Summary
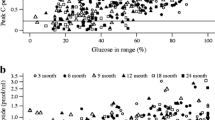
Many patients with Type 2 (non-insulin-dependent) diabetes mellitus are treated with insulin in order to control hyperglycaemia. We studied fasting plasma C-peptide, glucagon stimulated plasma C-peptide, and 24 h urinary C-peptide in relation to clinical type of diabetes in 132 insulin treated diabetic subjects. Patients were classified clinically as Type 1 (insulin-dependent) diabetic subjects in the presence of at least two of the following criteria: 1) significant ketonuria, 2) insulin treatment started within one year after diagnosis, 3) age of diagnosis ≤40 years, and 4) weight below 110% of ideal weight of the same age and sex. Eighty patients were classified as Type 1 and 52 as Type 2 diabetic subjects. A second classification of patients into 6 C-peptide classes was then performed. Class I consisted of patients without islet B-cell function. Class II-VI had preserved islet B-cell function and were separated according to the 20%, 40%, 60% and 80% C-peptide percentiles. The two classifications of patients were compared by calculating the prevalence of clinical Type 1 and Type 2 diabetes in each of the C-peptide classes. This analysis showed that patients with a fasting plasma C-peptide value <0.20 nmol/l, a glucagon stimulated plasma C-peptide value <0.32 nmol/l, and a urinary C-peptide value <3.1 nmol/l, or <0.54 nmol/mmol creatinine/24 h, or <5.4 nmol/24 h mainly were Type 1 diabetic patients; while patients with C-peptide levels above these values mainly were Type 2. At these limits the percentage, predictive value of positive tests as indicators of Type 2 diabetes were as follows: fasting C-peptide 83%, stimulated C-peptide 86%, and urinary C-peptide expressed as nmol/l 76%, as nmol/mmol creatinine/24 h 79%, and as nmol/24 h 78%. Similarly, the percentage predictive value of negative tests as indicators of Type 1 diabetes were as follows: fasting C-peptide 86%, stimulated C-peptide 88%, and urinary C-peptide expressed as nmol/l 79%, as nmol· mmol creatinine·24 h 81%, and as nmol/24 h 80%. If patients without detectable C-peptide were excluded, the predictive value of negative tests were as follows: fasting C-peptide 81%, stimulated C-peptide 88%, urinary C-peptide expressed as nmol/l 61%, as nmol/mmol creatinine/24 h 69%, and as nmol/24 h 64%. In conclusion, post glucagon C-peptide gives a good distinction between Type 1 and Type 2 diabetes mellitus in insulin treated diabetes while 24 h urinary C-peptide gives a less sensitive distinction between the clinical types of diabetes.
Article PDF
Similar content being viewed by others
References
World Health Organization (1985) Diabetes Mellitus. Technical Report Series 727. Geneva, Switzerland
Laakso M, Pyörälä K (1985) Age of onset and type of diabetes. Diabetes Care 8: 114–117
Faber OK, Binder C (1977) C-peptide response to glucagon. A test for the residual B-cell function in diabetes mellitus. Diabetes 26: 605–610
Sjöberg S, Gunnarsson R, Östman J (1983) Residual C-peptide production in type 1 diabetes mellitus. Acta Med Scand 214: 231–238
Siegler DE, Reeves ML, Skyler JS (1982) Lack of effect of improved glycemic control on C-peptide secretion in patients without residual B-cell function. Diabetes Care 5: 334–336
Gonen B, Goldman J, Baldwin D, Goldberg RB, Ryan WG, Blix PM, Schantzlin D, Frits KH, Rubenstein AH (1979) Metabolic control in diabetic patients. Effect of insulin secretory reserve (measured by plasma C-peptide levels) and circulating insulin antibodies. Diabetes 28: 749–753
Stiller CR, Dupre J, Gent M, Jenner MR, Keown PA, Laupacis A, Martell R, Rodger NW, Graffenried BV, Wolfe BMJ (1984) Effects of cyclosporine immunosuppression in insulin-dependent diabetes mellitus of recent onset. Science 223: 1362–1367
Madsbad S, Krarup T, McNair P, Christiansen C, Faber OK, Transbøl I, Binder C (1981) Practical clinical value of the C-peptide response to glucagon stimulation in the choice of treatment in diabetes mellitus. Acta Med Scand 210: 153–156
Koskinen P, Viikari J, Irjala K, Kaihola H-L, Seppälä P (1986) Plasma and urinary C-peptide in the classification of adult diabetics. Scand J Clin Lab Invest 46: 655–663
Groop L, Pelkonen R (1984) Treatment failures: A common problem in the management of patients with type 2 diabetes. Acta Endocrinologica 105 [Suppl 262]: 131–135
