Abstract
Rationale
Previous studies have implicated platelet amyloid precursor protein (APP) as a candidate biomarker for Alzheimer’s disease (AD). Platelets contain more than 95% of the circulating APP and enclose the enzymatic machinery for the APP metabolism yielding both soluble APP and amyloid-β peptides.
Objectives
The objective of this study is to compare the ratio of 130- to 110-kDa fragments of APP in platelets from patients with AD, mild cognitive impairment (MCI), and elderly controls.
Materials and methods
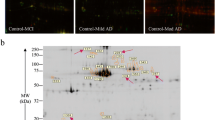
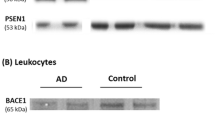
After subjects were grouped according to diagnosis, APP ratio in platelets was evaluated by means of Western blot analysis.
Results
The APP ratio was significantly lower in AD patients (1.01 ± 0.21) as compared to controls (1.24 ± 0.21, p = 0.001) and MCI patients (1.18 ± 0.21, p = 0.027), but no significant differences were found between MCI and controls (p = 0.904). In addition, we found positive correlations between the APP ratio and 1,6-diphenyl-1,3,5-hexatriene anisotropy (r = 0.3, p = 0.01), as well as with certain parameters of cognitive decline, namely, the mini-mental state examination score (r = 0.33, p = 0.003), the total Cambridge cognitive test (CAMCOG) score (r = 0.37, p = 0.001), and the score on the memory subscale of the CAMCOG (r = 0.38, p = 0.001).
Conclusions
The pattern of platelet APP fragments was altered in patients with AD but not in patients with MCI. The alteration of APP fragments was correlated with membrane fluidity and the cognitive decline.



Similar content being viewed by others
References
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Association, Washington
Army Individual Test Battery (1944) Manual of directions and scoring war department, Adjunt General’s Office Trail, Washington
Baskin F, Rosenberg RN, Iyer L, Hynan L, Cullum CM (2000) Platelet APP isoform ratios correlated with declining cognition in AD. Neurology 54:1907–1909
Blessed G, Tomlinson BE, Roth M (1968) The association between quantitative measures of dementia and of senile change in the cerebral grey matter of elderly subjects. Br J Psychiatry 114:797–811
Daly IVJ, Lahiri DK, Justus DE, Kotwal GJ (1998) Detection of the membrane-retained carboxy-terminal tail containing polypeptides of the amyloid precursor protein in tissue from Alzheimer’s disease brain. Life Sci 63:2121–2131
Davies TA, Long HJ, Sgro K, Rathbun WH, Mcmenamin ME, Seetoo K, Tibbles H, Billingslea AM, Fine RE, Fishman JB, Levesque CA, Smith SJ, Wells JM, Simons ER (1997) Activated Alzheimer disease platelets retain more beta amyloid precursor protein. Neurobiol Aging 18:147–153
Di Luca M, Pastorino L, Cattabeni F, Zanardi R, Scarone S, Racagni G, Smeraldi E, Perez J (1996) Abnormal pattern of platelet APP isoforms in Alzheimer disease and Down syndrome. Arch Neurol 53:1162–1166
Di Luca M, Pastorino L, Bianchetti A, Perez J, Viagnolo LA, Lenzi GL, Trabucchi M, Cattebeni F, Padovani A (1998) Differential level of platelet amyloid β precursor protein isoforms—an early marker for Alzheimer disease. Arch Neurol 55:1195–1200
Di Luca M, Colciaghi F, Pastorino L, Borroni B, Padovani A, Cattabeni F (2000) Platelets as a peripheral district were to study pathogenetic mechanisms of Alzheimer disease: the case of amyloid precursor protein. Eur J Pharmacol 405:277–283
Eckert GP, Cairns NJ, Maras A, Gattaz WF, Muller WE (2000) Cholesterol modulates the membrane-disordering effects of beta-amyloid peptides in the hippocampus: specific changes in Alzheimer’s disease. Dement Geriatr Cogn Disord 11(4):181–186
Erzigkeit H (1991) The development of the SKT project. In: Hindmarch I, Hippius H, Wilcock GK (eds) Dementia: molecules, methods and measures. Wiley, Chichester, England, pp 101–108
Fernandes MAS, Proença MT, Nogueira AJA, Oliveira LMV, Santiago B, Santana I, Oliveira CR (1999) Effects of apolipoprotein E genotype on blood lipid composition and membrane platelet fluidity in Alzheimer’s disease. Biochim Biophys Acta 1454:89–96
Flaks MK, Yassuda MS, Regina ACB, Cid AG, Camargo CHP, Gattaz WF, Forlenza OV (2006) The short test (SKT)—a transcultural test for early detection and discrimination of dementia: a preliminary study in Brazil. Int Psychogeriatr 18:121–133
Folstein MF, Folstein SE, Mchugh PR (1975) Mini-mental state: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Fuld P (1980) Guaranteed stimulus processing in the evaluation of memory and learning. Cortex 16:255–271
Haass C (2004) Take five-Bace and the γ-secretase quartet conduct Alzheimer’s amyloid β-peptide generation. EMBO J 23:483–488
Hachinski VC, Iliff ID, Zihkla E, Du Boulay GH, McAllister VL, Marshall J, Russel RW, Symon L (1975) Cerebral blood flow in dementia. Arch Neurol 32:632–637
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62
Hampel H, Teipel SJ, Fuchsberger T, Andreasen N, Wiltfang J, Otto M, Shen Y, Dodel R, Du Y, Farlow M, Möller H-J, Blennow K, Buerger K (2004) Value of CSF β-amyloid1–42 and tau as predictors of Alzheimer’s disease in patients with mild cognitive impairment. Mol Psychiatry 9:705–710
