Abstract
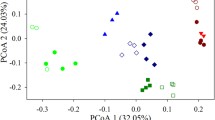
The spatial distribution of microbial communities has recently been reliably documented in the form of a distance–similarity decay relationship. In contrast, temporal scaling, the pattern defined by the microbial similarity–time relationships (STRs), has received far less attention. As a result, it is unclear whether the spatial and temporal variations of microbial communities share a similar power law. In this study, we applied the 454 pyrosequencing technique to investigate temporal scaling in patterns of bacterioplankton community dynamics during the process of shrimp culture. Our results showed that the similarities decreased significantly (P = 0.002) with time during the period over which the bacterioplankton community was monitored, with a scaling exponent of w = 0.400. However, the diversities did not change dramatically. The community dynamics followed a gradual process of succession relative to the parent communities, with greater similarities between samples from consecutive sampling points. In particular, the variations of the bacterial communities from different ponds shared similar successional trajectories, suggesting that bacterial temporal dynamics are predictable to a certain extent. Changes in bacterial community structure were significantly correlated with the combination of Chl a, TN, PO4 3-, and the C/N ratio. In this study, we identified predictable patterns in the temporal dynamics of bacterioplankton community structure, demonstrating that the STR of the bacterial community mirrors the spatial distance–similarity decay model.






Similar content being viewed by others
References
Allison SD, Martiny JBH (2008) Resistance, resilience, and redundancy in microbial communities. Proc Natl Acad Sci U S A 105:11512–11519
APHA (1976) Standard methods for the examination of water and wastewater 14ed. APHA American Public Health Association
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010) QIIME allows integration and analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Case M, Leca EE, Leitaoc SN, SantAnna EE, Schwamborn R, De Moraes Junior AT (2008) Plankton community as an indicator of water quality in tropical shrimp culture ponds. Mar Pollut Bull 56:1343–1352
Clarke KR (1993) Non-parametric multivariate analysis of changes in community structure. Aust J Ecol 18:117–143
Comte J, Del Giorgio PA (2010) Linking the patterns of change in composition and functional capacities in bacterioplankton successions along environmental gradients. Ecology 95:1466–1476
Comte J, Del Giorgio PA (2011) Composition influences the pathway but not the outcome of the metabolic response of bacterioplankton to resource shifts. PLoS One 6:e25266
Cotner JB, Biddanda BA (2002) Small players, large role: Microbial influence on biogeochemical processes in pelagic aquatic ecosystems. Ecosystems 5:105–121
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K et al (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72:5069–5072
DeSantis TZ, Hugenholtz P, Keller K, Brodie EL, Larsen N, Piceno YM et al (2006) NAST: a multiple sequence alignment server for comparative analysis of 16S rRNA genes. Nucleic Acids Res 34:394–399
Domingues RB, Barbosa A, Galvão H (2008) Constraints on the use of phytoplankton as a biological quality element within the water framework directive in Portuguese waters. Mar Pollut Bull 56:1389–1395
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Ferreira NC, Bonetti C, Seiffert WQ (2011) Hydrological and water quality indices as management tools in marine shrimp culture. Aquaculture 318:425–433
Fierer N, Nemergut DR, Knight R, Crainej JM (2010) Changes through time: integrating microorganisms into the study of succession. Res Microbiol 161:635–642
Gilbert JA, Field D, Swift P, Newbold L, Oliver A, Smyth T et al (2009) The seasonal succession of microbial communities in the Western English Channel using 16S rDNA-tag pyrosequencing. Environ Microbiol 11:3132–3139
Green JL, Holmes AJ, Westoby M, Oliver I, Briscoe D, Dangerfield M et al (2004) Spatial scaling of microbial eukaryote diversity. Nature 432:747–750
Griffiths RI, Thomson BC, James P, Bell T, Bailey M, Whiteley AS (2011) The bacterial biogeography of British soils. Environ Microbiol 13:1642–1654
Goldfarb KC, Karaoz U, Hanson CA, Santee CA, Bradford MA, Treseder KK, Wallenstein MD, Brodie EL (2011) Differential growth responses of soil bacterial taxa to carbon substrates of varying chemical recalcitrance. Front Microbiol 2:1–10
Gómez-Consarnau L, Lindh MV, Gasol JM, Pinhassi J (2012) Structuring of bacterioplankton communities by specific dissolved organic carbon compounds. Environ Microbiol 14:2361–2378
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Johnson EA, Miyanishi K (2008) Testing the assumptions of chronosequences in succession. Ecol Lett 11:419–431
Jones SE, Cadkin TA, Newton RJ, McMahon KD (2012) Spatial and temporal scales of aquatic bacterial beta diversity. Front Microbiol 3:318
Kim Y, Jeong J, Wells GF, Park H (2013) General and rare bacterial taxa demonstrating different temporal dynamic patterns in an activated sludge bioreactor. Appl Microbiol Biotechnol 97:1755–1765
Lemonnier H, Herbland A, Salery L, Soulard B (2006) “Summer syndrome” in Litopenaeus stylirostris grow out ponds in New Caledonia: zootechnical and environmental factors. Aquaculture 261:1039–1047
