Abstract
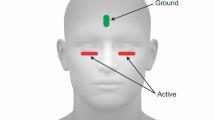
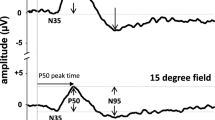
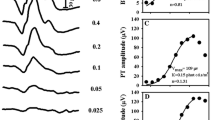
Pattern electroretinograms were recorded to phase-reversing checkerboard stimuli with DTL electrodes under conditions close to those of the ISCEV pattern electroretinogram guidelines. Both transient (2 reversals/s) and steady-state (16 reversals/s) stimulation was used. The check sizes were 0.4°, 0.8° and 16°; the mean luminance 45 cd/m2, the contrast 98%, and the field size 32°×27°. In 42 eyes of 21 subjects, measurements were repeated at the same time of day after 1 week. For each eye, the intersession coefficient of variation was calculated as a measure of reproducibility. We found a coefficient of variation (±standard deviation) of 7%±5% for the amplitude of the steady-state pattern electroretinogram, 9.5%±7% for the transient pattern electroretinogram and 1.5%±2% for the latency of the transient pattern electroretinogram. To assess the diurnal variability, during a 15-h period, three pattern electroretinograms were recorded in 10 subjects. No relationship was found between the P50 latency and the time of day. However, the mean amplitude showed a maximum in the morning (9∶30 am) and a minimum in the afternoon (2∶30 pm). This small effect (about 7%, p<0.001) was more pronounced for N95 and steady-state amplitudes than for P50 amplitudes (p<0.01). Diurnal contributions to the pattern electroretinogram ranged between 3% and 10%. We conclude that pattern electroretinogram amplitude reproduces within ±10% with a probability of 70%. The effect size of the diurnal variation is similar and might be relevant for longitudinal studies.
Similar content being viewed by others
References
Bach M, Gerling J, Geiger K. Optic atrophy reduces the pattern-electroretinogram for both fine and coarse stimulus patterns. Clin Vision Sci 1992; 7: 32–33.
Falsini B, Bardocci A, Porciatti V, Bolzani R, Piccardi M. Macular dysfunction in multiple sclerosis revealed by steady-state flicker and pattern ERGs. Electroencephalogr Clin Neurophysiol 1992; 82: 53–9.
Holder GE. Significance of abnormal pattern electroretinography in anterior visual pathway dysfunction. Br J Ophthalmol 1987; 71: 166–71.
Trick GL. Pattern evoked retinal and cortical potentials in diabetic patients. Clin Vision Sci 1991; 6: 209–17.
Zrenner E, Ziegler R, Voss B. Clinical application of pattern electroretinography: melanoma, retinal detachment and glaucoma. Doc Ophthalmol 1988; 68: 283–92.
Bach M, Pfeiffer N, Birkner-Binder D. Pattern-electroretinogram reflects diffuse retinal damage in early glaucoma. Clin Vision Sci 1992; 7: 335–40.
Korth M, Horn F, Storck B, Jonas J. The pattern-evoked electroretinogram (PERG): age-related alterations and changes in glaucoma. Graefes Arch Clin Exp Ophthalmol 1989; 227: 123–30.
Pfeiffer N, Tillmon B, Bach M. Predictive value of the pattern electroretinogram in high-risk ocular hypertension. Invest Ophthalmol Vis Sci 1993; 34: 1710–15.
Bartel P, Becker P, Robinson E. The intrasession repeatability of pattern electroretinograms and the effects of digital filtering. Doc Ophthalmol 1991; 76: 351–8.
Holopigian K, Snow J, Seiple W, Siegel I. Variability of the pattern electroretinogram. Doc Ophthalmol 1988; 70: 103–15.
Jacobi PC, Walter P, Brunner R, Kriegelstein GK. Reproducibility and intraindividual variability of the pattern electroretinogram. Ger J Ophthalmol 1994; 3: 216–9.
Odom VJ, Holder GE, Feghali JG, Cavender S. Pattern electroretinogram intrasession reliability: a two center comparison. Clin Vision Sci 1992; 7: 263–81.
Prager TC, Saad N, Schweitzer C, Garcia C, Arden GB. Electrode comparison in pattern electroretinography. Invest Ophthalmol Vis Sci 1992; 33: 390–4.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986; 1: 307–310.
Marmor MF, Holder GE, Porciatti V, Trick GL, Zrenner E. Guidelines for basic pattern electroretinography: recommendations by the International Society for Clinical Electrophysiology of Vision. Doc Ophthalmol 1996; 91: 291–8.
Buget A, Py P, Romanet JP. 24-hour (nyctohemeral) and sleep-related variations of intraocular pressure in healthy white individuals. Am J Ophthalmol 1994; 117: 342–7.