Hansson O, Zetterberg H, Buchhave P, Londos L, Blennow K, Minthon L (2006) Association between CSF biomarkers and incipient Alzheimer’s disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol 5:228–234
Jorm AF, Jacomb PA (1989) The informant questionnaire on cognitive decline in the elderly: sociodemographics correlates, reliability, validity and some norms. Psychol Med 19:1015–1022
Kukull WA, Hinds TR, Schellenberg GD, Belle Gv, Larson EB (1992) Increased platelet membrane fluidity as a diagnostic marker for Alzheimer’s disease. Neurology 42:607–614
Li QX, Whyte S, Tanner JE, Evin G, Beyreuther K, Masters CL (1998) Secretion of Alzheimer’s disease Aβ amyloid peptide by activated human platelets. Lab Invest 78:461–469
Maruyama M, Arai H, Sugita M, Tanji H, Higuchi M, Okamura N, Matsui T, Higuchi S, Matsushita S, Yoshida H, Sasaki H (2001) Cerebrospinal fluid amyloid β1–42 levels in the mild cognitive impairment stage of Alzheimer’s disease. Exp Neurol 172:433–439
Mc Khann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer disease: report of NINCDS–ADRDA work group under the auspices of Department of Health and Human Service Task Force on Alzheimer disease. Neurology 34:939–944
Müller WE, Kock S, Eckert A, Hartmann H, Scheuer K (1995) β-amyloid peptide decreases membrane fluidity. Brain Res 674:133–136
Müller WE, Koch S, Scheur K, Rostock A, Bartsch R (1997) Effects of piracetam on membrane fluidity in the aged mouse, rat, and human brain. Biochem Pharmacol 53(2):135–140
Oliveira R, Schmidt SL (1999) Teste Comportamental de Memória de Rivermead. Cognição, Rio de Janeiro
Padovani A, Borroni B, Colciaghi F, Pettenati C, Cottini E, Agosti C, Lenzi GL, Caltagirone C, Trabucchi M, Cattabeni F, Di Luca M (2002) Abnormalities in the pattern of platelet amyloid precursor protein forms in patients with mild cognitive impairment and Alzheimer disease. Arch Neurol 59:71–75
Petersen RC (2005) Mild cognitive impairment: useful or not? Alzheimer’s Dement 1:5–10
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E (1999) Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 56:303–308
Racchi M, Govoni S (2003) The pharmacology of amyloid precursor protein processing. Exp Gerontol 38:145–157
Rosenberg RN, Baskin F, Fosmire JA, Risser R, Adams P, Svetlik D, Honig LS, Cullun MC, Weiner MF (1997) Altered amyloid protein processing in platelets of patients with Alzheimer disease. Arch Neurol 54:139–144
Roth M, Hopkins B (1953) Psychological test performance in patients over 60. I. Senile psychosis and affective disorders of old age. J Ment Sci 99:439–450
Roth M, Tym E, Mountjoy CQ, Huppert FA, Hendrie H, Verma S, Goddard R (1986) CAMDEX: a standardized instrument for the diagnosis of mental disorders in the elderly with special reference to early detection of dementia. Br J Psychiatry 149:698–709
Skovronsky DM, Lee VMY, Praticò D (2001) Amyloid precursor protein and amyloid β peptide in human platelet. J Biol Chem 276:17036–17043
Sunderland T, Hill Jl, Mellow AM, Lawlor BA, Gundersheimer J, Newhouse PA, Grafman JH (1989) Clock drawing in Alzheimer’s disease. A novel measure of disease severity. J Am Geriatr Soc 37:725–729
Tang K, Hynan LS, Baskin F, Rosenberg RN (2006) Platelet amyloid precursor protein processing: a bio-marker for Alzheimer’s disease. J Neurol Sci 240:53–58
Wechsler DI (1981) Examiner’s manual: Wechsler adult intelligence scale-revised. Psychological Corporation, New York
Wilson B, Cockburn J, Baddeley AD (1985) Rivermead behavioural memory test. Thames Valley, Suffolk
Wood WG, Eckert GP, Igbavboa U, Müller WE (2003) Amyloid beta-protein interactions with membranes and cholesterol: causes or casualties of Alzheimer’s disease. Biochimica et Biophysica Acta 1610(2):281–290
Zubenko GS, Malinakova I, Chojnacki B (1987a) Proliferation of internal membranes in platelets from patients with Alzheimer’s disease. J Neuropathol Exp Neurol 46:407–418
Zubenko GS, Wusylko M, Cohen BM, Boller F, Teply I (1987b) Family study of platelet membrane fluidity in Alzheimer disease. Science 238:539–542
Zubenko GS, Kopp U, Seto T, Firestone LL (1999) Platelet membrane fluidity individuals at risk for Alzheimer’s disease: a comparison of results from fluorescence spectroscopy and electron spin resonance spectroscopy. Psychopharmacology 145:175–180
Acknowledgments
The present work was supported by FAPESP, Fundação de Amparo à Pesquisa do Estado de São Paulo (Project 02/13633-7). The authors are grateful to Dr. Breno S. O. Diniz and Carolina A. Torres. The Laboratory of Neurosciences receives financial support from Associação Beneficente Alzira Denise Hertzog da Silva.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zainaghi, I.A., Forlenza, O.V. & Gattaz, W.F. Abnormal APP processing in platelets of patients with Alzheimer’s disease: correlations with membrane fluidity and cognitive decline. Psychopharmacology 192, 547–553 (2007). https://doi.org/10.1007/s00213-007-0748-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0748-5