Abstract
Proton magnetic resonance spectroscopy (1H MRS) is beneficial in the lateralization of the epileptogenic zone in temporal lobe epilepsy; however, its role in extratemporal and, especially, MRI-negative epilepsy has not been established. This study seeks to verify how 1H MRS could help in localizing the epileptogenic zone in patients with MRI-negative extratemporal epilepsy. Seven patients (8–23 years) with MRI-negative refractory focal epilepsy were studied using 1H MRS on a 1.5T MR system. Chemical shift imaging sequence in the transversal plane was directed towards the suspected epileptogenic zone localized by seizure semiology, scalp video/EEG, ictal SPECT and 18FDG-PET. Spectra were evaluated using the program CULICH, and the coefficient of asymmetry was used for quantitative lateralization. MRS detected lateralization in all patients and was able to localize pathology in five. The most frequent findings were decreased ratios of N-acetylaspartate to choline compounds characterized by increasing choline concentration. The localization of the 1H MRS abnormality correlated well with ictal SPECT and subdural mapping. In all cases, histopathological analysis revealed MRI-undetected focal cortical dysplasias. 1H MRS could be more sensitive for the detection of discrete malformations of cortical development than conventional MRI. It is valuable in the presurgical evaluation of patients without MRI-apparent lesions.
Similar content being viewed by others
Introduction
Proton magnetic resonance spectroscopy (1H MRS) is a diagnostic method that provides a noninvasive window into brain biochemistry [1–4]. In epileptology, 1H MRS has proved to have a significant value in the lateralization of the epileptogenic zone in mesiotemporal epilepsy [5–10]. Two MRS techniques are generally used.
Single voxel spectroscopy acquiring a single spectrum has been predominantly targeted at the hippocampal regions [8], and the comparison of metabolite concentrations in ipsi- and contralateral hippocampus by using the coefficient of asymmetry, Ca, was developed [6]. Chemical shift imaging (CSI), also called MR spectroscopic imaging (MRSI) collects spectroscopic data from a large volume of interest in a single measurement and enables the creation of two- or three-dimensional maps of metabolites [5, 10, 11]. The advent of 1H CSI has also enabled its use in the localization of an epileptogenic zone outside of the temporal lobes.
In extratemporal epilepsies, the epileptogenic zone is frequently larger and more difficult to delineate in comparison with temporal lobe epilepsy. The most challenging patients are those with normal MRI findings, in whom only 1H MRS holds a promise for finding the metabolic changes corresponding to the epileptogenic zone. However, since the VOI positioning is rather difficult, studies focused on the use of 1H MRS in extratemporal epilepsy with negative MRI findings have been scarce.
Few reports have referred to 1H MRS abnormalities in patients with frontal or parietal lobe epilepsy, with or without MRI detected lesions [12–15]. These studies suggest that 1H MRS may help in the lateralization of extratemporal epilepsy; however, it is not clear whether it is useful in the localization of the epileptogenic zone. Moreover, there is an apparent absence of studies evaluating the relationship of 1H MRS abnormalities with the localization of the ictal onset zone as identified by monitoring from intracranial electrodes and correlating them with histopathological changes in resected brain tissue.
In the current study, metabolic spectroscopic images of patients with MRI-negative extratemporal epilepsy were evaluated by visual inspection to find the metabolic lesion corresponding to the epileptogenic region. We used Ca for the statistical evaluation of lateralization. The correlation with results of other imaging techniques and invasive monitoring as well as with histological analysis of the resected tissue was done.
Methods
Patients and presurgical diagnostic protocol
All patients were studied at the Motol Epilepsy Center (Prague) from 2002 to 2004. The inclusion criteria for the patients were the diagnosis of refractory extratemporal epilepsy (based on seizure semiology and EEG findings) and at least two negative MRI examinations. We retrospectively identified a cohort of 25 patients who had had extratemporal surgery from a total of 88 patients undergoing resective epilepsy surgery from 2002 to 2004 at our center. Seven (three females and four males aged from 8 to 23 years, mean age 11.7 years) of these 25 patients who had normal preoperative MRI examinations entered the study and were examined according to the diagnostic presurgical protocol for patients with refractory MRI-negative focal epilepsy. The protocol included video-EEG monitoring, 1H MRS, ictal 99mTc-ECD SPECT with subtraction, interictal 18FDG-PET, etc.
