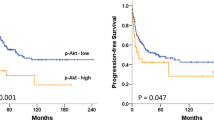
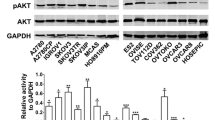
Abstract
Abnormalities in the phosphatidylinositol 3-kinase (PI3K)/Akt signaling pathway are commonly observed in human cancers and contribute to chemotherapy resistance. Combination therapy, involving the use of molecular targeted agents and traditional cytotoxic drugs, may represent a promising strategy to lower resistance and enhance cytotoxicity. Here, we demonstrate the efficacy of an Akt inhibitor, MK-2206, in increasing the cytotoxic effect of either paclitaxel (Taxol) or cisplatin against the ovarian cancer cell lines SKOV3 (with constitutively active Akt) and ES2 (with inactive Akt). Sequential treatment of Taxol or cisplatin, followed by MK-2206, induced a synergistic inhibition of cell proliferation and effectively promoted cell death, either by inhibiting the phosphorylation of Akt and its downstream effectors 4E-BP1 and p70S6K in SKOV3 cells or by restoring p53 levels, which were downregulated after Taxol or cisplatin treatment, in ES2 cells. Combination treatment also downregulated the pro-survival protein Bcl-2 in both SKOV3 and ES2 cells, which may have contributed to cell death. In addition, we discovered that Taxol/MK-2206 or cisplatin/MK-2206 combination treatment resulted in significant enhancement of intracellular reactive oxygen species (ROS) induced by MK-2206, in both SKOV3 and ES2 cells; however, MK-2206-induced growth inhibition was reversed by a ROS scavenger only in ES2 cells. MK-2206 also suppressed DNA repair, particularly in SKOV3 cells. Taken together, our results demonstrate that the Akt inhibitor MK-2206 enhances the efficacy of cytotoxic agents in both Akt-active and Akt-inactive ovarian cancer cells but through different mechanisms.







Similar content being viewed by others
References
Agarwal R, Kaye SB (2003) Ovarian cancer: strategies for overcoming resistance to chemotherapy. Nat Rev Cancer 3:502–516. doi:10.1038/nrc1123
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 15:6541–6551
Berkenblit A, Cannistra SA (2005) Advances in the management of epithelial ovarian cancer. J Reprod Med 50:426–438
Caro AA, Cederbaum AI (2006) Role of phosphatidylinositol 3-kinase/AKT as a survival pathway against CYP2E1-dependent toxicity. J Pharmacol Exp Ther 318:360–372. doi:10.1124/jpet.106.102921
Chen H, Ma Z, Vanderwaal RP, Feng Z, Gonzalez-Suarez I, Wang S, Zhang J, Roti Roti JL, Gonzalo S, Zhang J (2011) The mTOR inhibitor rapamycin suppresses DNA double-strand break repair. Radiat Res 175:214–224
Cheng Y, Ren X, Zhang Y, Patel R, Sharma A, Wu H, Robertson GP, Yan L, Rubin E, Yang JM (2011) eEF-2 kinase dictates crosstalk between autophagy and apoptosis induced by Akt inhibition, thereby modulating cytotoxicity of novel Akt inhibitor MK-2206. Cancer Res 71:2654–2663. doi:10.1158/0008-5472.CAN-10-2889
Chou TC, Talalay P (1984) Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzym Regul 22:27–55
Concin N, Stimpfl M, Zeillinger C, Wolff U, Hefler LJ, Sedlak J, Leodolter S, Zeillinger R (2003) Role of p53 in G2/M cell cycle arrest and apoptosis in response to gamma-irradiation in ovarian carcinoma cell lines. Int J Oncol 22:51–57
Courtney KD, Corcoran RB, Engelman JA (2010) The PI3K pathway as drug target in human cancer. J Clin Oncol 28:1075–1083. doi:10.1200/JCO.2009.25.3641
DiSaia PJ, Bloss JD (2003) Treatment of ovarian cancer: new strategies. Gynecol Oncol 90:S24–32
El-Gazzar A, Perco P, Eckelhart E, Anees M, Sexl V, Mayer B, Liu Y, Mikulits W, Horvat R, Pangeri T, Zheng D, Krainer M (2010) Natural immunity enhances the activity of a DR5 agonistic antibody and carboplatin in the treatment of ovarian cancer. Mol Cancer Ther 9:1007–1018. doi:10.1158/1535-7163.MCT-09-0933
