Abstract
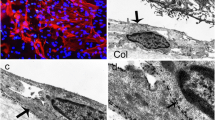
Cell growth in proliferative vitreoretinopathy (PVR) is activated by naturally occurring mitogenic substances; thus, growth factors seem to play an important role in PVR. Cell activation by growth factors requires the presence of specific cell-bound receptors by which the mitogenic signal is transmitted. Using the indirect immunoperoxidase method, we investigated frozen sections of 15 epiretinal membranes in PVR for the presence of epidermal growth factor receptor (EGFR), which is commonly utilized by epidermal growth factor (EGF) and transforming growth factor-alpha (TGF-alpha). This receptor could be localized only in one epiretinal membrane. The receptor-bearing cells stained positively for vimentin, cytokeratin, and macrophage marker Ki-M7, indicating that they most probably correspond to transformed macrophages. They stained negatively for desmin and glial fibrillary acidic protein. The cells were embedded in a fibronectin-positive intercellular matrix and were in an actively proliferating state as demonstrated by positive staining for the nuclear proliferation marker Ki-67. We propose that EGFR might be expressed only in certain as yet undefined stages of PVR.
Similar content being viewed by others
References
Berridge MJ (1986) Inositol lipids and cell proliferation. In: Kahn P, Graf Th (eds) Oncogenes and growth control. Springer, Berlin Heidelberg New York, pp 147–153
Burke JM, Twining SS (1987) Vitreous macrophage elicitation: generations of stimulants for pigment epithelium in vitro. Invest Ophthalmol Vis Sci 28:1100–1107
Campochiaro PA, Glaser BM (1985) Platelet-derived growth factor is chemotactic for human retinal pigment epithelial cells. Arch Ophthalmol 103:576–579
Campochiaro PA, Jerdan JA, Glaser BM, Cardin A, Michels RG (1985) Vitreous aspirates from patients with proliferative vitreoretinopathy stimulate retinal pigment epithelial cell migration. Arch Ophthalmol 103:1403–1405
Carpenter G, Cohen S (1976) [125I]-labeled human epidermal growth factor (hEGF): binding, internalization, and degradation in human fibroblasts. J Cell Biol 71:159–171
Carpenter G, Goodman L, Shaver L (1986) The physiology of epidermal growth factor. In: Kahn P, Graf Th (eds) Oncogenes and growth control. Springer, Berlin Heidelberg New York, pp 65–69
Caselitz J, Weber W, Seifert G (1986) Morphologische Untersuchungen des epidermalen Wachstumsfaktorrezeptor. Studien an menschlichen Tumoren und Tumortransplantaten auf athymische Mäuse mit besonderer Berücksichtigung von Speichel-drüsentumoren. (Morphological investigation of the epidermal growth factor receptor. Analysis of human tumors and tumor transplants in athymic mice, with special reference to salivary gland tumors.) Verh Dtsch Ges Pathol 70:343–346
Cohen S (1962) Isolation of a submaxillary gland protein accelerating incisor eruption and eyelid opening in the newborn animal. J Biol Chem 237:1555–1562
Couch JM, Cullen P, Casey TA, Fabre JW (1987) Mitotic activity of corneal endothelial cells in organ culture with recombinant human epidermal growth factor. Ophthalmology 94:1–6
Couch JM, Cullen P, Casey T, Fabre J (1988) Epidermal growth factor receptor sites in the human and monkey eye (abstr). Invest Ophthalmol Vis Sci 29 [Suppl]:86
Derynck R (1986) Transforming growth factor-alpha. In: Kahn P, Graf Th (eds) Oncogenes and growth control. Springer, Berlin Heidelberg New York, pp 58–64
Frati L, Daniele S, Delogu A, Covelli I (1972) Selective binding of the epidermal growth factor and its specific effects on the epithelial cells of the cornea. Exp Eye Res 14:135–141
Gerdes J, Schwab U, Lemke H, Stein H (1983) Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer 31:13–20
Gerdes J, Lemke H, Baisch H, Wacker H-H, Schwab U, Stein H (1984) Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 133:1710–1715