Clinical data and results of individual diagnostic tests in patients are depicted in Table 1. All patients but two [3, 7] had normal neurological examinations. Patient 3 had mild right-sided hemiparesis; patient 7 exhibited transient paresis of the right upper limb depending on the actual seizure frequency. All patients underwent thorough neuropsychological testing that showed different cognitive impairments in all the patients. Patient 3 had mild mental retardation; the others had normal intelligence. Severe attention disorder, depending on the actual seizure frequency, predominated.
Six patients (1, 2, 3, 4, 5, 6) had simple partial seizures with asymmetric tonic posturing typical for the activation of the supplementary sensorimotor area (SSMA), with or without secondary generalization. Patient 7 had several seizure types indicating the spread of seizure activity to the left parietal, frontal and temporal lobes. Interictal and ictal scalp EEG together enabled lateralization in six patients (1, 2, 3, 5, 6, 7).
All patients except one (4) also had a localized hyperperfusion zone on ictal 99mTc-ECD SPECT. Four patients had normal 18FDG-PET findings. Patients 3 and 7 had severe glucose hypometabolism of the whole left hemisphere with an occipital and frontal preponderance, respectively. Patient 6 had left frontal focal hypometabolism.
MRI study
Routine MRI examinations were performed using a 1.5-T MR imager (Gyroscan Intera, Philips) with a standard head coil. The initial MRI protocol included 5-mm-thick slices in all three directions [T2-weighted (T2w) turbo spin echo (TSE), fluid attenuated inversion recovery (FLAIR) and inversion recovery (IR)]. Patients with intractable focal epilepsy and a normal initial MRI were indicated for specialized MRI study, which consisted of 5-mm-thick transversal FLAIR slices and volumetric acquisition of 2-mm-thick coronal T1w-IR-TSE enabling 3D reconstruction of images. Two experienced neuroradiologists, who were not made aware of the clinical, EEG and other imaging data, independently analyzed the diagnostic MR scans (Philips Easy Vision Release 4.2 workstation). The following MRI criteria of focal cortical dysplasia were evaluated and excluded in the patients: increased cortical thickness, blurring of the gray matter–white matter junction, abnormal gyration, and FLAIR/TSE/IR white and gray matter signal changes.
1H Spectroscopic imaging
The patients underwent spectroscopic examination using a 1.5-T Siemens Vision MR system. Six healthy volunteers (mean age 29 years; range 25–32 years) were examined for comparison with patients. CSI spectra in controls were obtained from both white and gray matter of the frontoparietal regions bilaterally.
Both controls and patients signed the informed consent approved by the local ethics committee.
CSI spectra were obtained in the transversal plane by using a volume pre-selected PRESS-CSI hybrid sequence: FOV 160 × 160 mm, 16 × 16 steps, TR/TE = 1,500/135 ms, one acquisition, slice thickness 20 mm, nominal voxel volume 2 ml. Quality of the spectra was characterized by half-width of NAA signal in individual voxels between 2 and 4 Hz. The position of the CSI slice was chosen individually in each patient according to seizure semiology and the results of FDG-PET, ictal SPECT and EEG, i.e. it covered a region hypothesized to be the epileptogenic zone.
The evaluation of CSI data was done using the program CULICH, graphical LCModel-CSI data interface [16, 17]. Zero filling to a 32 × 32 matrix and mild k-space filtering to reduce point-spread function effects were performed prior to the calculation of metabolic images of the concentrations of N-acetylaspartate (NAA), creatine/phosphocreatine complex (Cr) and choline compounds (Cho) in laboratory units (mM) and their ratios. Error maps characterizing the statistical significance of the calculated results were used to exclude parts of the spectra with an error greater than 15% for metabolite images and 30% for their ratios [17, 18].
The description of metabolic abnormalities was done in two steps. First, metabolic images were evaluated visually and the position of the lesion was proposed on the basis of minima in the metabolic maps. In the second step, Ca was calculated from metabolite signals in the suspected lesion and contralaterally. It is given by the equation:
where x is the metabolite under investigation (NAA, Cr or Cho) and R is the metabolite concentration ratio in the left (sin) and right (dx) hemispheres (symmetrical with respect to the central line). The influence of CSF was eliminated using ratios of metabolite concentrations; the choice of voxels in gray and white matter was checked using visual segmentation. The minimum number of CSI voxels used for statistical evaluation was five for the side of the suspected lesion, and the data were compared with the coefficients of asymmetry obtained from the control group by ANOVA analysis with contrast evaluation and by paired t-tests. Significant differences between control and patient data were based on the null hypothesis H0—group means of Ca are not different from the control group—which was rejected if P < 0.05.