La Vail MM. Rod outer segment disk shedding in rat retina: relationship to cyclic lighting. Silence 1976; 194: 1071–3.
Roenneberg T, Lotze M, von Steinbüchel N. Diurnal variation in human visual sensitivity determined by incremental thresholds. Clin Vision Sci 1992; 2: 83–91.
Birch DG, Berson EL, Sandberg MA. Diurnal rhythm in the human rod ERG. Invest Ophthalmol Vis Sci 1984; 25: 236–8.
Wirz-Justice A, Da Prada M, Remé C. Circadian rhythm in rat retinal dopamine. Neurosci Lett 1984; 45: 21–5.
Masson G, Mestre D, Blin O. Dopaminergic modulation of visual sensitivity in man. Fundam Clin Pharmacol 1993; 7: 449–63.
Bach M. The “Freiburg Visual Acuity Test”: automatic measurement of the visual acuity. Optom Vision Sci 1996; 73: 49–53.
Bach M, Meigen T, Strasburger H. Raster-scan cathode ray tubes for vision research: limits of resolution in space, time and intensity, and some solutions. Spatial Vision 1997; 10:403–14.
Dawson WW, Trick GL, Litzkow CA. Improved electrode for electroretinography. Invest Ophthalmol Vis Sci 1979; 18: 988–91.
Thompson DA, Drasdo N. An improved method for using the DTL fibre in electroretinography. Ophthalmic Physiol Opt 1987; 7: 315–9.
Ederer F. Shall we count numbers of eyes or numbers of subjects? Arch Ophthalmol 1973; 89: 1–2.
Katz J, Zeger S, Liang KY. Appropriate statistical methods to account for similarities in binary outcome between fellow eyes. Invest Ophthalmol Vis Sci 1994; 35: 2461–5.
Bach M, Speidel-Fiaux A. Pattern electroretinogram in glaucoma and ocular hypertension. Doc Ophthalmol 1989; 73: 173–81.
Padovano S, Falsini B, Ciavarella P, Moretti G, Porciatti V. Spatial-temporal interactions in the steady-state pattern electroretinogram. Doc Ophthalmol 1996; 90: 169–76.
Shors TJ, Ary JP, Eriksen K, Wright KW. P100 amplitude variability of the pattern visual evoked potential. Electroencephalogr Clin Neurophysiol 1986; 65: 316–9.
Oken BS, Chiappa KH, Gill E. Normal temporal variability of the P100. Electroencephalogr Clin Neurophysiol 1987; 66: 153–6.
Fagan JE, Allen RG, Yolton RL. Factors contributing to the amplitude variability of steady-state visual evoked response. Am J Optom Physiol Opt 1984; 61: 453–64.
Berninger TA, Arden GB. The pattern electroretinogram. Eye 1988; 2: 257–83.
Weinstein GW, Arden GB, Hitchings RA, Ryan S, Calthorpe CM, Odom V. The pattern electroretinogram (PERG) in ocular hypertension and glaucoma. Arch Ophthalmol 1988; 106: 923–8.
Fujikado T. The effect of dopamine on the response to pattern stimulation: study of the chick ERG. Jpn J Ophthalmol 1994; 38: 368–74.
Jacobi PC, Rüther K, Miliczek KD, Völker M, Zrenner E. Klinische Elektroretinographie: Standardprotokoll und Normwerte. Klin Monatsbl Augenheilkd 1993; 202: 27–42.
Degaute JP, van de Borne P, Linkowski P, van Cauter E. Quantitative analysis of the 24-hours blood pressure and heart rate patterns in young men. Hypertension 1991; 18: 199–200.
Kremmer S, Tolksdorf-Kremmer A, Stodtmeister R. Simultaneous registration of VECP and pattern ERG during artificially raised intraocular pressure. Ophthalmologica 1995; 209: 233–41.
Siliprandi R, Bucci MG, Canella R, Carmignoto G. Flash and pattern electroretinograms during and after acute intraocular pressure elevation in cats. Invest Ophthalmol Vis Sci 1988; 29: 558–65.
Bach M, Holder GE. Check size tuning of the pattenr-ERG: a reappraisal. Doc Ophthalmol 1996; 92: 193–202.
Arden GB, Hogg CR, Holder GE. Gold foil electrodes: a two-center study of electrode reliability. Doc Ophthalmol 1994; 86: 275–84.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Otto, T., Bach, M. Retest variability and diurnal effects in the pattern electroretinogram. Doc Ophthalmol 92, 311–323 (1996). https://doi.org/10.1007/BF02584085
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02584085