Abstract
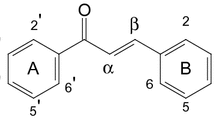
The methanol extract obtained from the aerial parts ofAceriphyllum rossii (Saxifragaceae) was fractionated into ethyl acetate (EtOAc),n-BuOH and H2O layers through solvent fractionation. Repeated silica gel column chromatography of EtOAc andn-BuOH layers afforded six flavonol glycosides. They were identified as kaempferol 3-O-β-D-glucopyranoside (astragalin,1), quercetin 3-O-β-D-glucopyranoside (isoquercitrin,2), kaempferol 3-O-α-L-rhamnopyranosyl (1→6)-β-D-glucopyranoside (3), quercetin 3-O-α-L-rhamnopyranosyl (1→6)-β-D-glucopyrano-side (rutin,4), kaempferol 3-O-[α-L-rhamnopyranosyl (1→4)-α-L-rhamnopyranosyl (1→6)-β-D-glucopyranoside] (5) and quercetin 3-O-[α-L-rhamnopyranosyl (1→4)-α-L-rhamnopyranosyl (1→6)-β-D-glucopyranoside] (6) on the basis of several spectral data. The antioxidant activity of the six compounds was investigated using two free radicals such as the ABTS free radical and superoxide anion radical. Compound1 exhibited the highest antioxidant activity in the ABTS2,2-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) radical scavenging method. 100 mg/L of compound1 was equivalent to 72.1±1.4 mg/L of vitamin C, and those of compounds3 and5 were equivalent to 62.7±0.5 mg/L and 54.3±1.3 mg/L of vitamin C, respectively. And in the superoxide anion radical scavenging method, compound5 exhibited the highest activity with an IC50 value of 17.6 ± 0.3 μM. In addition, some physical and spectral data of the flavonoids were confirmed.
Similar content being viewed by others
References
Arora, A., Nair, M. G., and Strasburg, G. M., Structure-activity relationships for antioxidant activities of a series of flavonoids in a liposomal system.Free Radic. Biol. Med., 24, 1355–1363 (1998).
Beck, M. A. and Häberlein, H., Flavonol glycoside fromEschscholtzia califormica.Phytochemistry, 50, 329–332 (1999).
Cao, G., Sofic, E., and Prior, R. L., Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships.Free Radic. Biol. Med., 22, 749–760 (1997).
Carini, R., Poli, G., Diazini, M. U., Maddix, S. P., Slater, T. F., and Cheesman K. H., Comparative evaluation of the antioxidant activity of α-tocopherol, α-tocopherol polyethylene glycol 1000 succinate and a-tocopherol succinate in isolated hepatocytes and liver microsomal suspensions.Biochem. Pharmacol., 39, 1597–1601 (1990).
Choi, W. H., Park, W. Y., Hwang, B. Y., Oh, G.-J., Kang, S. J., Lee, K. S., and Ro, J. S., Phenolic compounds from the stem bark ofCornus walteri Wagner.Kor. J. Pharmacog., 29(3), 217–224 (1998).
Cholbi, M. R., Paya, M., and Alcaraz, M. J., Inhibitory effects of phenolic compounds on CCI4-induced microsomal lipid peroxidation.Experientia, 47, 195–199 (1991).
Donovan, J. L., Meyer, A. S., and Waterhouse, A. L., Phenolic composition and antioxidant activity of prunes and prune juice (Prunus domestica).J. Agric. Food Chem., 1247–1252 (1998).
Duthie, S. J., Collins, G. G., and Dobson, V. L., Quercetin and myricetin protect against hydrogen peroxide-induced DNA damage (strand breaks and oxidized pyrimidines) in human lymphocytes.Mutation Research, 393, 223–231 (1997).
Gey, K. F., Prospects for the prevention of free radical disease, regarding cancer and cardiovascular disease.British Med. Bull., 49, 679–699 (1993).
Han, J. T., Kim, H. Y., Park, Y. D., Lee, Y. H., Lee, K. R., Kwon, B. M., and Baek, N. I., Aceriphyllic acid A, a new ACAT inhibitory triterpenoid, fromAceriphyllum rossii.Planta Medica, 68, 558–561 (2002).
Ho, H. M., Chen, R. Y., Leung, L. K., Cen, F. L., Huang, Y. and Chen, Z.-Y., Difference in flavonoid and isoflavone profile between soybean and soy leaf.Biomed. Pharmacother., 56, 289–295 (2002).
Jung, H. A., Kim, A. R., Chung, H. Y., and Choi, T. S.,In vitro antioxidant activity of some selected prunus species in Korea.Arch. Pharm. Res., 25, 825–827 (2002).
Kim, D. O., Lee, K. W., Lee, H. J., and Lee, C. Y., Vitamin C equivalent antioxidant capacity (VCEAC) of phenolic phytochemicals.J. Agric. Food Chem., 50, 3713–3717 (2002).
Kim, K. H. and Kim, M. N., Constituents ofCathamiflos.Yakhak Hoeji, 36, 556–562 (1992).
Kim, S. Y., Gao, J. J., Lee, W. C., Ruy, K. S., Lee, K. R., and Kim, Y. C., Antioxidative flavonoids from the leaves ofMorus alba.Arch. Pharm, Res., 22, 81–85 (1999).
Kweon, M. H., Hwang, H. J., and Sung, H. C., Identification and antioxidant activity of novel chlorogenic acid derivatives from bamboo (Phyllostachys edulis).J. Agric. Food Chem., 49, 4546–4655 (2001).
Liu, Y., Wu, Y., Ji, K. C., Hou, A., Yoshida, T., and Okuda, T., Astragalin 2″,6″-di-O-gallate fromLoropetalum chinense.Phytochemistry, 46(3), 389–391 (1997).
Nuutila, A. M., Puupponen-Pimia, R., Aarni, M., and Oksman-Caldentey, K. M., Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity.Food Chemistry, 80, 1–9 (2003).
Ohshima, H., Yoshie, Y., Auriol, S., and Gilibert, I., Antioxidant and pro-oxidant actions of flavonoids: effects on DNA damage induced by nitric oxide, peroxynitrite and nitroxyl anion.Free Radic. Biol. Med., 25, 1057–1065 (1998).
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M. and Rice-Evans, C., Antioxidnat activity applying an improved ABTs radical cation decolorization assay.Free Radic. Biol. Med., 26, 1231–1237 (1999).
Rice-Evans, C. A., Miller, N. J., and Paganga, G., Structure-antioxidant activity relationships of flavonoids and phenolic acids.Free Radic. Biol. Med., 20, 933–956 (1996).
Rice-Evans, C. A., Miller, N. J., and Paganga, G., Antioxidant properties of phenolic compounds.Trends in Plant Science, 2, 152–159 (1997).
Seto, T., Yasuda, I., and Akiyama, K., Purgative activity and principals of the fruits ofRosa multiflora andR. wichuraiana.Chem. Pharm. Bull., 40, 2080–2082 (1992).
Shin-Kim, J. S., Kim, H. J., and Park, H. K., Studies on the chemical constituents ofLysimachia clethroides.Yakhak Hoeji, 37, 325–330 (1993).
Webby, R. F. and Markham, K. R., Flavonol 3-O-triglycosides fromActinidia species.Phytochemistry, 29, 289–292 (1990).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, JT., Bang, MH., Chun, OK. et al. Flavonol glycosides from the aerial parts ofAceriphyllum rossii and their antioxidant activities. Arch Pharm Res 27, 390–395 (2004). https://doi.org/10.1007/BF02980079
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02980079