Summary
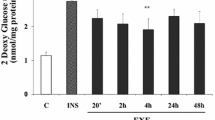
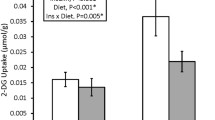
We investigated the influence of altered glucose levels on insulin-stimulated 3-0-methylglucose transport in isolated skeletal muscle obtained from NIDDM patients (n=13) and non-diabetic subjects (n=23). Whole body insulin sensitivity was 71% lower in the NIDDM patients compared to the non-diabetic subjects (p <0.05), whereas, insulin-mediated peripheral glucose utilization in the NIDDM patients under hyperglycaemic conditions was comparable to that of the non-diabetic subjects at euglycaemia. Following a 30-min in vitro exposure to 4 mmol/l glucose, insulin-stimulated 3-0-methylglucose transport (600 pmol/l insulin) was 40% lower in isolated skeletal muscle strips from the NIDDM patients when compared to muscle strips from the non-diabetic subjects. The impaired capacity for insulin-stimulated 3-0-methylglucose transport in the NIDDM skeletal muscle was normalized following prolonged (2 h) exposure to 4 mmol/l, but not to 8 mmol/l glucose. Insulin-stimulated 3-0-methylglucose transport in the NIDDM skeletal muscle exposed to 8 mmol/l glucose was similar to that of the non-diabetic muscle exposed to 5 mmol/l glucose, but was decreased by 43% (p <0.01) when compared to non-diabetic muscle exposed to 8 mmol/l glucose. Despite the impaired insulin-stimulated 3-0-methylglucose transport capacity demonstrated by skeletal muscle from the NIDDM patients, skeletal muscle glycogen content was similar to that of the non-diabetic subjects. Kinetic studies revel a Km for 3-0-methylglucose transport of 9.7 and 8.8 mmol/l glucose for basal and insulin-stimulated conditions, respectively. In conclusion, the impaired capacity for insulinstimulated glucose transport in skeletal muscle from patients with NIDDM appears to protect the cell from excessive glucose uptake under hyperglycaemic conditions. Furthermore, the in vitro normalization of the decreased insulin-stimulated glucose transport in NIDDM skeletal muscle following exposure to 4 mmol/l glucose suggests that glycaemia per se has a profound effect on the regulation of muscular glucose transport.
Article PDF
Similar content being viewed by others
Abbreviations
- NIDDM:
-
Non-insulin-dependent diabetes mellitus
- KHB:
-
Krebs-Henseleit bicarbonate buffer
- BSA:
-
bovine serum albumin
- ANOVA:
-
analysis of variance
- GLUT 4:
-
insulin regulated glucose transporter
References
Reaven GM, Olefsky JM (1978) The role of insulin resistance in the pathogenesis of diabetes mellitus. Adv Metab Res 9: 313–331
DeFronzo RA, Bonadonna RC, Ferrannini E (1992) Pathogenesis of NIDDM. A balanced overview. Diabetes Care 15: 318–368
Dohm GL, Tapscott EB, Pories WJ et al. (1988) An in vitro human muscle preparation suitable for metabolic studies. J Clin Invest 82: 486–494
Andréasson K, Galuska D, Thörne A, Sonnenfeld T, Wallberg-Henriksson H (1991) Decreased insulin-stimulated 3-0-methylglucose transport in in vitro incubated muscle strips from type II diabetic subjects. Acta Physiol Scand 142: 255–260
De Fronzo RA, Gunnarsson R, Björkman O, Olsson M, Wahren J (1985) Effects of insulin on peripheral and splanchnic glucose metabolism in noninsulin-dependent (type II) diabetes mellitus. J Clin Invest 76: 149–155
Rossetti L, Giaccari A, DeFronzo RA (1990) Glucose toxicity. Diabetes Care 13: 610–630
Sasson S, Edelson D, Cerasi E (1987) In vitro autoregulation of glucose utilization in rat soleus muscles. Diabetes 36: 1041–1046
Richter EA, Hansen BF, Hansen SA (1988) Glucose-induced insulin resistance of skeletal-muscle glucose transport and uptake. Biochem J 252: 733–737
Sasson S, Cerasi E (1986) Substrate regulation of the glucose transport system in rat skeletal muscle. J Biol Chem 261: 16827–16833
Yki-Järvinen H, Helve E, Koivisto VA (1987) Hyperglycemia decreases glucose uptake in type I diabetes. Diabetes 36: 892–896
Unger RH, Grundy S (1985) Hyperglycemia as an inducer as well as a consequence of impaired islet cell function and insulin resistance: implications for the management of diabetes. Diabetologia 28: 119–121
Kahn BB, Schulman GI, DeFronzo RA, Cushman SW, Rossetti L (1991) Normalization of blood glucose in diabetic rats with phlorizin treatment reverses insulin-resistant glucose transport in adipose cells without restoring glucose transporter gene expression. J Clin Invest 87: 561–570
Scarlett JA, Kolterman OG, Ciaraldi TP, Kao M, Olefsky JM (1983) Insulin treatment reverses the postreceptor defect in adipocyte 3-0-methylglucose transport in type II diabetes mellitus. J Clin Endocrinol Metab 56: 1195–1201
Foley JE, Kashiwagi A, Verso MA, Reaven G, Andrews J (1983) Improvement in in vitro insulin action after one month of insulin therapy in obese noninsulin-dependent diabetics. J Clin Invest 72: 1901–1909
Hollund E, Pedersen O, Sorensen NS (1987) Adipocyte insulin binding and action in moderately obese NIDDM patients after dietary control of plasma glucose: reversal of post-binding abnormalities. Diabetes Care 10: 306–312
