE, Keown PA and Groom AC, 1988, Microcirculatory changes in livers of mice infected with murine hepatitis virus. Evidence from microcorrosion casts and measurements of red cell velocity. Microvascular Research, 36, 140–149.
Jacoby F. Macrophages. In: Willmer EN, ed. Cells and Tissues in Culture, pp. 27–32. New York: Academic Press, 1965.
Fidler IJ. Patterns of tumor cell arrest and development. In: Weiss L, ed. Fundamental Aspects of Metastasis, pp. 275–289. North-Holland Publishing Co., 1976.
Uggla AH and Sundell-Bergman S, 1990, The induction and repair of DNA damage detected by the DNA precipitation assay in Chinese hamster ovary cells treated with acridine orange + visible light. Mutation Research, 236, 119–127.
Tatsuta M, Iishi H, Yamamura H, Yamamoto R and Okuda S, 1988, Comparison of photodynamic inactivation of experimental stomach tumors sensi tized by acridine orange or hematoporphyrin derivatives. Oncology, 45, 35–40.
Weiss L, Harlos JP and Elkin G, 1991, Measurements of compression of Ehrlich ascites cells and their relevance to hematogenous metastasis. Biorheology, 28, 185–193.
Barberá-Guillem E, Smith I and Weiss L, 1993, Cancer-cell traffic in the liver. II. Arrest, transit and death of B161710 and M5076 cells in the sinusoids. International Journal of Cancer, 53, 298–301.
Barberá-Guillem E, Alonso-Varona A and Vidal-Vanaclocha F, 1989, Selective implantation and growth in rats and mice of experimental liver meta stasis in acinar zone one. Cancer Research, 49, 4003–4010.
Nakata K, Leong GF and Brauer RW, 1960, Direct measurement of blood pressures in minute vessels of the liver. American Journal of Physiology, 199, 1181–1188.
Raz A, Barzilai R, Spira G and Inbar M, 1978, Oncogenicity and immunogenicity associated with membranes isolated from cell-free ascites fluid of lymphoma-bearing mice. Cancer Research, 38, 2480–2485.
Poste G and Nicolson GL, 1980, Arrest and metastasis of blood-borne tumor cells are modified by fusion of plasma membrane vesicles from highly metastatic cells. Proceedings of the National Academy of Sciences, USA, 77, 399–403.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Morris, V.L., MacDonald, I.C., Koop, S. et al. Early interactions of cancer cells with the microvasculature in mouse liver and muscle during hematogenous metastasis: videomicroscopic analysis. Clin Exp Metast 11, 377–390 (1993). https://doi.org/10.1007/BF00132981
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00132981