Abstract
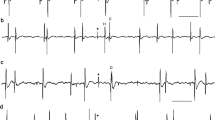
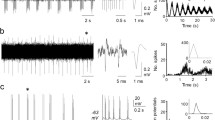
The response discharges of precentral motor cortical cells to brief trains of vibration applied to the tendon of biceps brachii were analyzed in two alert but passive monkeys. The activity of 20 phasictonic and 6 tonic cells was analyzed. All had functional linkages with flexor muscles during a preceding flexion task and responded to passive movement of the elbow. Taking as a reference the stereotyped reflex response in the stretched muscle, the effect of changes in the amplitude of a constant frequency vibration (4 vibrations at 58 Hz) was quantified statistically in peristimulus histograms of the cortical cell discharges. All cells were transiently influenced by low vibration amplitudes. Most responses (71 %) were excitatory and occurred at a mean latency of 24 ms, which is consistent with cells activated by input from stretch receptors. Excitatory, reproducible responses to the lowest vibration amplitudes were more frequent in phasictonic than in pure tonic cells. Large-amplitude vibrations always excited the motor cortical cells. The sign of the responses to vibration matched that to passive elbow movements for most cells. These findings show that elbowrelated motor cortical cells are very sensitive to proprioceptive input from primary spindle afferents.
Similar content being viewed by others
References
Albe-Fessard D, Liebeskind J (1966) Origine des messages so matosensitifs activant les cellules du cortex moteur chez le singe (in French). Exp Brain Res 1: 127–146
Ashby P, Stalberg E, Winkler T, Hunter JP (1987) Further observations on the depression of group Ia facilitation of motoneurons by vibration in man. Exp Brain Res 69: 1–6
Bauswein E, Fromm C, Werner W, Zieman U (1991) Phasic and tonic responses of premotor and primary motor cortex neurons to torque changes. Exp Brain Res 86: 303–310
Burke D, Hagbarth ICE, Löfstedt L, Wallin BG (1976) The responses of human spindle endings to vibration of noncon tracting muscles. J Physiol (Lond) 261: 673–693
Cheney PD, Fetz EE (1980) Functional classes of primate corticomotoneuronal cells and their relation to active force. J Neurophysiol 44: 773–791
Cheney PD, Fetz EE (1984) Corticomotoneuronal cells contribute to longlatency stretch reflexes in the rhesus monkey. J Physiol (Lond) 349: 249–272
Colebatch JG, Sayer RJ, Porter R, White OB (1991) Responses of monkey precentral neurones to passive movements and phasic muscle stretch: relevance to man. Electroencephalogr Clin Neurophysiol 75: 44–55
Conrad B, Meyer-Lohman J, Matsunami K, Brooks VB (1975) Precentral unit activity following torque pulse injections into elbow movements. Brain Res 94: 219–236
Eklund G (1972) Position sense and state of contraction; the effects of vibration. J Neurol Neurosurg Psychiatry 35: 606–611
Ellaway PH (1978) Cumulative sum technique and its application to the analysis of peristimulus time histograms. Electroencephalogr Clin Neurophysiol 45: 302–304
Evarts EV (1981) Role of motor cortex in voluntary movements in primates. In: Brooks VB (ed) Motor control. (Handbook of physiology, sect 1, The nervous system, vol II, part 2) Am Physiol Soc, Bethesda, MD, pp 1083–1120
Evarts EV, Fromm C (1977) Sensory responses in motor cortex neurons during precise motor control. Neurosci Lett 5:267–272
Evarts EV, Tanji J (1976) Reflex and intended responses in motor cortex pyramidal tract neurones of monkey. J Neurophysiol 39: 1069–1080
Flament D, Höre J (1988) Relations of motor cortex neural discharge to kinematics of passive and active elbow movements in the monkey. J Neurophysiol 60: 1268–1284
Fourment A, Belhaj-Saïf A, Maton B (1995) Functional linkages between motor cortical cells and elbow flexor muscles. Evidence for and characteristics of postspike facilitation. J Neurophysiol 74: 130–141
Fromm C, Wise SP, Evarts EV (1984) Sensory response properties of pyramidal tract neurons in the precentral motor cortex and postcentral gyrus of the rhesus monkey. Exp Brain Res 54: 177–185
Gandevia SC, Burke D (1992) Does the nervous system depend on kinesthetic information to control natural limb movements? Behav Brain Sci 15: 614–632
dwin GM, McCloskey DI, Matthews PBC (1972) The contribution of muscle afferents to kinaesthesia shown by vibration induced illusions of movement and by the effect of paralysing joint afferents. Brain 95: 705–748
Hepp-Reymond M-C (1988) Functional organization of motor cortex and its participation in voluntary movements. In: Seklis HD, Erwin J (eds) Comparative primate biology, vol 4. Liss, New York, pp 501–624
Hummelsheim H, Bianchetti M, Wiesendanger M, Wiesendanger R (1988) Sensory inputs to the agranular motor fields: a comparison between precentral, supplementarymotor and premotor areas in the monkey. Exp Brain Res 69: 289–298
Jami L (1992) Golgi tendon organs in mammalian skeletal muscle: functional properties and central actions. Physiol Rev 72:623–666
Matthews PBC (1972) Mammalian muscle receptors and their central action. Arnold, London
Matthews PBC (1984) Observations on the time course of the electromyographic response reflexly elicited by muscle vibration in man. J Physiol (Lond) 353: 447–461
Morin C, Pierrot Deseilligny E, Hultborn H (1984) Evidence for presynaptic inhibition of muscle spindle Ia afferents in man. Neurosci Lett 44: 137–142
Porter R, Lemon R (1993) Corticospinal function and voluntary movement, Clarendon, Oxford
Roll JP, Vedel JP (1982) Kinaesthetic role of muscle afferents in man, studied by tendon vibration and microneuronography. Exp Brain Res 47: 177–190
Roll JP, Bonnet M, Hugon M (1973) The baboon (Papio papio) as a model for the study of spinal reflexes. In: Desmedt JE (ed) New developments in EMG and clinical neurophysiology, vol 3. Karger, Basel, pp 194–208
Roll JP, Vedel JP, Ribot E (1989) Alteration of proprioceptive messages induced by tendon vibration in man: a microneuronographic study. Exp Brain Res 76: 213–222
Romaiguère P, Vedel JP, Azulay JP, Pagni S (1991) Differential activation of motor units in the wrist extensor muscles during the tonic vibration reflex in man. J Physiol (Lond) 444: 645–667
Vallbo AB (1973) Muscle spindle afferent discharge from resting and contracting muscles in normal human subjects. In: Desmedt JE (ed) New developments in EMG and clinical neurophysiology, vol 3. Karger, Basel, pp 251–262
Wannier TMJ, Maier MA, Hepp-Reymond M-C (1991) Contrasting properties of monkey somatosensory and motor cortex neurons activated during the control of force in precision grip. J Neurophysiol 65: 572–589
Wise SP, Tanji J (1981) Neuronal responses in sensorimotor cortex to ramp displacements and maintained positions imposed on hindlimb of the unanesthetized monkey. J Neurophysiol 45: 482–500
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fourment, A., Chennevelle, J.M., Belhaj-Saïf, A. et al. Responses of motor cortical cells to short trains of vibration. Exp Brain Res 111, 208–214 (1996). https://doi.org/10.1007/BF00227298
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227298