Zierath JR, Galuska D, Engström Å et al. (1992) Human islet amyloid polypeptide at pharmacological levels inhibits insulin and phorbol ester-stimulated glucose transport in in vitro incubated human muscle strips. Diabetologia 35: 26–31
Bergman RN, Ider YZ, Bowden CR, Cobelli C (1979) Quantitative estimation of insulin sensitivity. Am J Physiol 236: E667-E677
DeFronzo RA, Tobin JD, Andres R (1979) Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 237: E214-E223
Henriksson KG (1979) Semi-open muscle biopsy technique. Acta Neurol Scand 59: 317–323
Vinten J (1978) Cytochlasin B inhibition and temperature dependence of 3-0-methylglucose transport in fat cells. Biochim Biophy Acta 511: 259–273
Wallberg-Henriksson H, Nie Z, Henriksson J (1987) Reversibility of decreased insulin-stimulated glucose transport capacity in diabetic muscle with in vitro incubation: insulin is not required. J Biol Chem 262: 7665–7671
Lowery, OH, Passonneau JV (1972) A flexible system of enzymatic analysis. Academic Press, New York
Bourey RE, Koranyi L, James DE, Mueckler M, Permutt MA (1990) Effects of altered glucose homeostasis on glucose transporter expression in skeletal muscle of the rat. J Clin Invest 86: 542–547
James DE, Studelska DR, Rodnick KJ (1992) Glucose transporter gene expression in muscle. In: Devlin JT, Horton ES, Vranic M (eds) Diabetes mellitus and exercise. Smith-Gordon, London, pp 45–54
Ramlal T, Rastogi S, Vranic M, Klip A (1989) Decrease in glucose transporter number in skeletal muscle of mildly diabetic (streptozotocin-treated) rats. Endocrinology 125: 890–897
Handberg A, Vaag A, Damsbo P, Beck-Nielsen H, Vinten J (1990) Expression of insulin-regulatable glucose transporters in skeletal muscle from type 2 (non-insulin-dependent) diabetic patients. Diabetologia 33: 625–627
Pedersen O, Bak JF, Andersen PH et al. (1990) Evidence against altered expression of GLUT1 or GLUT4 in skeletal muscle of patients with obesity or NIDDM. Diabetes 39: 865–870
Garvey WT, Maianu L, Hancock JA, Golichowski AM, Baron A (1992) Gene expression of GLUT4 in skeletal muscle from insulin-resistant patients with obesity, IGT, GDM, and NIDDM. Diabetes 41: 465–475
Vogt B, Mühlbacher C, Carrascosa J et al. (1992) Subcellular distribution of GLUT 4 in the skeletal muscle of lean type 2 (non-insulin-dependent) diabetic patients in the basal state. Diabetologia 35: 456–463
Vaag A, Damsbo P, Hother-Nielsen O, Beck-Nielsen H (1992) Hyperglycaemia compensates for the defect in insulin-mediated glucose metabolism and in the activation of glycogen synthase in the skeletal muscle of patients with type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 35: 80–88
Revers RR, Fink R, Griffin J, Olefsky JM, Kolterman OG (1984) Influence of hyperglycemia on insulin's in vivo effects in type II diabetes. J Clin Invest 73: 664–672
Kelley DE, Mandarino LJ (1990) Hyperglycemia normalizes insulin-stimulated skeletal muscle glucose oxidation and storage in noninsulin-dependent diabetes mellitus. J Clin Invest 86: 1999–2007
Gottesman I, Mandarino L, Verdonk C, Rizza R, Gerich J (1982) Insulin increases the maximum velocity for glucose uptake without altering the Michaelis constant in man. Evidence that insulin increases glucose uptake merely by providing additional transport sites. J Clin Invest 70: 1310–1314
Laakso M, Edelman SV, Olefsky JM, Brechtel G, Wallace P, Baron AD (1990) Kinetics of in vivo muscle insulin-mediated glucose uptake in human obesity. Diabetes 39: 965–974
Edelman SV, Laakso M, Wallace P, Brechtel G, Olefsky JM, Baron AD (1990) Kinetics of insulin-mediated and non-insulin-mediated glucose uptake in humans. Diabetes 39: 955–964
Fink RI, Wallace P, Brechtel G, Olefsky JM (1992) Evidence that glucose transport is rate-limiting for in vivo glucose uptake. Metabolism 41: 897–902
Laakso M, Edelman SV, Brechtel G, Baron AD (1992) Impaired insulin-mediated skeletal muscle blood flow in patients with NIDDM. Diabetes 41: 1076–1083
Boström M, Nie Z, Goertz G, Henriksson J, Wallberg-Henriksson H (1989) Indirect effect of catecholamines on development of insulin resistance in skeletal muscle from diabetic rats. Diabetes 38: 906–910
Laakso M, Edelman SV, Brechtel G, Baron A (1992) Effects of epinephrine on insulin-mediated glucose uptake in whole body and leg muscle in humans: role of blood flow. Am J Physiol 263: E199-E204
Nesher R, Karl I, Kipnis DM (1985) Dissociation of effects of insulin and contraction on glucose transport in rat epitrochlearis muscle. Am J Physiol 249: C226-C232
Karl IE, Gavin JR III, Levy J (1990) Effect of insulin on glucose utilization in epitrochlearis muscle of rats with streptozotocin-induced NIDDM. Diabetes 39: 1106–1115
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zierath, J.R., Galuska, D., Nolte, L.A. et al. Effects of glycaemia on glucose transport in isolated skeletal muscle from patients with NIDDM: in vitro reversal of muscular insulin resistance. Diabetologia 37, 270–277 (1994). https://doi.org/10.1007/BF00398054
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00398054