Abstract
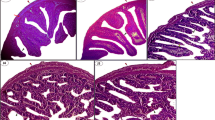
Nigerian Bonny light crude oil (BLCO) is commonly used by the local population in folklore medicine for the management of various forms of gastrointestinal problems and male reproductive capacity. The study investigated the effects of BLCO on the antioxidant systems of the testes and epidydimal sperm in rats by oral exposure to 0, 200, 400 and 800 mg/kg BLCO for 7 days. In testes and sperm, BLCO treatment at all doses significantly (p < 0.05) decreased superoxide dismutase (SOD), catalase (CAT), and glutathione S-transferase (GST) activities, whereas it markedly increased glucose 6-phosphate dehydrogenase (G6PD) and gamma glutamyl transferase (GGT) activities as well as increased glutathione (GSH), hydrogen peroxide, and malondialdehyde (MDA) levels in all treatment groups. Although epididymal sperm number (ESN), daily spermatozoa production (DSP), and sperm motility were significantly decreased, total sperm abnormalities were significantly increased without affecting sperm viability at all dose levels compared with controls. The adverse effect of BLCO on TSN was noted at the 800 mg/kg dose only. Histopathology results showed treatment-related lesions of the testes characterized by severe congestion of interstitial vessels, decreased germinal epithelium, and increased number of vacuolization. These results suggest that exposure to BLCO, such as its use in ailment management, may promote infertility by altering the function of the testes and sperm, particularly by way of induction of oxidative stress.




Similar content being viewed by others
References
Abarikwu SO, Adesiyan AC, Oyejola TO, Oyeyemi MO, Farombi EO (2009) Changes in sperm characteristics and induction of oxidative stress in the testis and epididymis of experimental rats by a herbicide, atrazine. Arch Environ Contam Toxicol. doi:10.1007/s00244-009-9371-2
Aitken RJ, Buckingham D, Harkiss D (1993) Relationship between iron-catalyzed lipid peroxidation potential and human sperm function. J Reprod Fertil 98:257–265
Aitkin RJ, Clarkson JS, Fishel S (1989) Generation of reactive oxygen species, lipid peroxidation and human sperm function. Biol Reprod 41:183–187
Akintonwa A, Ebere AG (1990) Toxicity of Nigerian crude oil and chemical dispersants to Barbus Sp. and Clarias Sp. Bull Environ Contam Toxicol 45:729–733
Alvarez JG, Touchstone JC, Blasco L, Storey B (1987) Spontaneous lipid peroxidation and production of hydrogen peroxide and superoxide in human spermatozoa. J Androl 8:338–348
Amstad P, Peskin A, Shah AG, Mirault ME, Moret R, Zbinden I et al (1991) The balance between Cu, Zn-superoxide dismutase and catalase affects the sensitivity of mouse epidermal cells to oxidative stress. Biochemistry 30:9305–9313
Bauche F, Fouchard H, Jegou B (1994) Antioxidant system in rat testicular cells. FEBS Lett 349:392–396
Carpenter DO, Arcaro K, Spink DC (2002) Understanding the human health effects of chemical mixtures. Environ Health Perspect 110:25–42
Clairborne A (1995) Catalase activity. In: Greewald AR (ed) Handbook of methods for oxygen radical research. CRC Press, Boca Raton, FL, pp 237–242
CONCAWE (2001) Environmental classification of petroleum substances—summary data and rationale. Report No. 01/54. CONCAWE, Brussels
Dawson JP, Thayer WW, Desforges JF (1958) Acute hemolytic anaemia in the newborn infant due to naphthalene poisoning: report of two cases with investigations into the mechanism of the disease. Hematology 13:1113–1125
Dickinson DA, Moellering DR, Iles KE, Patel RP, Levonen AL, Wigley A et al (2003) Cytoprotection against oxidative stress and the regulation of glutathione synthesis. Biol Chem 384:527–537
Dimitrova MST, Tsinova V, Velcheva V (1994) Combined effect of zinc and lead on the hepatic superoxide dismutase-catalase system in carp (cyprinus carpio). Comp Biochem Physiol 108:43–46
Farombi EO, Tahnteng JG, Agboola AO, Nwankwo JO, Emerole GO (2000) Chemoprevention of 2-acetylaminofluorene-induced hepatotoxicity and lipid peroxidation in rats by kolaviron-a garcinia kola seed extract. Food Chem Toxicol 38:353–541
Farombi EO, Abarikwu SO, Adedara IA, Oyeyemi MO (2007) Curcumin and Kolaviron ameliorate Di-n-butylphthalate-induced testicular damage in rats. Basic Clin Pharmacol Toxicol 100:43–48
Farombi EO, Ugwuezunmba MC, Ezenwadu TT, Oyeyemi MO, Ekor M (2008) Tetracycline-induced reproductive toxicity in male rats: Effects of vitamin C and N-acetylcysteine. Exp Toxicol Pathol 60:77–85
Fraga CG, Motchnik PA, Shigenaga MK, Helbock HJ, Jacob RA, Ames BN (1991) Ascorbic acid protects against endogenous oxidative damage in human sperm. Proc Natl Acad Sci USA 88:11003–11006
Franca LR, Leal MC, Sasso-Cerri E, Vasconcelos A, Debeljuk L, Russell LD (2000) Cimetidine (Tagamet) is a reproductive toxicant in male rats affecting peritubular cells. Biol Reprod 63:1403–1412
Fridovich I (1995) Superoxide radical and superoxide dismutases. Annu Rev Biochem 64:97–112
Girotti AW (1985) Mechanisms of lipid peroxidation. J Free Radic Biol Med 1:87–95
