Abstract
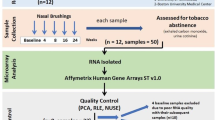
The earliest morphologic evidence of changes in the airways associated with chronic cigarette smoking is in the small airways. To help understand how smoking modifies small airway structure and function, we developed a strategy using fiberoptic bronchoscopy and brushing to sample the human small airway (10th–12th order) bronchial epithelium to assess gene expression (Affymetrix HG-U133A and HG-133 Plus 2.0 array) in phenotypically normal smokers (n = 16, 25 ± 7 pack-years) compared to matched nonsmokers (n = 17). Compared to samples from large (second to third order) bronchi, the small airway samples had a higher proportion of ciliated cells, but less basal, undifferentiated, and secretory cells, and contained Clara cells. Even though the smokers were phenotypically normal, microarray analysis of gene expression of the small airway epithelium of the smokers compared to the nonsmokers demonstrated up- and downregulation of genes in multiple categories relevant to the pathogenesis of chronic obstructive lung disease (COPD), including genes coding for cytokines/innate immunity, apoptosis, mucin, response to oxidants and xenobiotics, and general cellular processes. In the context that COPD starts in the small airways, these gene expression changes in the small airway epithelium in phenotypically normal smokers are candidates for the development of therapeutic strategies to prevent the onset of COPD.






Similar content being viewed by others
References
Pauwels RA, Buist AS, Calverley PM, Jenkins CR, Hurd SS (2001) Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) workshop summary. Am J Respir Crit Care Med 163:1256–1276
Celli BR, MacNee W (2004) Standards for the diagnosis and treatment of patients with COPD: a summary of the ATS/ERS position paper. Eur Respir J 23:932–946
Shapiro SD, Ingenito EP (2005) The pathogenesis of chronic obstructive pulmonary disease: advances in the past 100 years. Am J Respir Cell Mol Biol 32:367–372
Hogg JC, Macklem PT, Thurlbeck WM (1968) Site and nature of airway obstruction in chronic obstructive lung disease. N Engl J Med 278:1355–1360
Yanai M, Sekizawa K, Ohrui T, Sasaki H, Takishima T (1992) Site of airway obstruction in pulmonary disease: direct measurement of intrabronchial pressure. J Appl Physiol 72:1016–1023
Hogg JC (2004) Pathophysiology of airflow limitation in chronic obstructive pulmonary disease. Lancet 364:709–721
Barnes PJ, Shapiro SD, Pauwels RA (2003) Chronic obstructive pulmonary disease: molecular and cellular mechanisms. Eur Respir J 22:672–688
Niewoehner DE, Kleinerman J, Rice DB (1974) Pathologic changes in the peripheral airways of young cigarette smokers. N Engl J Med 291:755–758
Cosio MG, Hale KA, Niewoehner DE (1980) Morphologic and morphometric effects of prolonged cigarette smoking on the small airways. Am Rev Respir Dis 122:265–321
Saetta M, Finkelstein R, Cosio MG (1994) Morphological and cellular basis for airflow limitation in smokers. Eur Respir J 7:1505–1515
Cosio Piqueras MG, Cosio MG (2001) Disease of the airways in chronic obstructive pulmonary disease. Eur Respir J Suppl 34:41s–49s
Willemse BW, ten Hacken NH, Rutgers B, Postma DS, Timens W (2005) Association of current smoking with airway inflammation in chronic obstructive pulmonary disease and asymptomatic smokers. Respir Res 6:38
Cosio M, Ghezzo H, Hogg JC, Corbin R, Loveland M, Dosman J, Macklem PT (1978) The relations between structural changes in small airways and pulmonary-function tests. N Engl J Med 298:1277–1281
Hogg JC, Chu F, Utokaparch S, Woods R, Elliott WM, Buzatu L, Cherniack RM, Rogers RM, Sciurba FC, Coxson HO, Pare PD (2004) The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med 350:2645–2653
Saetta M, Turato G, Maestrelli P, Mapp CE, Fabbri LM (2001) Cellular and structural bases of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 163:1304–1309
Barnes PJ (2004) Mediators of chronic obstructive pulmonary disease. Pharmacol Rev 56:515–548
Spurzem JR, Rennard SI (2005) Pathogenesis of COPD. Semin Respir Crit Care Med 26:142–153