Subdural electrode mapping, surgery and outcome
Because of negative MRI results and difficult localization of the epileptogenic zone, all the patients underwent the implantation of subdural electrodes prior to resection. The electrodes were implanted according to the results of the noninvasive tests and in all cases covered the area investigated by 1H MRS (Table 1). The extent of neocortical resection was delineated according to the results of the invasive monitoring and extraoperative cortical mapping.
Follow-up periods and seizure outcomes in individual patients are depicted in the second part of Table 1.
Histopathology
The resected brain tissue was orientated according to a drawing made during surgery, and tissue blocks for histological processing were taken topographically. To assess the degree of gliosis and the altered antigenic properties of cells in cortical dysplasia, immunoperoxidase staining was performed using antibodies against glial fibrillary acidic protein and neurofilaments from selected tissue blocks. The antigen-antibody complexes were visualized by biotin-streptavidin detection systems with 3,3′-diaminobenzidine as the chromogen.
In all cases, histological examination revealed a focal cortical dysplasia (FCD). According to the new classification of FCD [19], six patients were evaluated as Taylor-type FCD (type II). The diagnosis of FCD type IIb was established after extensive examination of the whole resected tissue in the respective cases. Balloon cells were frequently present in a limited number of foci only. The changes seen in the resected cortex of the last patient (5) were classified as FCD type Ib due to the presence of both immature and giant neurons in the dyslaminated cortex.
Comparison of the results
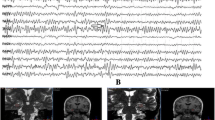
The localization of 1H MRS, SPECT and PET abnormalities was compared by coregistration. The position of subdural electrodes was visually compared with the location of 1H MRS pathology. The localization of the resection cavity on postoperative MRI was coregistered with preoperative 1H MRS metabolite maps (Fig. 1).
CSI results and T2w images of patients 2 (a–c), 5 (d–f) and 7 (g–i). (NAA N-acetylaspartate, Cho choline compounds, Cr creatine/phosphocreatine; all colored images are in the same scale for the ratio NAA/Cho). a Projection of the NAA/Cho CSI map before surgery. A large hypointensive area A (blue) was considered to be a metabolic anomaly, and Ca calculated from areas A and B confirmed this. b 1H MR spectra from symmetrical voxels in areas A and B (see arrows) show differences in the NAA/Cho ratios: A/B ∼ 4.6/7.6. c Hyperintensive lesion in T2w image after surgery corresponds to area A in CSI image. d Projection of the NAA/Cho CSI map before surgery. A hypointensive NAA/Cho area A (blue) with two minima was observed in the left frontal lobe; and Ca calculated from areas A and B confirmed this. In the CSI image, another metabolic anomaly in region C was also visible in the right hemisphere; however, these pixels did not influence the calculation of Ca. e 1H MR spectra from symmetrical voxels in areas A and B (see arrows) show differences in the NAA/Cho ratios: A/B ∼ 4.2/6.0. f T2w image obtained after resection. The location of surgery corresponds to one of the minima in the CSI image from area A. g Projection of the NAA/Cho minima CSI map before surgery. A much larger hypointensive area was observed in the left parietal lobe (A) compared to the right one (B). In area A, a hypointensive minimum was found in the gray matter (C) and the coefficient of asymmetry in Table 2 was calculated for voxels in the gray matter (C) and contralaterally (D). h 1H MR spectra from symmetrical voxels in areas C and D represent the differences in the relative concentrations of NAA/Cho: C/D ∼ 4.8/8.3. Also, the decrease in the total intensity of the signals in the spectrum from area C is clearly visible. i T2w image obtained 1 month after resection. The location of surgery corresponds to the minima in the CSI image (C)
Results
All patients had a visible asymmetry in spectroscopic images that corresponded to local changes in metabolite concentrations. To quantify these findings, the coefficients of asymmetry from the metabolic abnormality and symmetrical contralateral part of the brain were calculated using values obtained from LCModel calculation of spectroscopic images. The CSI results of controls and patients are summarized in Table 2.
In the controls, we did not find any significant difference in the Ca of the NAA/Cho and NAA/Cr ratios in either white or gray matter. This is in agreement with previous findings [20] in which comparison of the Ca in the white and gray matter of controls revealed only a difference in the Cho/Cr ratio.
