Abstract
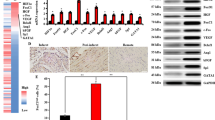
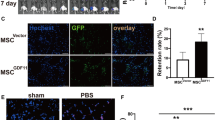
We previously reported that mesenchymal stem cells (MSC) co-expressing Akt and angiopoietin-1 (Ang-1) preserved infarcted heart function via angiomyogenesis. The present study determined the mechanism of co-overexpression of Akt and Ang-1 in promoting endothelial commitment of MSC. The cells were transduced with vectors encoding for Akt (AktMSC), Ang-1 (Ang-1MSC), and both Akt and Ang-1 (AAMSC) using Empty vector transduced MSC (EmpMSC) as control. Molecular studies indicated a coordinated interaction between Akt and Ang-1 in AAMSC and led to non-hypoxic stabilization of hypoxia inducible factor-1α (HIF-Iα) which accentuated under 4-h anoxia. We also observed HIF-Iα dependent induction of hemeoxygenase-1, endothelial specific markers and VEGF in AAMSC. Vascular commitment of AAMSC was confirmed by immunostaining, Western blotting and flow cytometry for endothelial specific early and late markers including Flt1, Flk1, Tie2, VCAM-1, and von Willebrand Factor-VIII (vWF-VIII) in HIF-Iα dependent fashion besides exhibiting higher emigrational activity and angiogenesis in vitro. AAMSC transplanted into rat model of myocardial infarction showed higher Flk1 and Flt1 positivity and also promoted intrinsic Flk1+ and Flt1+ cell mobilization into the infarcted heart. Given the ease of availability of MSC and simplicity of approach to co-overexpress Ang-1 and Akt to enhance their endothelial commitment, the strategy will be significant for cellular angiogenesis to treat ischemic heart.






Similar content being viewed by others
Abbreviations
- Ang-1:
-
Angiopoietin-1
- BM:
-
Bone marrow
- DMEM:
-
Dulbecco’s Modified Eagle Medium
- HIF-Iα:
-
Hypoxia inducible factor-1α
- HO-1:
-
Hemeoxygenase-1
- AAMSC:
-
Mesenchymal stem cells overexpressing Akt and angiopoietin-1
- EmpMSC:
-
Empty vector trnasduced MSC
- MSC:
-
Mesenchymal stem cells
- PGF:
-
Placental growth factor
- VEGF:
-
Vascular endothelial growth factor
- Sc:
-
Scramble
References
Noiseux N, Gnecchi M, Lopez-Ilasaca M, Zhang L, Solomon SD, Deb A, Dzau VJ, Pratt RE (2006) Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Mol Ther 14:840–850
Haider H, Jiang S, Idris NM, Ashraf M (2008) IGF-1-overexpressing mesenchymal stem cells accelerate bone marrow stem cell mobilization via paracrine activation of SDF-1alpha/CXCR4 signaling to promote myocardial repair. Circ Res 103:1300–1308
Yau TM, Kim C, Li G, Zhang Y, Weisel RD, Li RK (2005) Maximizing ventricular function with multimodal cell-based gene therapy. Circulation 112:I123–I128
Uemura R, Xu M, Ahmad N, Ashraf M (2006) Bone marrow stem cells prevent left ventricular remodeling of ischemic heart through paracrine signaling. Circ Res 98:1414–1421
Jiang S, Haider H, Idris NM, Salim A, Ashraf M (2006) Supportive interaction between cell survival signaling and angiocompetent factors enhances donor cell survival and promotes angiomyogenesis for cardiac repair. Circ Res 99:776–784
Duffy GP, D'Arcy S, Ahsan T, Nerem RM, O'Brien T, Barry F (2010) Mesenchymal stem cells overexpressing ephrin-b2 rapidly adopt an early endothelial phenotype with simultaneous reduction of osteogenic potential. Tissue Eng Part A 16:2755–2768
Mangi AA, Noiseux N, Kong D, He H, Rezvani M, Ingwall JS, Dzau VJ (2003) Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med 9:1195–1201
Kim SH, Moon HH, Kim HA, Hwang KC, Lee M, Choi D (2010) Hypoxia-inducible vascular endothelial growth factor-engineered mesenchymal stem cells prevent myocardial ischemic injury. Mol Ther 19:741–750
Ye L, Haider H, Jiang S, Tan RS, Toh WC, Ge R, Sim EK (2007) Angiopoietin-1 for myocardial angiogenesis: a comparison between delivery strategies. Eur J Heart Fail 9:458–465
Shujia J, Haider HK, Idris NM, Lu G, Ashraf M (2008) Stable therapeutic effects of mesenchymal stem cell-based multiple gene delivery for cardiac repair. Cardiovasc Res 77:525–533
Iyer NV, Kotch LE, Agani F, Leung SW, Laughner E, Wenger RH, Gassmann M, Gearhart JD, Lawler AM, Yu AY et al (1998) Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1 alpha. Genes Dev 12:149–162
Lee BL, Kim WH, Jung J, Cho SJ, Park JW, Kim J, Chung HY, Chang MS, Nam SY (2008) A hypoxia-independent up-regulation of hypoxia-inducible factor-1 by Akt contributes to angiogenesis in human gastric cancer. Carcinogenesis 29:44–51
Kim HW, Haider HK, Jiang S, Ashraf M (2009) Ischemic preconditioning augments survival of stem cells via miR-210 expression by targeting caspase-8-associated protein 2. J Biol Chem 284:33161–33168
Ahmed RP, Haider KH, Shujia J, Afzal MR, Ashraf M (2010) Sonic Hedgehog gene delivery to the rodent heart promotes angiogenesis via iNOS/netrin-1/PKC pathway. PLoS One 5:e8576