Hackett NR, Heguy A, Harvey BG, O’Connor TP, Luettich K, Flieder DB, Kaplan R, Crystal RG (2003) Variability of antioxidant-related gene expression in the airway epithelium of cigarette smokers. Am J Respir Cell Mol Biol 29:331–343
Heguy A, Harvey BG, O’Connor TP, Hackett NR, Crystal RG (2003) Sampling-dependent up-regulation of gene expression in sequential samples of human airway epithelial cells. Mol Med 9:200–208
Kaplan R, Luettich K, Heguy A, Hackett NR, Harvey BG, Crystal RG (2003) Monoallelic up-regulation of the imprinted H19 gene in airway epithelium of phenotypically normal cigarette smokers. Cancer Res 63:1475–1482
Spira A, Beane J, Shah V, Liu G, Schembri F, Yang X, Palma J, Brody JS (2004) Effects of cigarette smoke on the human airway epithelial cell transcriptome. Proc Natl Acad Sci USA 101:10143–10148
Burrows B, Knudson RJ, Cline MG, Lebowitz MD (1977) Quantitative relationships between cigarette smoking and ventilatory function. Am Rev Respir Dis 115:195–205
Troisi RJ, Speizer FE, Rosner B, Trichopoulos D, Willett WC (1995) Cigarette smoking and incidence of chronic bronchitis and asthma in women. Chest 108:1557–1561
Lindstrom M, Kotaniemi J, Jonsson E, Lundback B (2001) Smoking, respiratory symptoms, and diseases: a comparative study between northern Sweden and northern Finland: report from the FinEsS study. Chest 119:852–861
Bailey CJ, Crystal RG, Leopold PL (2003) Association of adenovirus with the microtubule organizing center. J Virol 77:13275–13287
Irizarry RA, Hobbs B, Collin F, Beazer-Barclay YD, Antonellis KJ, Scherf U, Speed TP (2003) Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4:249–264
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B57:289–300
Barrett T, Suzek TO, Troup DB, Wilhite SE, Ngau WC, Ledoux P, Rudnev D, Lash AE, Fujibuchi W, Edgar R (2005) NCBI GEO: mining millions of expression profiles-database and tools. Nucleic Acids Res 33:D562–D566
Breeze RG, Wheeldon EB (1977) The cells of the pulmonary airways. Am Rev Respir Dis 116:705–777
McDowell EM, Barrett LA, Glavin F, Harris CC, Trump BF (1978) The respiratory epithelium. I. Human bronchus. J Natl Cancer Inst 61:539–549
Mercer RR, Russell ML, Roggli VL, Crapo JD (1994) Cell number and distribution in human and rat airways. Am J Respir Cell Mol Biol 10:613–624
Danel C, Erzurum SC, McElvaney NG, Crystal RG (1996) Quantitative assessment of the epithelial and inflammatory cell populations in large airways of normals and individuals with cystic fibrosis. Am J Respir Crit Care Med 153:362–368
Knight DA, Holgate ST (2003) The airway epithelium: structural and functional properties in health and disease. Respirology 8:432–446
Hermans C, Bernard A (1999) Lung epithelium-specific proteins: characteristics and potential applications as markers. Am J Respir Crit Care Med 159:646–678
Plopper CG, Hyde DM, Buckpitt AR (1997) Clara cells. In: RG Crystal, JB West, ER Weibel, PJ Barnes (eds) The lung: scientific foundations. Lippincott-Raven, Philadelphia, p 535
Massaro GD, Singh G, Mason R, Plopper CG, Malkinson AM, Gail DB (1994) Biology of the Clara cell. Am J Physiol 266:L101–L106
Boers JE, Ambergen AW, Thunnissen FB (1999) Number and proliferation of Clara cells in normal human airway epithelium. Am J Respir Crit Care Med 159:1585–1591
Mueller TD, Zhang JL, Sebald W, Duschl A (2002) Structure, binding, and antagonists in the IL-4/IL-13 receptor system. Biochim Biophys Acta 1592:237–250
D’Ambrosio D, Mariani M, Panina-Bordignon P, Sinigaglia F (2001) Chemokines and their receptors guiding T lymphocyte recruitment in lung inflammation. Am J Respir Crit Care Med 164:1266–1275
Fujimoto K, Imaizumi T, Yoshida H, Takanashi S, Okumura K, Satoh K (2001) Interferon-gamma stimulates fractalkine expression in human bronchial epithelial cells and regulates mononuclear cell adherence. Am J Respir Cell Mol Biol 25:233–238
Kazanskaya O, Glinka A, del Barco Barrantes I, Stannek P, Niehrs C, Wu W (2004) R-Spondin2 is a secreted activator of Wnt/beta-catenin signaling and is required for Xenopus myogenesis. Dev Cell 7:525–534
Gelbman B, Heguy A, O’Connor TP, Zabner J, Crystal RG (2005) Adenovirus-mediated gene transfer demonstrates that pirin, a transcription factor up-regulated in the bronchial epithelium by cigarette smoke, mediates bronchial epithelial cell apoptosis. Mol Ther 2:A803