Giwa-Osagie OO (2003) Nigeria has twelve million infertile persons. Pharmanews 25:48–49
Griendling KK, Sorescu D, Ushio-Fukai M (2000) NADPH oxidase: role in cardiovascular biology and disease. Circ Res 86:494–501
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferase. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hasspielar BM, Behar JV, DiGiulio RT (1994) Glutathione dependent defense in channel catfish (Ictalurus punctatus) and brown bullhead (Ameiurus nebulosus). Ecotoxicol Environ Saf 28:82–90
Iwasaki A, Gagnon C (1992) Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil Steril 57:409–416
Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR (1974) Bromobenzene induced liver necrosis: Protective role of glutathione and evidence for 3,4 bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 11:151–169
Joyce KL, Porcelli J, Cooke PS (1993) Neonatal goitrogen treatment increases adult testes size and sperm production in the mouse. J Androl 14:448–455
Kumar S (2008) Is environmental exposure associated with reproductive health impairments? J Turk Ger Gynecol Assoc 9:60–69
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Lucesoli F, Caligiuri M, Roberti MF, Perazzo JC, Fraga CG (1999) Dose-dependent increase of oxidative damage in the testis of rats subjected to acute iron overload. Arch Biochem Biophys 372:37–43
Markelewicz RJ, Hall SJ, Boekeldeide K (2004) 2,5-hexanedione and carbendazim coexposure synergistically disrupts rats spermatogenesis despite opposing molecular effects on microtubules. Toxicol Sci 80:92–100
McLachlan R, de Krester D (2001) Male infertility: the case for continued research. Med J Aust 174:116–117
Misra HP, Fridovich I (1972) The role of superoxide anion in the auto-oxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Oehninger S (2001) Strategies for the infertile man. Semin Reprod Med 19:231–237
Orisakwe OE, Njan AA, Afonne JO, Akumka DD, Orish N, Udemezue OO (2004a) Investigation into the nephrotoxicity of Nigerian bonny light crude oil in albino rats. Int J Environ Res Public Health 1:106–110
Orisakwe OE, Akumka DD, Njan AA, Afonne JO (2004b) Testicular toxicity of Nigerian bonny light crude oil in male albino rats. Reprod Toxicol 18:439–442
Oruambo IF, Jones AB (2007) Alterations in the concentrations of liver mitochondrial DNA: cytoplasmic total hydrocarbon and calcium in guinea pigs after treatment with Nigerian light crude oil. Int J Environ Res Public Health 4:23–27
Oruambo IF, Kachikwu S, Idabor L (2007) Dose-related increased binding of nickel to chromatin proteins and changes to DNA concentration in the liver of guinea pigs treated with Nigerian light crude oil. Int J Environ Res Public Health 4:211–215
Pant N, Srivastava SP (2003) Testicular and spermatotoxic effects of quinalphos in rats. J Appl Toxicol 23:271–274
Pant N, Prasad AK, Srivastava SC, Shanker R, Srivastava SP (1995) Effect of oral administration of carbofuran on reproductive system of rat. Hum Exp Toxicol 14:889–894
Perreault SD (1998) Gamete toxicology: the impact of new technologies. In: Korach KS (ed) Reproductive and developmental toxicology. Marcel Dekker, New York, NY, pp 635–654
Sharlip ID, Jarow JP, Belker AM, Lipshultz LI, Sigman M, Thomas AJ et al (2002) Best practice policies for male infertility. Fertil Steril 77:873–882
Stephensen E, Sturve J, Forlin L (2002) Effect of redox cycling compounds on glutathione content and activity of glutathione-related enzymes in rainbow trout liver. Comp Biochem Physiol C 133:435–442
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med 18:321–336
Szasz G (1974) Gamma glutamyltranspeptidase. In: Bergmeye A (ed) Methods of enzymatic analysis, 2nd edn. Academic Press, New York, NY, p 715
Teuschler L, Klauring J, Carney E, Chambers J, Conolly R, Gennings C et al (2002) Support of the science-based decisions concerning the evaluation of the toxicology of mixtures: a new beginning. Regul Toxicol Pharmacol 36:34–39
Wells ME, Awa OA (1970) New technique for assessing acrosomal characteristics of spermatozoa. J Dairy Sci 53:227
Wolff SP (1994) Ferrous ion oxidation in the presence of ferric ion indicator xylenol orange for measurement of hydroperoxides. Methods Enzymol 233:182 (Academic Press 189)
Zemjanis R (1970) Collection and evaluation of semen. In: Diagnostic and therapeutic technique in animal reproduction, 2nd ed. William and Wilkins, Baltimore, MD, pp 139–153
Acknowledgments
The authors gratefully acknowledge the technical assistance of Omoko Ejiro of the Department of Veterinary Surgery and Reproduction, University of Ibadan. This work was supported by Senate Research Grant Code No. SRG/Com/2006/34A from the University of Ibadan, Nigeria (E. O. F.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farombi, E.O., Adedara, I.A., Ebokaiwe, A.P. et al. Nigerian Bonny Light Crude Oil Disrupts Antioxidant Systems in Testes and Sperm of Rats. Arch Environ Contam Toxicol 59, 166–174 (2010). https://doi.org/10.1007/s00244-009-9443-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-009-9443-3