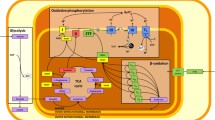
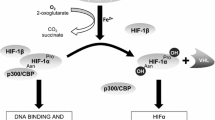
Abstract
Hypoxia refers to environmental or clinical settings that potentially threaten tissue oxygen homeostasis. One unique aspect of skeletal muscle is that, in addition to hypoxia, oxygen balance in this tissue may be further compromised when exercise is superimposed on hypoxia. This review focuses on the cellular and molecular responses of human skeletal muscle to acute and chronic hypoxia, with emphasis on physical exercise and training. Based on published work, it is suggested that hypoxia does not appear to promote angiogenesis or to greatly alter oxidative enzymes in skeletal muscle at rest. Although the HIF-1 pathway in skeletal muscle is still poorly documented, emerging evidence suggests that muscle HIF-1 signaling is only activated to a minor degree by hypoxia. On the other hand, combining hypoxia with exercise appears to improve some aspects of muscle O2 transport and/or metabolism.
Similar content being viewed by others
References
Niermeyer S, Zamudio S, Moore LG (2001) The people. In: Hornbein TF, Schoene RB (eds) High altitude, an exploration of human adaptation. Marcel Dekker, New York, pp 43–100
Harris AL (2002) Hypoxia—a key regulatory factor in tumour growth. Nat Rev Cancer 2:38–47
Richardson RS, Noyszewski EA, Kendrick KF, Leigh JS, Wagner PD (1995) Myoglobin O2 desaturation during exercise. Evidence of limited O2 transport. J Clin Invest 96:1916–1926
Wang GL, Semenza GL (1995) Purification and characterization of hypoxia-inducible factor 1. J Biol Chem 270:1230–1237
Ke Q, Costa M (2006) Hypoxia-inducible factor-1 (HIF-1). Mol Pharmacol 70:1469–1480
Huang LE, Bunn HF (2003) Hypoxia-inducible factor and its biomedical relevance. J Biol Chem 278:19575–19578
Semenza GL (2002) Signal transduction to hypoxia-inducible factor 1. Biochem Pharmacol 64:993–998
Dm Stroka, Tobi Burkhardt, Isab Desbaillets, Rh Wenger, Dah Neil, Chri Bauer, Max Gassmann, Dani Candinas (2001) HIF-1 is expressed in normoxic tissue and displays an organ-specific regulation under systemic hypoxia. FASEB J 15:2445–2453
Ameln H, Gustafsson T, Sundberg CJ, Okamoto K, Jansson E, Poellinger L, and Makino Y (2005) Physiological activation of hypoxia inducible factor-1 in human skeletal muscle. FASEB J, 04-2304fje
Pisani DF, Dechesne CA (2005) Skeletal Muscle HIF-1α expression is dependent on muscle fiber type. J Gen Physiol 126:173–178
Chavez JC, Agani F, Pichiule P, LaManna JC (2000) Expression of hypoxia-inducible factor-1alpha in the brain of rats during chronic hypoxia. J Appl Physiol 89:1937–1942
Vigano A, Ripamonti M, De Palma S, Capitanio D, Vasso M, Wait R, Lundby C, Cerretelli P, Gelfi C (2008) Proteins modulation in human skeletal muscle in the early phase of adaptation to hypobaric hypoxia. Proteomics 8:4668–4679
Robach P, Cairo G, Gelfi C, Bernuzzi F, Pilegaard H, Vigano A, Santambrogio P, Cerretelli P, Calbet JAL, Mouterou S, Lundby C (2007) Strong iron demand during hypoxia-induced erythropoiesis is associated with down-regulation of iron-related proteins and myoglobin in human skeletal muscle. Blood 109:4724–4731
Mason SD, Howlett RA, Kim MJ, Olfert IM, Hogan MC, McNuly W, Hickey RP, Wagner PD, Kahn CR, Giordano FJ, Johnson RS (2004) Loss of skeletal muscle HIF-1alpha results in altered exercise endurance. PLoS Biol 2:e288
Hoppeler H, Howald H, Conley K, Lindstedt SL, Claassen H, Vock P, Weibel ER (1985) Endurance training in humans: aerobic capacity and structure of skeletal muscle. J Appl Physiol 59:320–327
Lundby C, Gassmann M, Pilegaard H (2005) Regular endurance training reduces the exercise induced HIF-1a and HIF-2a mRNA expression in human skeletal muscle in normoxic conditions. Eur J Appl Physiol 96:363–369
Mason SD, Johnson RS (2007) The role of HIF-1 in hypoxic response in the skeletal muscle. Adv Exp Med Biol 618:229–244