The lateralization of a metabolic abnormality in patients was based on the calculation of the Ca and the comparison with Ca of controls. In patients 2, 5, 6 and 7, the lateralization and localization of the metabolic abnormalities corresponded well with the position of the resection (Fig. 1); the Ca also confirmed the lateralization (Table 2).
In patient 4, visual inspection showed two visible metabolite minima (in both frontal lobes), without any significant difference with respect to Ca of controls (Table 2). Detailed inspection of the MR images revealed an enlarged perivascular space colocalizing with the 1H MRS abnormality in the right hemisphere. Since this MRI abnormality was initially regarded as a possible epileptogenic lesion, subdural electrodes were first implanted over the right frontal region; invasive monitoring however failed to show ictal onsets there. Electrodes were therefore additionally implanted over the contralateral frontal lobe (MRI-negative area) and revealed an epileptogenic zone in the left hemisphere that was eventually proven by histopathology. Localization of the lesion agreed with the left hemisphere metabolite changes.
In patients 1 and 3, only lateralization based on overall changes in Ca in gray matter was described because no local minima in the examined CSI areas were visible. In these subjects, the resection was outside the regions examined by CSI. However, the lateralization resulting from CSI evaluation (Table 2) was confirmed by the results of the surgery, supporting the assumption that metabolite changes are usually more extensive.
In the patients with a localized 1H MRS abnormality, metabolite changes colocalized with results of ictal SPECT in all four cases with a localized hyperperfusion zone (2, 5, 6, 7). Comparison of metabolite maps with results of subdural electrode mapping showed that in both patients without a localized 1H MRS abnormality (1, 3), the ictal onset zone was found outside the position of CSI slides. In the remaining patients with a clearly localized metabolic abnormality (2, 4, 5, 6, 7), the ictal onset zone overlapped with 1H MRS changes. Correlation of 1H MRS abnormalities with the localization of the resection cavity on postoperative MRI showed an overlap of these regions in all the patients with localized 1H MRS pathology.
Discussion
Success in epilepsy surgery is directly related to the ability to precisely localize the region of seizure onset. When the MRI is normal, localization of the epileptogenic zone is very challenging and a less favorable surgical outcome is generally assumed. Some studies therefore seriously question even the consideration of epileptosurgical treatment when the MRI is negative. However, some recent studies have reported promising surgical outcomes in this group of patients [21–23]. Siegel reported on a series of 43 patients with normal MRI. All the patients underwent invasive monitoring with intracranial electrodes. Twenty-five (58%) of them were subsequently indicated for resective epilepsy surgery, and 20 of these (83%) had a satisfactory postoperative seizure outcome. The reason for these good results was considered to be both the proper use of intracranial electrodes and the correct interpretation of all noninvasive diagnostic tests, especially ictal SPECT [21].
A common strategy of clinical MR spectroscopic examination is based on a comparison of a diseased, or suspected to be diseased, region of interest with the contralateral, healthy part of the brain. In those cases where MR images show no structural abnormality, the coefficient of asymmetry of patients and controls can be used for the characterization of metabolic changes in the clinically suspected area.
It is known that, with the exception of the thalamus and Wernicke area, there are no significant differences in metabolite concentrations between hemispheres in the healthy brain [20]. The comparison of coefficients of asymmetry is thus a suitable procedure for evaluating 1H MRS examinations in patients with normal MRI findings.
Some previous studies suggested a role for 1H MRS in MRI-negative temporal lobe epilepsy patients [5, 6, 8]. Only a few studies focused on extratemporal epilepsies. In the largest series of 20 patients with extratemporal focal epilepsy without apparent MRI lesions, Stanley et al. [13] reported decreased NAA/Cr, NAA/Cho and NAA/(Cho+Cr) ratios as compared with controls. Other ratios (Cho/Cr, Lac/Cr) did not significantly differ. The most prominent abnormalities were found in the probable epileptogenic zone; however, only 42% of the patients had such a localized abnormality. Similarly to other studies [12, 14], a predominance of widespread alterations in metabolite concentrations was seen. Minor abnormalities were also found contralateral to the epileptogenic focus in some patients. Guye et al. [15] described in their group of 12 patients four cases with negative MRI findings. Histology confirmed Taylor-type FCD lesion, and MRS revealed decreased values of NAA/Cr and NAA/(Cr+Cho). 1H MRS CSI protocol was demonstrated to be a useful method for the FCD description. To our knowledge, no study has correlated 1H MRS findings with the localization of the seizure onset zone from intracranial electrodes and subsequent histopathological examination.