Lemonnier H, Courties C, Mugnier C, Torréton J, Herbland A (2010) Nutrient and microbial dynamics in eutrophying shrimp ponds affected or unaffected by vibriosis. Mar Pollut Bull 60:402–411
Legendre P, Legendre L (1998) Numerical ecology, 2nd English edn. Developments in environmental modeling. Dev Environ Model 20:1–853
Lucas R, Courties C, Herbland A, Goulletquer P, Marteau AL, Lemonnier H (2010) Etrophication in a tropical pond: understanding the bacterioplankton and phytoplankton dynamics during a vibriosis outbreak using flow cytometric analyses. Aquaculture 310:112–121
Lundin D, Severin I, Logue JB, Östman Ö, Andersson AF, Lindström ES (2012) Which sequencing depth is sufficient to describe patterns in bacterial α- and β-diversity? Environ Microbiol Rep 4:367–372
Ma Z, Song X, Wan R, Gao L (2013) A modified water quality index for intensive shrimp ponds of Litopenaeus vannamei. Ecol Indic 24:287–293
Or A, Shtrasler L, Gophna U (2012) Fine-scale temporal dynamics of a fragmented lotic microbial ecosystem. Sci Rep 2:207
Paver SF, Hayek KR, Gano KA, Fagen JR, Brown CT et al (2013) Interactions between specific phytoplankton and bacteria affect lake bacterial community succession. Environ Microbiol 15:2489–2504
Preston FW (1960) Time and space and the variation of species. Ecology 41:612–627
R Development Team (2012) R: A language and environment for statistical computing. http://cran.r-project.org
Redford A, Fierer N (2009) Bacterial succession on the leaf surface: a novel system for studying successional dynamics. Microb Ecol 58:189–198
Shade A, Caporaso JG, Handelsman J, Knight R, Fierer N (2013) A meta-analysis of changes in bacterial and archaeal communities with time. ISME J 7:1493–1506
Sørensen TA (1948) A method of establishing groups of equal amplitude in plant sociology based on similarity of species and its application to analyses of the vegetation on Danish commons. Biol Skr 5:1–34
Snieszko SF (1974) The effect of environmental stress on outbreaks of infectious diseases of fish. J Fish Biol 6:197–208
Sung H, Hsu S, Chen C, Ting Y, Chao W (2001) Relationships between disease outbreak in cultured tiger shrimp (Penaeus monodon) and the composition of Vibrio communities in pond water and shrimp hepatopancreas during cultivation. Aquaculture 192:101–110
Teeling H, Fuchs BM, Becher D, Klockow C, Gardebrecht A, Bennke CM et al (2012) Substrate-controlled succession of marine bacterioplankton populations induced by a phytoplankton bloom. Science 336:608–611
Van Der Gast CJ, Ager D, Lilley AK (2008) Temporal scaling of bacterial taxa is influenced by both stochastic and deterministic ecological factors. Environ Microbiol 10:1411–1418
Wang J, Shen J, Wu Y, Tu C, Soininen J, Stegen JC et al (2013) Phylogenetic beta diversity in bacterial assemblages across ecosystems: deterministic versus stochastic processes. ISME J 7:1310–1321
Wells GF, Park HD, Eggleston B, Francis CA, Criddle CS (2011) Fine-scale bacterial community dynamics and the taxa–time relationship within a full-scale activated sludge bioreactor. Water Res 45:5476–5488
White EP, Adler PB, Lauenroth WK, Gill RA, Greenberg D et al (2006) A comparison of the species–time relationship across ecosystems and taxonomic groups. Oikos 112:185–195
Wu QL, Hahn MW (2006) High predictability of the seasonal dynamics of a species-like Polynucleobacter population in a freshwater lake. Environ Microbiol 8:1660–1666
Xiong J, Wu L, Tu S, Van Nostrand JD, He Z, Zhou J, Wang G (2010) Microbial communities and functional genes associated with soil arsenic contamination and rhizosphere of the arsenic hyper-accumulating plant Pteris vittata L. Appl Environ Microbiol 76:7277–7284
Xiong J, He Z, Van Nostrand JD, Luo G, Tu S, Zhou J, Wang G (2012) Assessing the microbial community and functional genes in a vertical soil profile with long-term arsenic contamination. PLoS One 7:e50507
Xiong J, Liu Y, Lin X, Zhang H, Zeng J, Hou J et al (2012) Geographic distance and pH drive bacterial distribution in alkaline lake sediments across Tibetan Plateau. Environ Microbiol 14:2457–2466
Xu H, Zhu M, Jiang Y, Al-Rasheid KS (2010) Temporal species distributions of planktonic protist communities in semi-enclosed mariculture waters and responses to environmental stress. Acta Oceanol Sin 29:74–83
Zhang D, Wang X, Xiong J, Zhu J, Wang Y, Zhao Q et al (2013) Bacterioplankton assemblages as biological indicators for shrimp healthy states. Ecol Indic. doi:10.1016/j.ecolind.2013.11.002
Zhou J, Liu W, Deng Y, Jiang Y, Xue K, He s et al (2013) Stochastic assembly leads to alternative communities with distinct functions in a bioreactor microbial community. mBio 4:e00584–12
Acknowledgments
This work was financially supported by the National High Technology Research and Development Program of China (863 Program, 2012AA092000), the Science and Technology Project of the Ministry of Education (Grant No. 208053), the Natural Science Foundation of Ningbo City (2013A610169), the Research Fund from 2011 Center of Modern Marine Aquaculture of East China Sea, and the KC Wong Magna Fund of Ningbo University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiong and Zhu contributed equally to this work
Rights and permissions
About this article
Cite this article
Xiong, J., Zhu, J., Wang, K. et al. The Temporal Scaling of Bacterioplankton Composition: High Turnover and Predictability during Shrimp Cultivation. Microb Ecol 67, 256–264 (2014). https://doi.org/10.1007/s00248-013-0336-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-013-0336-7