Abstract
Purpose
To compare interview data on drug use during pregnancy with data identified from a register of prescriptions.
Materials
We compared information from the Swedish Prescribed Drug Register with the Swedish Medical Birth Register on antidepressant use. In order to evaluate the clinical significance of the difference in ascertainment with the two methods, the rate of preterm births among singletons and of neonatal symptoms were studied.
Results
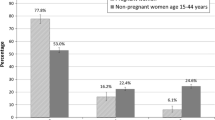
During the year before the last menstrual period, 1.5% of the women filled prescriptions for antidepressants each month. Already before the pregnancy was known, the rate of filled prescriptions decreased and reached 0.5% towards the end of the pregnancy. Twenty-two percent of first-trimester use of antidepressants was unidentified using interview data and prescriptions during the 2nd and 3rd pregnancy months covered only 55% of actual use. When women who filled prescriptions 1 or 3 months before the last menstrual period were included, 17 and 43% respectively of women were included who probably did not use the drugs in the first trimester. Prescriptions gave a more complete ascertainment of drug use after the first trimester than data from antenatal care, which seemed to overestimate drug use.
Conclusions
Interview data seem to give the most valid results on early use. When interview data are not available, prescription data could be used, but should not include prescriptions given earlier than 1 month before the last menstrual period. Studies of drug use later in pregnancy are best based on prescription data in the absence of interview data.

Similar content being viewed by others
References
Källén B (2005) Methodological issues in the epidemiological study of the teratogenicity of drugs. Congenit Anom 45:44–51
Chambers CD, Johnson KA, Dick LM, Felix RJ, Jones KL (1996) Birth outcomes in pregnant women taking fluoxetine. N Engl J Med 335:1010–1015
McElhatton PR, Garbis HM, Elefant E, Vial T, Bellemin B, Mastroiacovo P, Arnon J, Rodriguez-Pinilla E, Schaefer C, Pexieder T, Merlob P, Dal Verme S (1996) The outcome of pregnancy in 689 women exposed to therapeutic doses of antidepressants. A collaborative study of the European Network of Teratology Information Services (ENTIS). Reprod Toxicol 10:285–294
Diav-Citrin O, Schechtman S, Weinbaum D, Wajnberg R, Avgil M, Di Gianantonio E, Clementi M, Weber-Schoendorfer C, Schaefer C, Ornoy A (2008) Paroxetine and fluoxetine in pregnancy: a prospective, multicentre, controlled, observational study. Br J Clin Pharmacol 66:695–705
Oberlander TF, Warburton W, Misri S, Aghajanian J, Hertzman C (2008) Effect of timing and duration of gestational exposure to serotonin reuptake inhibitor antidepressants: population-based study. Br J Psychiatry 192:338–343
Cole JA, Ephross SA, Cosmatos IS, Walker AM (2007) Paroxetine in the first trimester and the prevalence of congenital malformations. Pharmacoepidemiol Drug Saf 16:1075–1085
Malm H, Klaukka T, Neuvonen PJ (2005) Risks associated with selective serotonin reuptake inhibitors in pregnancy. Obstet Gynecol 106:1289–1296
Wogelius P, Norgaard M, Gislum M, Pedersen L, Munk E, Mortensen PB, Lipworth L, Sørensen HT (2006) Maternal use of selective serotonin reuptake inhibitors and risk of congenital malformations. Epidemiology 17:701–704
Pedersen LH, Henriksen TB, Vestergaard M, Olsen J (2009) Selective serotonin reuptake inhibitors in pregnancy and congenital malformations: population based cohort study. BMJ 339:b3569
Espnes MG, Bjørge T, Engeland A (2011) Comparison of recorded medication use in the Medical Birth Registry of Norway with prescribed medicines registered in the Norwegian Prescription Database. Pharmacoepidemiol Drug Saf 20:243–248
Stephansson O, Granath F, Svensson T, Haglund B, Ekbom A, Kieler H (2011) Drug use during pregnancy in Sweden—assessed by the Prescribed Drug Register and the Medical Birth Register. Clin Epidemiol 3:43–50
National Board of Health and Welfare, Centre for Epidemiology (2003) The Swedish Medical Birth Registry—a summary of content and quality. Available at: http://www.sos.se/publikationer2003/2003-112-3
Reis M, Källén B (2010) Delivery outcome after maternal use of antidepressant drugs in pregnancy: an update using Swedish data. Psychol Med 10:1723–1733
Wettermark B, Hammar N, Fored CM, Leimanis A, Otterblad Olausson P, Bergman U, Persson I, Sundström A, Westerholm B, Rosén M (2007) The new Swedish Prescribed Drug Register—opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf 16:726–735
Källén B, Otterblad Olausson P (2001) Monitoring of maternal drug use and infant congenital malformations: does loratadine cause hypospadias? Int J Risk Saf Med 14:115–119
Acknowledgement
The study was supported by a grant from Evy and Gunnar Sandberg Foundation, Lund to BK.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Källén, B., Nilsson, E. & Olausson, P.O. Antidepressant use during pregnancy: comparison of data obtained from a prescription register and from antenatal care records. Eur J Clin Pharmacol 67, 839–845 (2011). https://doi.org/10.1007/s00228-011-1021-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1021-8