It appears that the cause of epilepsy in many MRI-negative patients is FCD. Subtle FCD cannot be discerned on MRI [24, 25]. Several reports referred to the sensitivity of 1H MRS for different types of malformations [26–28]. The comparison of MRI and 1H MRS findings showed that metabolite alterations could also be detected outside an MRI lesion in patients with FCD [29, 30]. This could reflect the well-known fact that FCD are usually widespread and extend beyond the visible lesion [31–33]. These findings represent a theoretical background for the application of 1H MRS in patients with refractory epilepsy and no structural abnormality on MRI.
We report on seven patients with refractory extratemporal epilepsy and normal MRI. In five patients, there was a localized 1H MRS abnormality; in the remaining two patients, the 1H MRS findings had value for lateralization. With the exception of one patient (4), the 1H MRS abnormalities supported the hypothesis about the epileptogenic zone localization based on seizure semiology and other noninvasive examinations.
We found an especially good agreement between the 1H MRS findings and the localization of the hyperperfusion zone in ictal SPECT (in all patients with both a localized 1H MRS abnormality and hyperperfusion zone). In two patients (1, 3) with a discrepancy between semiological and SPECT localization, 1H MRS supported the ictal SPECT findings, which were finally confirmed by subdural electrode mapping.
In agreement with previous studies [34, 35], 18FDG-PET was not able to lateralize or localize the epileptogenic zone in all patients. In our study, 18FDG-PET was effective in only in three patients; the remaining four subjects had normal glucose metabolism. Localized hypometabolism correlating with a 1H MRS abnormality was encountered in only one patient. The remaining two patients had widespread left hemisphere hypometabolism; in one of them, the maximum 1H MRS abnormality colocalized with the region of the most prominent hypometabolism.
Subdural electrode mapping showed a good agreement between the maximum 1H MRS abnormality and seizure onsets in five subjects (two patients with no clear metabolite abnormality maximum had an ictal onset zone outside the regions examined by 1H MRS). With the exception of one patient (5) whose seizures originated from the eloquent cortex, at least part of the ictal onset zone was resected and processed for histopathological analysis. Based on the precise correlation of postoperative MRI defects with 1H MRS maps, we can be certain that the histologically examined tissue colocalized with the regions of the most prominent 1H MRS changes.
The 1H MRS findings are characterized by decreasing NAA/Cho and NAA/Cr ratios in the ipsilateral part of the brain (or in the “lesion”). The most frequent abnormalities were characterized by significant differences in the Ca of the NAA/Cho (in five of seven patients) and NAA/Cr (in three of seven patients) ratios. Two patients had significant differences in the Cho/Cr ratio compared to controls. Observed changes can reflect an increase in Cho and Cr concentrations and/or a decrease in NAA concentrations. To distinguish these two possibilities, absolute quantification of Cho, Cr and NAA signals has to be performed. Several aspects make absolute quantification of CSI data difficult. Apart from the existence of point-spread function [11], inhomogeneous B1 field in the examined tissue may influence the accuracy of calculated concentrations. This becomes an important issue when signals from different locations are compared. The B1 homogeneity of the used head coil had been tested on a water phantom. The greatest difference between left and corresponding right voxels was below 5%. Figure 2 shows the graphical presentation of the signal distribution and its asymmetry in the excited area. It should be noted that the B1 inhomogeneity has no effect on the metabolite ratios and corresponding Ca as long as the relative displacements of the slice profiles of individual metabolites due to chemical shift artifact can be neglected.
Influence of the coil B1 inhomogeneity. a Distribution of the water signal measured by CSI sequence in the water phantom using a transmit/receive head coil. Asymmetry in the signal distribution can be recognized. b The dependence of the relative signal difference SR = 2 × (S sin - S dx) / (S sin + S dx) between the signal from the left (S dx) and corresponding right (S dx) voxel on the distance from the coil center. The dependence for the profile depicted in Fig. 2a is shown by the dashed line
In our patient group, increasing choline intensity in the ipsilateral part of the brain was observed in six cases. When taking the correction mentioned above into consideration, the mean value of choline concentration increase was approximately 19%. In the remaining patient (7), we observed all compounds decreased compared to the contralateral part (Fig. 1h). A decrease in Cr and NAA (greater than 5%) was observed in three cases (patients 3, 4, 5). Our findings are in accordance with previous studies that observed increased Cho concentration [14, 15] in cortical dysplasia and decreased relative NAA concentration [26, 27].
