Summary
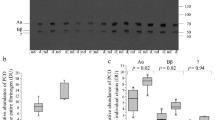
Polymerisation and crosslinking of fibrin monomers was studied in 35 healthy volunteers and in 42 poorly controlled diabetic patients. Polymerisation did not show any difference between control subjects (n = 10) and diabetic patients (n = 11) (p>0.1), although fibrinogen was 35% more glycated in the diabetic patients (p<0.001). Alpha chain crosslinking in the diabetic patients, however, was impaired as is shown from an increase in intermediate alpha polymers with a concomitant decrease in alpha monomer disappearance. A significant positive correlation was found between the degree of glycation of fibrinogen and the defective alpha chain polymerisation (r = 0.86, p<0.005). These results were consistent with the results of thrombin and reptilase experiments. The reaction rate with reptilase did not show any difference between the two groups (p>0.1), whereas the reaction rate with thrombin was significantly slower in the diabetic group compared to the control subjects (p<0.001). Purified fibrin clots obtained from the diabetic patients were more susceptible to plasmin than clots obtained from control subjects. It is concluded that in poorly controlled diabetic patients polymerisation of fibrin monomers is normal, but crosslinking of the alpha chains is impaired, leading to a higher susceptibility of the clots to plasmin degradation.
Article PDF
Similar content being viewed by others
References
Farquahr A, MacDonald MK, Ireland JT (1972) The role of fibrin-depositions in diabetic glomerulosclerosis: a light electron and immunofluorescence microscopy study. J Clin Pathol 25: 657–661
Brownlee M, Vlassara H, Cerami A (1983) Nonenzymatic glycosylation reduces the susceptibility of fibrin to degradation by plasmin. Diabetes 32: 680–684
Jones RL, Jovanovic L, Forman S, Peterson ChM (1984) Time course of reversibility of accelerated fibrinogen disappearance in diabetes mellitus: association with intravascular volume shifts. Blood 63: 22–30
Tsianos EB, Stathakis NE (1980) Soluble fibrin complexes and fibrinogen heterogeneity in diabetes mellitus. Thromb Haemost 44: 130–134
Gurewich V, Lipinska I, Pulini M, Gordon E, Lipinski B (1977) Fibrinogen, fibrinogen heterogeneity and fibrinolytic activity in diabetes mellitus. Thromb Haemost 38: 103
Jones RL, Peterson ChM (1979) Reduced fibrinogen survival in diabetes mellitus. J Clin Invest 63: 485–493
DaSilva SR, Shawe JEH, Patel H, Cudworth AG (1979) Plasma fibrinogen in diabetes mellitus. Diabete Metab 5: 201–206
Wieland OH (1983) Protein modification by nonenzymatic glycosylation: a possible role in the development of diabetic complications. Mol Cell Endocrinol 29: 125–131
McVerry BA, Thorpe S, Joe F, Gaffney P, Huehns ER (1981) Non enzymatic glycosylation of fibrinogen. Haemostasis 10: 261–270
Lütjens A, te Velde AA, v d Veen EA, v d Meer J (1985) Glycosylation of human fibrinogen in vivo. Diabetologia 28: 87–89
Lorand L, Ong HH, Lipinski B, Rule HG, Downey J, Jacobsen A (1966) Lysine as amine donor in fibrin crosslinking. Biochem Biophys Res Comm 25: 629–637
WHO Study Group Diabetes Mellitus (1985) Technical Report Series 727. World Health Organisation, Geneva, Switzerland
Belitser VA, Varetskaja TV, Malneva GV (1968) Fibrinogen-fibrin interaction. Biochim Biophys Acta 154: 367–369
Olexa SA, Budzynski AZ (1980) Evidence for four different polymerisation sites involved in human fibrin formation. Proc Natl Acad Sci USA 77: 1374–1378
McDonagh J, Messel H, McDonagh RP jr, Murano G, Blomback B (1972) Molecular weight analysis of fibrinogen and fibrin chains by an improved sodium dodecylsulfate gel electrophoresis method. Biochim Biophys Acta 257: 135–142
Owren PA, Aas K (1951) The control fo dicoumarol therapy and the quantitative determination of prothrombin and proconvertin. Scand J Clin Lab Invest 3: 201–208
Deutsch DG, Mertz ET (1970) Plasminogen. Purification from human plasma by affinity chromatography. Science 170: 1095–1096
Strengers Th, Asberg EGMTh (1963) Een screeningtest, gevolgd door een snelle kwantitatieve microbepaling van fibrinogeen in plasma. Ned Tijdschr Geneeskd 107: 2044–2045
Kendall MG (1970) Rank correlation methods, 4th edn. Griffin, London
Bell WR (1982) Defibrinating enzymes. In: Colman RW, Hirsch J, Marder VJ, Salzman EW (eds) Hemostasis and thrombosis. Lippincott, Philadelphia, pp 1013–1027
Kloczko J, Wojtukiewicz M, Dielawiec M, Larzycka B, Kinalska J (1986) Plasma factor XIII and some other haemostasis parameters in patients with diabetic angiopathy. Acta Haematol (Basel) 76: 81–85
Kirkpatrick JP, McIntire LV, Moake JL (1978) Streptokinase-induced degradation of crosslinks and uncrosslinked clots. Thromb Res 13: 569–575
Francis CW, Marder VJ, Barlow G (1980) Plasmic degradation of corsslinked fibrin. J Clin Invest 66: 1033–1043
Geiger M, Binder BR (1986) Non enzymatic glucosylation as a contributing factor to defective fibrinolysis in diabetes mellitus. Haemostasis 16: 439–446
Brownlee M, Vlassara H, Cerami A (1983) Non enzymatic glycosylation reduces the susceptibility of fibrin to degradation by plasmin. Diabetes 32: 680–684
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lütjens, A., Jonkhoff-Slok, T.W., Sandkuijl, C. et al. Polymerisation and crosslinking of fibrin monomers in diabetes mellitus. Diabetologia 31, 825–830 (1988). https://doi.org/10.1007/BF00277485
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00277485