Mason SD, Rundqvist H, Papandreou I, Duh R, McNulty WJ, Howlett RA, Olfert IM, Sundberg CJ, Denko NC, Poellinger L, Johnson RS (2007) HIF-1α in endurance training: suppression of oxidative metabolism. Am J Physiol Regul Integr Comp Physiol 293:R2059–R2069
Vogt M, Puntschart A, Geiser J, Zuleger C, Billeter R, Hoppeler H (2001) Molecular adaptations in human skeletal muscle to endurance training under simulated hypoxic conditions. J Appl Physiol 91:173–182
Zoll J, Ponsot E, Dufour S, Doutreleau S, Ventura-Clapier R, Vogt M, Hoppeler H, Richard R, Fluck M (2006) Exercise training in normobaric hypoxia in endurance runners. III. Muscular adjustments of selected gene transcripts. J Appl Physiol 100:1258–1266
Reynafarje B (1962) Myoglobin content and enzymatic activity of muscle and altitude adaptation. J Appl Physiol 17:301–305
Jackson CG, Sillau AH, Banchero N (1987) Fiber composition and capillarity in growing guinea pigs acclimated to cold and cold plus hypoxia. Proc Soc Exp Biol Med 185:101–106
Lundby C, Pilegaard H, Andersen JL, van Hall G, Sander M, Calbet JAL (2004) Acclimatization to 4,100 m does not change capillary density or mRNA expression of potential angiogenesis regulatory factors in human skeletal muscle. J Exp Biol 207:3865–3871
Mizuno M, Savard GK, Areskog NH, Lundby C, Saltin B (2008) Skeletal muscle adaptations to prolonged exposure to extreme altitude: a role of physical activity? High Alt Med Biol 9:311–317
Hoppeler H, Kleinert E, Schlegel C, Claassen H, Howald H, Kayar SR, Cerretelli P (1991) Morphological adaptations of human skeletal muscle to chronic hypoxia. Int J Sports Med 11:S3–S9
MacDougall JD, Green HJ, Sutton JR, Coates G, Cymerman A, Young P, Houston CS (1991) Operation Everest II: structural adaptations in skeletal muscle in response to extreme simulated altitude. Acta Physiol Scand 142:427
Jagoe RT, Engelen MPKJ (2003) Muscle wasting and changes in muscle protein metabolism in chronic obstructive pulmonary disease. Eur Respir J 22:52s–563
Kayser B (1992) Nutrition and high altitude exposure. Int J Sports Med 13:S129–S132
Gelfi C, De Palma S, Ripamonti M, Wait R, Eberini I, Bajracharya A, Marconi C, Schneider A, Hoppeler H, and Cerretelli P (2004) New aspects of altitude adaptation in Tibetans: a proteomic approach. FASEB J, DOI:10.1096/fj.03-1077fje
Juel C, Lundby C, Sander M, Calbet JAL, van Hall G (2003) Human skeletal muscle and erythrocyte proteins involved in acid–base homeostasis: adaptations to chronic hypoxia. J Physiol 15:639–648
Green H, Roy B, Grant S, Burnett M, Tupling R, Otto C, Pipe A, McKenzie D (2000) Downregulation in muscle Na+-K+-ATPase following a 21-day expedition to 6,194 m. J Appl Physiol 88:634–640
Hochachka PW, Buck LT, Doll CJ, Land SC (1996) Unifying theory of hypoxia tolerance: molecular/metabolic defense and rescue mechanisms for surviving oxygen lack. Proc Natl Acad Sci USA 93:9493–9498
Lundby C, Calbet JAL, Sander M, van Hall G, Mazzeo RS, Stray-Gundersen J, Stager JM, Chapman RF, Saltin B, Levine BD (2007) Exercise economy does not change after acclimatization to moderate to very high altitude. Scand J Med Sci Sports 17:281–291
Pilegaard H, Saltin B, Neufer PD (2003) Exercise induces transient transcriptional activation of the PGC-1alpha gene in human skeletal muscle. J Physiol 546:851–858
Ponsot E, Dufour SP, Zoll J, Doutrelau S, N’Guessan B, Geny B, Hoppeler H, Lampert E, Mettauer B, Ventura-Clapier R, Richard R (2006) Exercise training in normobaric hypoxia in endurance runners. II. Improvement of mitochondrial properties in skeletal muscle. J Appl Physiol 100:1249–1257
Masuda KOK, Kuno S, Asano K, Shimojo H, Katsuna S (2001) Endurance training under 2,500-m hypoxia does not increase myoglobin content in human skeletal muscle. Eur J Appl Physiol 85:486–490
Terrados N, Melinchna J, Sylven C, Jansson E, Kaijser L (1988) Effects of training at simulated altitude on performance and muscle metabolic capacity in competitive road cyclists. Eur J Appl Physiol 57:203–209