Dechend R, Hirano F, Lehmann K, Heissmeyer V, Ansieau S, Wulczyn FG, Scheidereit C, Leutz A (1999) The Bcl-3 oncoprotein acts as a bridging factor between NF-kappaB/Rel and nuclear co-regulators. Oncogene 18:3316–3323
Orzaez D, de Jong AJ, Woltering EJ (2001) A tomato homologue of the human protein PIRIN is induced during programmed cell death. Plant Mol Biol 46:459–468
Wendler WM, Kremmer E, Forster R, Winnacker EL (1997) Identification of pirin, a novel highly conserved nuclear protein. J Biol Chem 272:8482–8489
Hofmann TG, Stollberg N, Schmitz ML, Will H (2003) HIPK2 regulates transforming growth factor-beta-induced c-Jun NH(2)-terminal kinase activation and apoptosis in human hepatoma cells. Cancer Res 63:8271–8277 (Dec 1)
Shi M, Zhang X, Wang P, Zhang HW, Zhang BH, Wu MC (2005) TIP30 regulates apoptosis-related genes in its apoptotic signal transduction pathway. World J Gastroenterol 11:221–227
Takekawa M, Saito H (1998) A family of stress-inducible GADD45-like proteins mediate activation of the stress-responsive MTK1/MEKK4 MAPKKK. Cell 95:521–530
Bowler RP, Crapo JD (2002) Oxidative stress in airways: is there a role for extracellular superoxide dismutase? Am J Respir Crit Care Med 166:S38–S43
Fischer BM, Voynow JA (2002) Neutrophil elastase induces MUC5AC gene expression in airway epithelium via a pathway involving reactive oxygen species. Am J Respir Cell Mol Biol 26:447–452
Mio T, Romberger DJ, Thompson AB, Robbins RA, Heires A, Rennard SI (1997) Cigarette smoke induces interleukin-8 release from human bronchial epithelial cells. Am J Respir Crit Care Med 155:1770–1776
Profita M, Chiappara G, Mirabella F, Di GR, Chimenti L, Costanzo G, Riccobono L, Bellia V, Bousquet J, Vignola AM (2003) Effect of cilomilast (Ariflo) on TNF-alpha, IL-8, and GM-CSF release by airway cells of patients with COPD. Thorax 58:573–579
Shao MX, Nakanaga T, Nadel JA (2004) Cigarette smoke induces MUC5AC mucin overproduction via tumor necrosis factor-alpha-converting enzyme in human airway epithelial (NCI-H292) cells. Am J Physiol Lung Cell Mol Physiol 287:L420–L427
Willems LN, Kramps JA, Stijnen T, Sterk PJ, Weening JJ, Dijkman JH (1989) Antileukoprotease-containing bronchiolar cells. Relationship with morphologic disease of small airways and parenchyma. Am Rev Respir Dis 139:1244–1250
Ning W, Li CJ, Kaminski N, Feghali-Bostwick CA, Alber SM, Di YP, Otterbein SL, Song R, Hayashi S, Zhou Z, Pinsky DJ, Watkins SC, Pilewski JM, Sciurba FC, Peters DG, Hogg JC, Choi AM (2004) Comprehensive gene expression profiles reveal pathways related to the pathogenesis of chronic obstructive pulmonary disease. Proc Natl Acad Sci USA 101:14895–14900
Vayssier M, Banzet N, Francois D, Bellmann K, Polla BS (1998) Tobacco smoke induces both apoptosis and necrosis in mammalian cells: differential effects of HSP70. Am J Physiol 275:L771–L779
Li X, Shu R, Filippatos G, Uhal BD (2004) Apoptosis in lung injury and remodeling. J Appl Physiol 97:1535–1542
Bartalesi B, Cavarra E, Fineschi M, Lucattelli B, Martorana PA, Lungarelia G (2005) Different lung responses to cigarette smoke in two strains of mice sensitive to oxidants. Eur Respir J 25:15–22
Calabrese F, Giacometti C, Beghe B, Rea F, Loy M, Zuin R, Marulli G, Baraldo S, Saetta M, Valente M (2005) Marked alveolar apoptosis/proliferation imbalance in end-stage emphysema. Respir Res 6:14
Hodge S, Hodge G, Holmes M, Reynolds PN (2005) Increased airway epithelial and T-cell apoptosis in COPD remains despite smoking cessation. Eur Respir J 25:447–454
Zheng T, Kang MJ, Crothers K, Zhu Z, Liu W, Lee CG, Rabach LA, Chapman HA, Homer RJ, Aldous D, Desanctis G, Underwood S, Graupe M, Flavell RA, Schmidt JA, Elias JA (2005) Role of cathepsin S-dependent epithelial cell apoptosis in IFN-gamma-induced alveolar remodeling and pulmonary emphysema. J Immunol 174:8106–8115
Repine JE, Bast A, Lankhorst I (1997) Oxidative stress in chronic obstructive pulmonary disease. Oxidative Stress Study Group. Am J Respir Crit Care Med 156:341–357
