Abstract
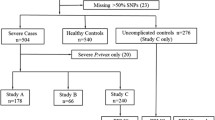
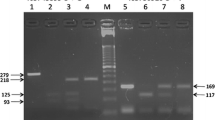
The clinical course of malaria varies between affected individuals and host genetic factors have been shown to influence the outcome of malaria. The role of FOXO3-driven pathway in modulating inflammatory responses, including mediation of distinct functions of regulatory T effector cell populations (Tregs) by the transcription factor FOXO3, has recently been recognized. We aimed to study possible associations of a non-coding polymorphism in intron 2 of the FOXO3A gene (rs12212067T>G) that was shown earlier to modulate the FOXO3 expression and to be associated with the prognosis of distinct inflammatory and infectious diseases. The FOXO3A polymorphism rs12212067T>G was genotyped by direct sequencing in a group of Gabonese children with confirmed Plasmodium falciparum malaria. Severe cases of malaria were compared with asymptomatic/mild cases. The FOXO3A variant rs12212067T>G was associated with the phenotype of severe malaria, but not with asymptomatic/mild malaria (allelic model: OR = 1.54, 95 % CI = 1.15–2.05, P = 0.0028; dominant model: OR = 1.94, 95 % CI = 1.36–2.77, P = 0.0002). The FOXO3A variant rs12212067T>G is associated with increased inflammatory responses to Plasmodium falciparum malaria, indicating a role of the FOXO3-dependent pathway in malaria.
Similar content being viewed by others
References
Alessi DR, James SR, Downes CP, Holmes AB, Gaffney PR, Reese CB, Cohen P (1997) Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Balpha. Curr Biol 7:261–269
Brustoski K, Moller U, Kramer M, Hartgers FC, Kremsner PG, Krzych U, Luty AJ (2006) Reduced cord blood immune effector-cell responsiveness mediated by CD4+ cells induced in utero as a consequence of placental Plasmodium falciparum infection. J Infect Dis 193:146–154
Carter ME, Brunet A (2007) FOXO transcription factors. Curr Biol 17:R113–R114
Dejean AS, Beisner DR, Ch’en IL, Kerdiles YM, Babour A, Arden KC, Castrillon DH, DePinho RA, Hedrick SM (2009) Transcription factor FOXO3 controls the magnitude of T cell immune responses by modulating the function of dendritic cells. Nat Immunol 10:504–513
Eijkelenboom A, Mokry M, de Wit E, Smits LM, Polderman PE, van Triest MH, van Boxtel R, Schulze A, de Laat W, Cuppen E, Burgering BM (2013) Genome-wide analysis of FOXO3 mediated transcription regulation through RNA polymerase II profiling. Mol Syst Biol 9:638
Essaghir A, Dif N, Marbehant CY, Coffer PJ, Demoulin JB (2009) The transcription of FOXO genes is stimulated by FOXO3 and repressed by growth factors. J Biol Chem 284:10334–10342
Esser C, Tomluk J (2005) Reporting Hardy-Weinberg tests in case-control studies: reasons for caution but not for panic reactions. J Investig Dermatol 124:1082–1083
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567
Hanel SA, Velavan TP, Kremsner PG, Kun JF (2011) Novel and functional regulatory SNPs in the promoter region of FOXP3 gene in a Gabonese population. Immunogenetics 63:409–415
Hedrick SM (2009) The cunning little vixen: FOXO and the cycle of life and death. Nat Immunol 10:1057–1063
Hedrick SM, Hess MR, Doedens AL, Goldrath AW, Stone EL (2012) FOXO transcription factors throughout T cell biology. Nat Rev Immunol 12:649–661
Huang X, Kuhne V, Kun JF, Soboslay PT, Lell B, Tp V (2012) In-vitro characterization of novel and functional regulatory SNPs in the promoter region of IL2 and IL2R alpha in a Gabonese population. BMC Med Genet 13:117
Kalmbach Y, Boldt AB, Fendel R, Mordmuller B, Kremsner PG, Kun JF (2006) Increase in annexin V-positive B cells expressing LILRB1/ILT2/CD85j in malaria. Eur Cytokine Netw 17:175–180
Karlsson EK, Kwiatkowski DP, Sabeti PC (2014) Natural selection and infectious disease in human populations. Nat Rev Genet 15:379–393
Kerdiles YM, Stone EL, Beisner DR, McGargill MA, Ch’en IL, Stockmann C, Katayama CD, Hedrick SM (2010) FOXO transcription factors control regulatory T cell development and function. Immunity 33:890–904
Koukouikila-Koussounda F, Ntoumi F, Ndounga M, Tong HV, Abena AA, Velavan TP (2013) Genetic evidence of regulatory gene variants of the STAT6, IL10R and FOXP3 locus as a susceptibility factor in uncomplicated malaria and parasitaemia in Congolese children. Malar J 12:9
Kun JF, Mordmuller B, Lell B, Lehman LG, Luckner D, Kremsner PG (1998) Polymorphism in promoter region of inducible nitric oxide synthase gene and protection against malaria. Lancet 351:265–266