Krause DS (2002) Plasticity of marrow-derived stem cells. Gene Ther 9:754–958
Daly C, Wong V, Burova E, Wei Y, Zabski S, Griffiths J, Lai KM, Lin HC, Ioffe E, Yancopoulos GD et al (2004) Angiopoietin-1 modulates endothelial cell function and gene expression via the transcription factor FKHR (FOXO1). Genes Dev 18:1060–1071
DeBusk LM, Hallahan DE, Lin PC (2004) Akt is a major angiogenic mediator downstream of the Ang1/Tie2 signaling pathway. Exp Cell Res 298:167–177
Rey S, Semenza GL (2010) Hypoxia-inducible factor-1-dependent mechanisms of vascularization and vascular remodelling. Cardiovasc Res 86:236–242
Misra S, Fu AA, Misra KD, Shergill UM, Leof EB, Mukhopadhyay D (2010) Hypoxia-induced phenotypic switch of fibroblasts to myofibroblasts through a matrix metalloproteinase 2/tissue inhibitor of metalloproteinase-mediated pathway: implications for venous neointimal hyperplasia in hemodialysis access. J Vasc Interv Radiol 21:896–902
Lange C, Heynen SR, Tanimoto N, Thiersch M, Le YZ, Meneau I, Seeliger MW, Samardzija M, Caprara C, Grimm C (2011) Normoxic activation of hypoxia inducible factors in photoreceptors provides transient protection against light induced retinal degeneration. Invest Ophthalmol Vis Sci 52:5872–5880
Palladino MA, Pirlamarla PR, McNamara J, Sottas CM, Korah N, Hardy MP, Hales DB, Hermo L (2011) Normoxic expression of hypoxia-inducible factor 1 in rat Leydig cells in vivo and in vitro. J Androl 32:307–323
Xu J, Peng Z, Li R, Dou T, Xu W, Gu G, Liu Y, Kang Z, Tao H, Zhang JH (2009) Normoxic induction of cerebral HIF-Ialpha by acetazolamide in rats: role of acidosis. Neurosci Lett 451:274–278
Carver DJ, Gaston B, Deronde K, Palmer LA (2007) Akt-mediated activation of HIF-I in pulmonary vascular endothelial cells by S-nitrosoglutathione. Am J Respir Cell Mol Biol 37:255–263
Chen JX, Stinnett A (2008) Ang-1 gene therapy inhibits hypoxia-inducible factor-1alpha (HIF-Ialpha)-prolyl-4-hydroxylase-2, stabilizes HIF-Ialpha expression, and normalizes immature vasculature in db/db mice. Diabetes 57:3335–3343
Alam H, Weck J, Maizels E, Park Y, Lee EJ, Ashcroft M, Hunzicker-Dunn M (2009) Role of the phosphatidylinositol-3-kinase and extracellular regulated kinase pathways in the induction of hypoxia-inducible factor (HIF-I)-1 activity and the HIF-I target vascular endothelial growth factor in ovarian granulosa cells in response to follicle-stimulating hormone. Endocrinology 150:915–928
Huang H, Tindall DJ (2007) Dynamic FoxO transcription factors. J Cell Sci 120:2479–2487
Asahara T, Bauters C, Zheng LP, Takeshita S, Bunting S, Ferrara N, Symes JF, Isner JM (1995) Synergistic effect of vascular endothelial growth factor and basic fibroblast growth factor on angiogenesis in vivo. Circulation 92:365–371
Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M, Wu Y, Bono F, Devy L, Beck H (2001) Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 7:575–583
Ryu JK, Cho CH, Shin HY, Song SU, Oh SM, Lee M, Piao S, Han JY, Kim IH, Koh GY et al (2006) Combined angiopoietin-1 and vascular endothelial growth factor gene transfer restores cavernous angiogenesis and erectile function in a rat model of hypercholesterolemia. Mol Ther 13:705–715
Okuyama H, Krishnamachary B, Zhou YF, Nagasawa H, Bosch-Marce M, Semenza GL (2006) Expression of vascular endothelial growth factor receptor 1 in bone marrow-derived mesenchymal cells is dependent on hypoxia-inducible factor 1. J Biol Chem 281:15554–15563
Reyes M, Dudek A, Jahagirdar B, Koodie L, Marker PH, Verfaillie CM (2002) Origin of endothelial progenitors in human postnatal bone marrow. J Clin Invest 109:337–346
Nagaya N, Kangawa K, Itoh T, Iwase T, Murakami S, Miyahara Y, Fujii T, Uematsu M, Ohgushi H, Yamagishi M (2005) Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation 112:1128–1135
Angiopoietins RY (2010) Recent Results Cancer Res 180:3–13
Askari A, Unzek S, Goldman CK, Ellis SG, Thomas JD, DiCorleto PE, Topol EJ, Penn MS (2004) Cellular, but not direct, adenoviral delivery of vascular endothelial growth factor results in improved left ventricular function and neovascularization in dilated ischemic cardiomyopathy. J Am Coll Cardiol 43:1908–1914
Acknowledgments
This work was supported by NIH Grants# [R37 HL074272;HL-080686;HL-087246 (M.A) and HL-087288;HL-089535; HL106190-01 (Kh.H.H)].
Conflict of Interest Disclosure Statement
Nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 190 kb)
Rights and permissions
About this article
Cite this article
Lai, V.K., Afzal, M.R., Ashraf, M. et al. Non-hypoxic stabilization of HIF-Iα during coordinated interaction between Akt and angiopoietin-1 enhances endothelial commitment of bone marrow stem cells. J Mol Med 90, 719–730 (2012). https://doi.org/10.1007/s00109-011-0852-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0852-1