Garvey WT, Olefsky JM, Griffin J, Hamman RF, Kolterman OG (1985) The effect of insulin treatment on insulin secretion and insulin action in type 2 diabetes mellitus. Diabetes 34: 222–234
Hoekstra JBL, Van Rijn HJ, Thijssen JHH, Erkelens DW (1982) C-peptide reactivity as a measure of insulin dependency in obese diabetic patients treated with insulin. Diabetes Care 5: 585–591
Natvig H (1956) Nye Høyde-Vekttabeller for Norske Kvinner og Menn. Landsforeningen for Kosthold og Helse, Oslo
Gjessing HJ, Matzen LE, Faber OK, Frøland A (1989) Sensitivity and reproducibility of urine C-peptide as estimate of islet B-cell function in insulin treated diabetes. Diabetic Med 6: 329–333
Heding LG (1975) Radioimmunologic determination of human C-peptide in serum. Diabetologia 11: 541–548
Faber OK, Binder C, Markussen J, Heding LG, Naithani VK, Kuzuya H, Blix P, Horwitz V, Rubenstein AH (1978) Characterization of seven C-peptide antisera. Diabetes 27 [Suppl 1]: 170–177
Hendriksen C, Faber OK, Drejer J, Binder C (1977) Prevalence of residual B-cell function in insulin treated diabetics evaluated by the plasma C-peptide response to intravenous glucagon. Diabetologia 13: 615–619
Crossley JT, James AG, Elliott RB, Berryman CC, Edgar BW (1981) Residual B-cell function and islet cell antibodies in diabetic children. Pediatr Res 15: 62–65
Meistas MT, Rendell M, Margolis S, Kowarski AA (1982) Estimation of the secretion rate of insulin from urinary excretion rate of C-peptide. Study in obese and diabetic subjects. Diabetes 31: 449–453
Heding LG (1972) Determination of total serum insulin (IRI) in insulin treated diabetic patients. Diabetologia 8: 260–266
Schmidt FH (1973) Enzymatische Teste zur Schnell-Diagnose. In: Beringer A (ed) 3. Internationales Donau-Symposium über Diabetes Mellitus. Verlag W Maudrich, Wien München Bern, pp 938
Gjessing HJ, Matzen LE, Pedersen PC, Faber OK, Frøland A (1988) Insulin requirement in non-insulin-dependent diabetes mellitus: Relation to simple tests of islet B-cell function and insulin sensitivity. Diabetic Med 5: 328–332
Gjessing HJ, Matzen LE, Frøland A, Faber OK (1987) Correlations between fasting plasma C-peptide, glucagon-stimulated plasma C-peptide, and urinary C-peptide in insulin-treated diabetics. Diabetes Care 10: 487–490
Welborn TA, Garcia-Webb P, Bonser A (1981) Basal C-peptide in the discrimination of Type 1 from Type 2 diabetes. Diabetes Care 4: 614–619
Meistas MT, Zadik Z, Margolis S, Kowarski A (1981) Correlation of urinary excretion of C-peptide with the integrated concentration and secretion rate of insulin. Diabetes 30: 639–643
Tillil H, Shapiro ET, Given BD, Rue P, Rubenstein AH, Galloway JA, Polonsky KS (1988) Reevaluation of urine C-peptide as measure of insulin secretion. Diabetes 37: 1195–1201
Arnold-Larsen S, Madsbad S, Kühl C (1987) Reproducibility of the glucagon test. Diabetic Med 4: 299–303
Bonser A, Garcia-Webb P (1981) C-peptide measurement and its clinical usefulness: a review. Ann Clin Biochem 18: 200–206
Massi-Benedetti M, Burrin JM, Capaldo B, Alberti KGMM (1981) A comparative study of the activity of biosynthetic human insulin and pork insulin using the glucose clamp technique in normal subjects. Diabetes Care 4: 163–167
Garvey WT, Revers RR, Kolterman OG, Rubenstein AH, Olefsky JM (1985) Modulation of insulin secretion by insulin and glucose in Type II diabetes mellitus. J Clin Endocrinol Metab 60: 559–568
Agner T, Damm P, Binder C (1987) Remission in IDDM: Prospective study of basal C-peptide and insulin dose in 268 consecutive patients. Diabetes Care 10: 164–169
Meistas MT, Zadik Z, Margolis S, Kowarski A (1981) Correlation of urinary excretion of C-peptide with the integrated concentration and secretion rate of insulin. Diabetes 30: 639–643
Hother-Nielsen O, Faber OK, Sørensen NS, Beck-Nielsen H (1988) Classification of newly diagnosed diabetic patients as insulin-requiring or non-insulin-requiring based on clinical and biochemical variables. Diabetes Care 11: 531–537
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gjessing, H.J., Matzen, L.E., Faber, O.K. et al. Fasting plasma C-peptide, glucagon stimulated plasma C-peptide, and urinary C-peptide in relation to clinical type of diabetes. Diabetologia 32, 305–311 (1989). https://doi.org/10.1007/BF00265547
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00265547