Lee JC, Espeli M, Anderson CA, Linterman MA, Pocock JM, Williams NJ, Roberts R, Viatte S, Fu B, Peshu N, Hien TT, Phu NH, Wesley E, Edwards C, Ahmad T, Mansfield JC, Gearry R, Dunstan S, Williams TN, Barton A, Vinuesa CG, Parkes M, Lyons PA, Smith KG (2013) Human SNP links differential outcomes in inflammatory and infectious disease to a FOXO3-regulated pathway. Cell 155:57–69
Levings MK, Bacchetta R, Schulz U, Roncarolo MG (2002) The role of IL-10 and TGF-beta in the differentiation and effector function of T regulatory cells. Int Arch Allergy Immunol 129:263–276
Litvak V, Ratushny AV, Lampano AE, Schmitz F, Huang AC, Raman A, Rust AG, Bergthaler A, Aitchison JD, Aderem A (2012) A FOXO3-IRF7 gene regulatory circuit limits inflammatory sequelae of antiviral responses. Nature 490:421–425
Maizels RM (2009) Parasite immunomodulation and polymorphisms of the immune system. J Biol 8:62
Malaguarnera L, Musumeci S (2002) The immune response to Plasmodium falciparum malaria. Lancet Infect Dis 2:472–478
Miller LH, Baruch DI, Marsh K, Doumbo OK (2002) The pathogenic basis of malaria. Nature 415:673–679
Sakaguchi S (2000) Regulatory T cells: key controllers of immunologic self-tolerance. Cell 101:455–458
Sakaguchi S (2004) Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu Rev Immunol 22:531–562
Scholzen A, Mittag D, Rogerson SJ, Cooke BM, Plebanski M (2009) Plasmodium falciparum-mediated induction of human CD25FOXP3 CD4 T cells is independent of direct TCR stimulation and requires IL-2, IL-10 and TGFbeta. PLoS Pathog 5:e1000543
Stokoe D, Stephens LR, Copeland T, Gaffney PR, Reese CB, Painter GF, Holmes AB, McCormick F, Hawkins PT (1997) Dual role of phosphatidylinositol-3,4,5-trisphosphate in the activation of protein kinase B. Science 277:567–570
Taylor A, Verhagen J, Blaser K, Akdis M, Akdis CA (2006) Mechanisms of immune suppression by interleukin-10 and transforming growth factor-beta: the role of T regulatory cells. Immunology 117:433–442
Timmann C, Thye T, Vens M, Evans J, May J, Ehmen C, Sievertsen J, Muntau B, Ruge G, Loag W, Ansong D, Antwi S, Asafo-Adjei E, Nguah SB, Kwakye KO, Akoto AO, Sylverken J, Brendel M, Schuldt K, Loley C, Franke A, Meyer CG, Agbenyega T, Ziegler A, Horstmann RD (2012) Genome-wide association study indicates two novel resistance loci for severe malaria. Nature 489:443–446
Velavan TP, Ojurongbe O (2011) Regulatory T cells and parasites. J Biomed Biotechnol 2011:520940
Velavan TP, Bechlars S, Tomiuk J, Kremsner PG, Kun JF (2011) Molecular characterization of regulatory polymorphisms in the promoter region of the STAT6 gene in a Gabonese population. Mem Inst Oswaldo Cruz 106:65–69
Velavan TP, Buyukyazici B, Kremsner PG, Kun JF (2012) Combined promoter haplotypes of the IL10R genes are associated with protection against severe malaria in Gabonese children. Immunogenetics 64:87–95
Vigario AM, Gorgette O, Dujardin HC, Cruz T, Cazenave PA, Six A, Bandeira A, Pied S (2007) Regulatory CD4+ CD25+ FOXP3+ T cells expand during experimental Plasmodium infection but do not prevent cerebral malaria. Int J Parasitol 37:963–973
Wang Y, Zhou L, Chen J, Li J, He L, Wu P, Wang M, Tong N, Zhang Z, Fang Y (2014) Association of the 3′UTR FOXO3a polymorphism rs4946936 with an increased risk of childhood acute lymphoblastic leukemia in a Chinese population. Cell Physiol Biochem 34:325–332
Weatherall DJ, Clegg JB (2002) Genetic variability in response to infection: malaria and after. Genes Immun 3:331–337
Weigel D, Jackle H (1990) The fork head domain: a novel DNA binding motif of eukaryotic transcription factors? Cell 63:455–456
WHO (2010) Guidelines for the treatment of malaria
WHO (2013) World malaria report: 2013
Zintzaras E (2010) Impact of Hardy-Weinberg equilibrium deviation on allele-based risk effect of genetic association studies and meta-analysis. Eur J Epidemiol 25:553–560
Acknowledgments
We would like to thank the staff and technicians of Lambaréné, Gabon, for their assistance in collecting the blood samples of patients during the study. We also express our gratitude to all the study participants involved in this study.
Conflicts of interest
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nguetse, C.N., Kremsner, P.G. & Velavan, T.P. FOXO3A regulatory polymorphism and susceptibility to severe malaria in Gabonese children. Immunogenetics 67, 67–71 (2015). https://doi.org/10.1007/s00251-014-0816-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-014-0816-z