Gusterson B, Cowley G, Smith JA, Ozanne B (1984) Cellular localization of human epidermal growth factor receptor. Cell Biol Int Rep 8:649–657
Harvey AK, Roberge F, Hjelmeland LM (1987) Chemotaxis of rat retinal glia to growth factors found in repairing wounds. Invest Ophthalmol Vis Sci 28:1092–1099
Hiscott PS, Grierson I, McLeod D (1984) Retinal pigment epithelial cells in epiretinal membranes: an immunohistochemical study. Br J Ophthalmol 68:708–715
Ho PC, Davis WH, Elliott JH, Cohen S (1974) Kinetics of corneal epithelial regeneration and epidermal growth factor. Invest Ophthalmol Vis Sci 13:804–809
Hui YN, Goodnight R, Zhang XJ, Sorgente N, Ryan J (1988) Glial epiretinal membranes and contraction. Immnnohistochemical and morphological studies. Arch Ophthalmol 106:1280–1285
Kampik A, Kenyon KR, Michels RG, Green WR, Cruz ZC de la (1981) Epiretinal and vitreous membranes. Comparative study of 56 cases. Arch Ophthalmol 99:1445–1454
Kirchhof B, Kirchhof E, Ryan SJ, Dixon JFP, Barton BE, Sorgente N (1989) Macrophage modulation of retinal pigment epithelial cell migration and proliferation. Graefe's Arch Clin Exp Ophthalmol 227:60–66
Massague J (1983) Epidermal growth factor-like transforming growth factor: II. Interactions with epidermal growth factor receptors in human placenta membranes and A431 cells. J Biol Chem 258:13614–13620
Miller B, Miller H, Patterson R, Ryan SJ (1986) Retinal wound healing. Cellular activity at the vitreoretinal interface. Arch Ophthalmol 104:281–285
Nishida T, Nakamura M, Mishima H, Otori T (1990) Kinetics of stimulatory action of fibronectin and EGF on corneal epithelial migration (abstr). Invest Ophthalmol Vis Sci 31 [Suppl 4]:54
Rappolee DA, Mark D, Banda MJ, Werb Z (1988) Wound macrophages express TGF-alpha and other growth factors in vivo: analysis by mRNA phenotyping. Science 241:708–711
Rowen SL, Glaser BM (1985) Retinal pigment epithelial cells release a chemoattractant for astrocytes. Arch Ophthalmol 103:704–707
Savage RC Jr, Cohen S (1973) Proliferation of corneal epithelium induced by epidermal growth factor. Exp Eye Res 15:361–366
Schreiber AB, Winkler ME, Derynck R (1986) Transforming growth factor-a is a more potent angiogenic mediator than epidermal growth factor. Science 232:1250–1253
Vinores SA, Campochiaro PA, Conway BP (1990) Ultrastructural and electron-immunocytochemical characterization of cells in epiretinal membranes. Invest Ophthalmol Vis Sci 31:14–28
Weller M, Heimann K, Wiedemann P (1988) Demonstration of mononuclear phagocytes in a human epiretinal membrane using a monoclonal anti-human macrophage antibody. Graefe's Arch Clin Exp Ophthalmol 226:252–254
Weller M, Wiedemann P, Heimann K, Zilles K (1988) The significance of fibronectin in vitreoretinal pathology. A critical evaluation. Graefe's Arch Clin Exp Ophthalmol 226:294–298
Wiedemann P, Ryan SJ, Novar P, Sorgente N (1985) Vitreous stimulates proliferation of fibroblasts and retinal pigment epithelial cells. Exp Eye Res 41:619–628
Yamashita H, Hori S, Masuda K (1986) Population and proportion of component cells in preretinal membranes. Jpn J Ophthalmol 30:269–281
Zhu Z-R, Begg IS, Palcic B (1990) Macrophage modulatio of fibroblast migration and proliferation in vitro (abstr). Invest Ophthalmol Vis Sci 31 [Suppl 4]:443
Author information
Authors and Affiliations
Additional information
Offprint requests to: H.-P. Heidenkummer
Rights and permissions
About this article
Cite this article
Heidenkummer, HP., Kampik, A. Immunohistochemical localization of epidermal growth factor receptor in a human epiretinal membrane. Graefe's Arch Clin Exp Ophthalmol 229, 492–496 (1991). https://doi.org/10.1007/BF00166316
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00166316