Abstract
Objective
Intravenous artesunate is commonly used in the emergency treatment of patients with severe falciparum malaria in Asia. The choice of doses used has been empirical. To inform dosage recommendations we assessed the pharmacokinetics of intravenous artesunate after the first dose.
Methods
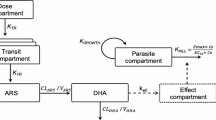
As part of a clinical trial of artesunate in adults with severe falciparum malaria in western Thailand, we assayed plasma concentrations of artesunate and the principal biologically active metabolite dihydroartemisinin (DHA) in 17 patients given an initial dose of 2.4 mg/kg body weight of intravenous artesunate. Drug levels were measured using high performance liquid chromatography with mass spectroscopy-electrospray ionisation detection.
Results
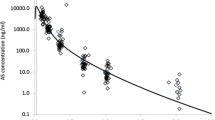
Median (range) observed DHA Cmax was 2128 (513–5789) nmol/L, elimination half-life was 0.34 (0.14–0.87) h, and the time to the last detectable DHA was 2 h.
Conclusion
The large inter-individual variability (10 fold) in DHA Cmax and AUC in patients with potentially lethal, severe malaria, suggests that 2.4 mg/kg should be the minimum daily dose in severe malaria.


Similar content being viewed by others
References
Angus BJ, Thaiaporn I, Chanthapadith K, Suputtamongkol Y, White NJ (2002) Oral artesunate dose-response relationship in acute falciparum malaria. Antimicrob Agents Chemother 46:778–782
Barradell LB, Fitton A (1995) Artesunate. Drugs 50:714–741
Batty K, Thu LTA, Davis TME, Ilett KF, Mai TX, Hung NC, Tien NP, Powell SM, Thien HV, Binh TQ, Kim NV (1998) A pharmacokinetic and pharmacodynamic study of intravenous vs oral artesunate in uncomplicated falciparum malaria. B J Clin Pharmacol 45:123–129
Bethell DB, Teja-Isavadharm P, Phuong CXT, Thuy PTT, Mai TTT, Thuy JTN, Ha NTT, Phuong PT, Kyle D, Day NJP, White NJ (1997) Pharmacokinetics of oral artesunate in children with moderately severe Plasmodium falciparum malaria. Trans R Soc Trop Med Hyg 91:195–198
Bunnag D, Viravan C, Looareesuwan S, Karbwang J, Harinasuta T (1991) Double blind randomized clinical trial of two different regimens of oral artesunate in falciparum malaria. Southeast Asian J Trop Med Publ Hlth 22:543–548
Davis TME, Phuong HL, Ilett KF, Hung NC, Batty KT, Phuong VDB, Powell SM, Thien HV, Binh TQ (2001) Pharmacokinetics and pharmacodynamics of intravenous artesunate in severe falciparum malaria. Antimicrob Agents Chemther 45:181–186
Dondorp A, Nosten F, Stepniewska K, Day N, White N, South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group (2005) Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet 366:717–725
Hien TT, Davis TME, Chuong LV, Ilett KF, Mai NTH, Chau TTH, Agus C, Chiswell GM, White NJ, Farrar J (2004) Comparative pharmacokinetics of intramuscular artesunate and artemether in severe falciparum malaria. Antimicrob Agents Chemother 48:4234–4239
Li Q, Guo X, Jiang R (1982) Clinical studies on treatment of cerebral malaria with qinghaosu and its derivatives. J Trad Chin Med 2:125–130
Meshnick SR, Taylor TE, Kamchonwongpaisan S (1996) Artemisinin and the antimalarial endoperoxides: from herbal remedy to targeted chemotherapy. Micro Rev 60:301–315
Na-Bangchang K, Karbwang J, Congpoung K, Thanavibul A, Ubalee R (1998) Pharmacokinetic and bioequivalence evaluation of two generic formulations of oral artesunate. Eur J Clin Pharmacol 53:375–376
Newton PN, Suputtamongkol Y, Teja-Isavadharm P, Pukrittayakamee S, Navartnam V, Bates I, White NJ (2000) Antimalarial bioavailability and disposition of artesunate in acute falciparum malaria. Antimicrob Agents Chemother 44:972–977
Newton PN, Angus BJ, Chierakul W, Dondorp A, Ruangveerayuth R, Silamut K, Teerapong P, Suputtamongkol Y, Looareesuwan S, White NJ (2003) A randomised comparison of artesunate and quinine in the treatment of severe falciparummalaria. Clin Inf Dis 37:7–16
Newton PN, Ward S, Angus BJ, Chierakul W, Dondorp A, Ruangveerayuth R, Silamut K, Teerapong P, Suputtamongkol Y, Looareesuwan S, White NJ (2005) Early treatment failure in severe malaria resulting from abnormally low plasma quinine concentrations. Trans Royal Soc Trop Med Hyg 100:184–186
Nosten F, Luxemburger C, ter Kuile FO, Woodrow C, Pa Eh J, Chongsuphajaisiddhi T, White NJ (1994) Treatment of multidrug resistant falciparum malaria with a 3 day artesunate-mefloquine combination. J Infect Dis 170:971–977
Sangster T, Spence M, Sinclair P, Payne R, Smith C (2004) Unexpected observation of ion suppression in a liquid chromatography/atmospheric pressure chemical ionization mass spectrometric bioanalytical method. Rapid Comm Mass Spec 18:1361–1364
Silamut K, Hough R, Eggelte T, Pukrittayakamee S, Angus B, White NJ (1995) A simple method for assessing quinine pre-treatment in acute malaria. Trans R Soc Trop Med Hyg 89:665–667
Souppart C, Gauducheau N, Sandrenan N, Richard F (2002) Development and validation of a HPLC-MS assay for determination of AM and DHA in human plasma. J Chrom B 774:195–203
White NJ (1994) Clinical pharmacokinetics and pharmacodynamics of artemisinin and derivatives. Trans R Soc Trop Med Hyg 88 Suppl 1:S41–43
White NJ (1996) The treatment of malaria. N Eng J Med 335:800–806
White NJ (1997) Assessment of the pharmacodynamic properties of antimalarial drugs in-vivo. Antimicrob Agents Chemother 41:1413–1422
Acknowledgements
This study was a part of the Wellcome Trust-Mahidol University Oxford Tropical Medicine Research Programme, funded by the Wellcome Trust of Great Britain. We are very grateful to the Director, doctors and nurses of Mae Sot Hospital, Thanongsak Teewarakulpana, Paktiya Teja-Isavadharm, Kamolrat Silamut, Kesinee Chotivanich, Sayan Langla, Kongpop Pupae, Nitirat Thima, Niklas Lindegårdh and Nicholas Day for their help and advice.
[The study complies with the laws of Thailand inclusive of ethical approval].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Newton, P.N., Barnes, K.I., Smith, P.J. et al. The pharmacokinetics of intravenous artesunate in adults with severe falciparum malaria. Eur J Clin Pharmacol 62, 1003–1009 (2006). https://doi.org/10.1007/s00228-006-0203-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-006-0203-2