Perkins KA (1992) Metabolic effects of cigarette smoking. J Appl Physiol 72:401–409
Goris AH, Vermeeren MA, Wouters EF, Schols AM, Westerterp KR (2003) Energy balance in depleted ambulatory patients with chronic obstructive pulmonary disease: the effect of physical activity and oral nutritional supplementation. Br J Nutr 89:725–731
Acknowledgements
We thank the Pulmonary Fellows and the nurses of the bronchoscopy suite of the Division of Pulmonary and Critical Care Medicine for helping with bronchoscopies; Igor Dolgalev and Tina Raman for excellent technical assistance; T. O’Connor and N. Hackett for helpful discussions; and N. Mohamed for help in preparing this manuscript. These studies were supported, in part, by NIH R01 HL074326 and Weill Cornell GCRC M01RR00047 and the Will Rogers Memorial Fund, Los Angeles, CA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ben-Gary Harvey and Adriana Heguy contributed equally to this study.
An erratum to this article can be found at http://dx.doi.org/10.1007/s00109-008-0351-1
Electronic supplementary material
Below is the link to the electronic supplementary material
Supplemental Fig. 1
Fluoroscopy of brushing to obtain large and small airway epithelial cells through a fiberoptic bronchoscope. a Proximal brush, large airway (third order), and right lower lobe. b. Distal brush, small airway (10th to 12th order), and right lower lobe. Arrows mark the tip of the brush(PDF 476 kb)
Supplemental Fig. 2
Hierarchical cluster analysis of airway epithelial gene expression changes in the small airways of healthy smokers vs nonsmokers. a Dendrogram resulting from cluster analysis for all expressed genes (30,963 probe sets, present in at least one array), using MAS5-generated values and the GeneSpring software (Spearman correlation) by sample. Note that this analysis does not segregate smokers from nonsmokers, indicating that cigarette smoking does not change the global pattern of gene expression in the epithelium of the small airways. b. Cluster analysis for genes differentially expressed in smokers vs nonsmokers. The cluster was generated for 118 significant probe sets [p < 0.05, Welch t test with the Benjamini–Hochberg false discovery rate multiple test correction, by both MAS5 and RMA preprocessing algorithms; >1.5-fold change in smokers vs nonsmokers (upregulated) or nonsmokers vs smokers (downregulated), calculated by either the MAS5 values or the RMA values], using the GeneSpring program (Spearman correlation) by sample, and by gene. Smokers and nonsmokers were not grouped a priori. Genes expressed above average are represented in red, genes expressed below average in blue, and average levels in white. The degree of red or blue intensity indicates the degree of up- or downregulation relative to the average level of expression across all samples. The genes are represented vertically, and the individual samples [S for smokers (n = 10) and NS for nonsmokers (n = 12)] horizontally(PDF 218 kb)
Supplemental Table 1
Demographics of the Study Population1 (PDF 91 kb).
Supplemental Table 2
Genes Differentially Expressed in the Small Airway Epithelium of 6 Healthy Smokers vs 5 Healthy Non-smokers (PDF 82 kb).
Supplemental Table 3
Examples of Genes Differentially Expressed in the Small Airway Epithelium in Smokers vs Non-smokers from Group A in Functional Categories that are Relevant for the Pathogenesis of COPD (PDF 40 kb).
Supplemental Table 4
Genes Differentially Expressed in the Small Airway Epithelium of 10 Healthy Smokers vs 12 Healthy Non-smokers (PDF 155 kb).
Supplemental Table 5
Examples of Genes Differentially Expressed in the Small Airway Epithelium of 10 Smokers vs 12 Non-smokers, in Functional Categories that are Relevant for the Pathogenesis of COPD1 (PDF 34 kb).
Supplemental Table 6
Comparison of HG-U133 2.0 vs. HG-U133A results (PDF 104 kb).
Rights and permissions
About this article
Cite this article
Harvey, BG., Heguy, A., Leopold, P.L. et al. Modification of gene expression of the small airway epithelium in response to cigarette smoking. J Mol Med 85, 39–53 (2007). https://doi.org/10.1007/s00109-006-0103-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-006-0103-z