Hanrahan AJ, Schultz N, Westfal ML, Sakr RA, Giri DD, Scarperi S, Janakiraman M, Olvera N, Stevens EV, She QB, Aghajanian C, King TA, Stanchina E, Spriggs DR, Heguy A, Taylor BS, Sander C, Rosen N, Levine DA, Solit DB (2012) Genomic complexity and AKT dependence in serous ovarian cancer. Cancer Discov 2:56–67
Hirai H, Sootome H, Nakatsuru Y, Miyama K, Taguchi S, Tsujioka K, Ueno Y, Hatch H, Majumder PK, Pan BS, Kotani H (2010) MK-2206, an allosteric Akt inhibitor, enhances antitumor efficacy by standard chemotherapeutic agents or molecular targeted drugs in vitro and in vivo. Mol Cancer Ther 9:1956–1967. doi:10.1158/1535-7163.MCT-09-1012
Hsu LC, Kapali M, DeLoia JA, Gallion HH (2005) Centrosome abnormalities in ovarian cancer. Int J Cancer 113:746–751
Li Z, Yan S, Attayan N, Ramalingam S, Thiele CJ (2012) Combination of an allosteric Akt inhibitor MK-2206 with etoposide or rapamycin enhances the antitumor growth effect in neuroblastoma. Clin Cancer Res 18:3603–3615. doi:10.1158/1078-0432.CCR-11-3321
Lindsley CW, Barnett SF, Layton ME, Bilodeau MT (2008) The PI3K/Akt pathway: recent progress in the development of ATP-competitive and allosteric Akt kinase inhibitors. Curr Cancer Drug Targets 8:7–18
Liu P, Cheng T, Roberts TM, Zhao JJ (2009) Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov 8:627–644. doi:10.1038/nrd2926
Mendoza MC, Er EE, Blenis J (2011) The Ras-ERK and PI3K-mTOR pathways: cross-talk and compensation. Trends Biochem Sci 36:320–328
Paull TT, Rogakou EP, Yamazaki V, Kirchgessner CU, Gellert M, Bonner WM (2000) A critical role for histone H2AX in recruitment of repair factors to nuclear foci after DNA damage. Curr Biol 10:886–895
Peng DJ, Wang J, Zhou JY, Wu GS (2010) Role of the Akt/mTOR survival pathway in cisplatin resistance in ovarian cancer cells. Biochem Biophys Res Commun 394:600–605. doi:10.1016/j.bbrc.2010.03.029
Scully R, Chen J, Ochs RL, Keegan K, Hoekstra M, Feunteun J, Livingston DM (1997) Dynamic changes of BRCA1 subnuclear location and phosphorylation state are initiated by DNA damage. Cell 90:425–435
Sun M, Wang G, Paciga JE, Feldman RI, Yuan ZQ, Ma XL, Shelley SA, Jove R, Tsichlis PN, Nicosia SV, Cheng JQ (2001) AKT1/PKBalpha kinase is frequently elevated in human cancers and its constitutive activation is required for oncogenic transformation in NIH3T3 cells. Am J Pathol 159:431–437
West KA, Castillo SS, Dennis PA (2002) Activation of the PI3K/Akt pathway and chemotherapeutic resistance. Drug Resist Updat 5:234–248
Yu YM, Pace SM, Allen SR, Deng CX, Hsu LC (2008) A PP1-binding motif present in BRCA1 plays a role in its DNA repair function. Int J Biol Sci 4:352–361. doi:10.7150/ijbs.4.352
Yuan ZQ, Sun M, Feldman RI (2000) Frequent activation of AKT2 and induction of apoptosis by inhibition of phosphoinositide-3-OH kinase/Akt pathway in human ovarian cancer. Oncogene 19:2324–2330. doi:10.1038/sj.onc.1203598
Zhao L, Wientjes MG, Au JL (2004) Evaluation of combination chemotherapy: integration of nonlinear regression, curve shift, isobologram, and combination index analyses. Clin Cancer Res 10:7994–8004. doi:10.1158/1078-0432.CCR-04-1087
Acknowledgments
This study was supported by funds from the National Taiwan University (L.C.H.). We thank Jui-Ling Hsu, She-Hung Chan, Cheng-Hsiang Huang, and Ching-Chun Huang for technical support and helpful discussion.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 223 kb)
Rights and permissions
About this article
Cite this article
Lin, YH., Chen, B.YH., Lai, WT. et al. The Akt inhibitor MK-2206 enhances the cytotoxicity of paclitaxel (Taxol) and cisplatin in ovarian cancer cells. Naunyn-Schmiedeberg's Arch Pharmacol 388, 19–31 (2015). https://doi.org/10.1007/s00210-014-1032-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-014-1032-y