Terrados N, Jansson E, Sylven C, Kaijser L (1990) Is hypoxia a stimulus for synthesis of oxidative enzymes and myoglobin? J Appl Physiol 68:2369–2372
Melissa L, Tarnopolsky M, Cipriano N, Green HJ (1997) Skeletal muscle adaptations to training under normobaric hypoxic versus normoxic conditions. Med Sci Sports Exerc 29:238–243
Saltin B, Kim CK, Tearrads N, Larsen H, Svedenhag J, Rolf CJ (1995) Morphology, enzyme activities and buffer capacity in leg muscles of Kenyan and Scandinavian runners. Scand J Med Sci Sports 5:222–230
Desplanches D, Hoppeler H, Tuscher L, Mayet MH, Spielvogel H, Ferretti G, Kayser B, Leuenberger M, Grunenfelder A, Favier R (1996) Muscle tissue adaptations of high altitude natives to training in chronic hypoxia or acute normoxia. J Appl Physiol 81:1946–1951
Robach P, Fulla Y, Westerterp KP, Richalet JP (2004) Comparative response of EPO and soluble transferrin receptor at high altitude. Med Sci Sports Exerc 36:1493–1498
Hoppeler H, Fluck M (2002) Normal mammalian skeletal muscle and its phenotypic plasticity. J Exp Biol 205:2143–2152
Ordway GA, Garry DJ (2004) Myoglobin: an essential hemoprotein in striated muscle. J Exp Biol 207:3441–3446
Garry DJ, Ordway GA, Lorenz JN, Radford NB, Chin ER, Grange RW, Bassel-Duby R, Williams RS (1998) Mice without myoglobin. Nature 395:905–908
Lundby C, Møller P, Kanstrup IL, Olsen NV (2001) Heart rate response to hypoxic exercise: role of dopamine D2-receptors and effect of oxygen supplementation. Clin Sci (Lond) 101:377–383
Lundby C, Sander M, van Hall G, Saltin B, Calbet JAL (2006) Maximal exercise and muscle oxygen extraction in acclimatizing lowlanders and high altitude natives. J Physiol 573:535–547
Calbet JAL, Boushel R, Radegran G, Sondergaard H, Wagner PD, Saltin B (2003) Why is VO2 max after altitude acclimatization still reduced despite normalization of arterial O2 content? Am J Physiol Regul Integr Comp Physiol 284:R304–R316
Robach P, Recalcati S, Girelli D, Gelfi C, achmann-Andersen NJ, Thomsen JJ, Norgaard AM, Alberghini A, Campostrini N, Castagna A, Vigano A, Santambrogio P, Kempf T, Wollert KC, Moutereau S, Lundby C, Cairo G (2009) Alterations of systemic and muscle iron metabolism in human subjects treated with low dose recombinant erythropoietin. Blood 113(26):6707–6715. doi:10.1182/blood_2008_09_178095
Dehne N, Kerkweg U, Otto T, Fandrey J (2007) The HIF-1 response to simulated ischemia in mouse skeletal muscle cells neither enhances glycolysis nor prevents myotube cell death. Am J Physiol Regul Integr Comp Physiol 293:R1693–R1701
Kubis HP, Hanke N, Scheibe RJ, Gros G (2005) Accumulation and nuclear import of HIF1 alpha during high and low oxygen concentration in skeletal muscle cells in primary culture. Biochim Biophys Acta Mol Cell Res 1745:187–195
Young AJ, Evans WJ, Fisher EC, Sharp RL, Costill DL, Maher JT (1984) Skeletal muscle metabolism of sea-level natives following short-term high altitude residence. Eur J Appl Physiol 52:463–466
Bigard AX, Brunet A, Guezennec CY, Monod H (1991) Skeletal muscle changes after endurance training at high altitude. J Appl Physiol 71:2114–2121
McClelland GB, Brooks GA (2002) Changes in MCT 1, MCT 4, and LDH expression are tissue specific in rats after long-term hypobaric hypoxia. J Appl Physiol 92:1573–1584
Green HJ, Sutton JR, Cymerman A, Young PM, Houston CS (1989) Operation Everest II: adaptations in human skeletal muscle. J Appl Physiol 66:2454–2461
Dill RP, Chadan SG, Li C, Parkhouse WS (2001) Aging and glucose transporter plasticity in response to hypobaric hypoxia. Mech Ageing Dev 122:533–545
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lundby, C., Calbet, J.A.L. & Robach, P. The response of human skeletal muscle tissue to hypoxia. Cell. Mol. Life Sci. 66, 3615–3623 (2009). https://doi.org/10.1007/s00018-009-0146-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-009-0146-8