Abstract.
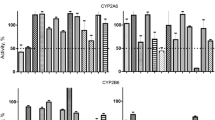
The first step in the oxidative metabolism of cocaine is N-demethylation to norcocaine, which is further N-hydroxylated to more toxic N-hydroxynorcocaine. In this study we examined the kinetics of norcocaine N-hydroxylation mediated by cytochrome P450 (CYP) in mouse and human liver microsomes. N-hydroxynorcocaine was identified by analytical HPLC-MS after incubation of norcocaine with mouse liver microsomes in the presence of NADPH. In mouse liver microsomes, there was no apparent difference in Km values for norcocaine N-hydroxylation between male and female microsomes, while the Vmax rate was approximately two times higher in female than in male microsomes (34±10 v 16±4 pmol/min per mg protein). The Km value for norcocaine N-hydroxylation in human liver microsomes was approximately three times higher than that observed in comparable incubations using mouse liver microsomes, whereas the Vmax rate was ten times lower. Both cocaine and norcocaine induced type I difference spectra upon interaction with CYP in mouse liver microsomes. In contrast, in human microsomes both type I and type II spectra were recorded. In the 0.01 to 1 mM concentration range, cocaine and norcocaine inhibited mouse microsomal testosterone 6α-, 7α- and 16α-hydroxylation reactions by 20% to 30%. Testosterone 6β- and 15α-hydroxylations were blocked by 60% and 50%, respectively, by 1 mM norcocaine, while only 40% inhibition was obtained with 1 mM cocaine. Coumarin 7-hydroxylation and pentoxyresorufin O-deethylation were inhibited by 50% by 1 and 0.4 mM norcocaine, respectively. In contrast, 10 and 2 mM cocaine, respectively, were needed to obtain the same degrees of inhibition. In human liver microsomes, 1 mM norcocaine and cocaine blocked testosterone 6β-hydroxylase by 60% and 40%, respectively. Coumarin 7-hydroxylation was inhibited by only 30% by norcocaine (5.4 mM) and cocaine (10 mM). Norcocaine N-hydroxylation in mouse and human liver microsomes was blocked by 30% and 60%, respectively, by α-naphthoflavone (0.1 mM). The reaction was inhibited by 30–40% by metyrapone, cimetidine and gestodene at a concentration of 1 mM in mouse microsomes, while in human microsomes, 70% inhibition was obtained with 1 mM metyrapone and cimetidine. Taken together, these results indicate that (1) norcocaine N-hydroxylation is at least partly a CYP-mediated reaction, (2) the rate of reaction is considerably lower in human liver microsomes than in mouse liver microsomes and (3) several CYP subfamilies including 1A, 2A, 3A and possibly 2B may contribute to the formation of N-hydroxynorcocaine.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Pellinen, P., Kulmala, L., Konttila, J. et al. Kinetic characteristics of norcocaine N-hydroxylation in mouse and human liver microsomes: involvement of CYP enzymes. Arch Toxicol 74, 511–520 (2000). https://doi.org/10.1007/s002040000154
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002040000154