A decreasing ratio of NAA/Cr and NAA/Cho (or NAA/(Cho+Cr) ratio) was also described in many cases of mesiotemporal sclerosis [5, 6]. Besides the decrease in the NAA concentration due to the loss or damage of neurons, another theory supposes a change in mitochondrial metabolism [36] that leads to increasing creatine and decreasing NAA concentrations in the epileptogenic lesion. The most significant change we observed was a decrease in the NAA/Cho ratio, which corresponded to an increase in the choline compound content. Major contributors to the choline signal at a magnetic field of 1.5 T are phosphocholine (PCho) and glycerophosphocholine (GPCho). The other contributions to signal intensity at 3.2 ppm are from choline, acetylcholine, cytidine diphosphate choline and betaine [37]. Metabolic pathways describing possibilities for the increasing concentration of choline compounds in the brain show that it is phospholipase A2 (PLA2) that controls the formation of GPCho and PCho from phosphatydylcholine. PLA2 was observed in increased concentrations in an experimental epilepsy model, and this lipase is responsible for turnover of membrane fatty acids [38]. PLA2 in the epilepsy model decreases the concentration of fatty acid precursors and increases the concentration of small MR-visible choline metabolites in the brain. On the other hand, because the group of choline metabolites contributing to the MR signal at 3.2 is large, different metabolic pathways may be expected that can be regionally dependent [37, 38]. Changes in relaxation times of the metabolite in diseased tissue should also be considered, however, sufficient data are still missing.
Histopathological analysis showed FCD in all patients. Severe type II FCD was predominantly encountered. Only in one patient was a type Ib dysplasia found, and in this patient the greatest changes in the Ca were also observed. On the other hand, we could not clearly distinguish between different types of FCD based only on the 1H MRS results in our patients. This is in agreement with a study that analyzed 1H MRS findings in patients with malformations caused by abnormalities of cortical development [30]. While the spectra from lesions usually showed a decreased NAA concentration, abnormalities in the perilesional white matter and contralateral regions were diverse.
The large number of type II and especially IIb FCD findings in our MRI-negative patients is rather surprising, since MRI is usually reported to be highly sensitive for this type of pathology [39]. The reason for the normal MRI findings in our patients could be the relatively small number of balloon cells found. It should be stressed that the proportion of MRI-negative cases among our FCD patients is similar to that reported in previous studies on FCD patients [39, 40].
In conclusion, the present study found promising results concerning the sensitivity of 1H MRS in patients with MRI-undetectable malformations of cortical development. 1H MRS helped not only to lateralize, but also to localize the epileptogenic zone. The most important message of the study is the good correlation among 1H MRS, ictal SPECT and subdural mapping, which was subsequently confirmed by histopathological analysis of the resected tissue. We suggest that 1H MRS may provide important additional data in the presurgical evaluation of patients without apparent MRI lesions.
References
Garcia PA, Laxer KD, Ng T (1995) Application of spectroscopic imaging in epilepsy. Magn Reson Imaging 13:1181–1185
Zimmerman RA, Wang Z (1999) Proton magnetic resonance spectroscopy. Crit Rev Neurosurg 9:161–166
Bonavita S, Di Salle F, Tedeschi G (1999) Proton MRS in neurological disorders. Eur J Radiol 30:125–131
Ranjeva JP, Confort-Gouny S, Le Fur Y, Cozzone PJ (2000) Magnetic resonance spectroscopy of brain in epilepsy. Childs Nerv Syst 16:235–241
Cendes F, Caramanos Z, Andermann F, Dubeau F, Arnold DL (1997) Proton magnetic resonance spectroscopic imaging and magnetic resonance imaging volumetry in the lateralization of temporal lobe epilepsy: a series of 100 patients. Ann Neurol 42:737–746
Hajek M, Dezortova M, Komarek V (1998) 1H MR spectroscopy in patients with mesial temporal epilepsy. Magn Reson Mater Phy 7:95–114
Hammen T, Stefan H, Eberhardt KE, W-Huk BH, Tomandl BF (2003) Clinical applications of 1H-MR spectroscopy in the evaluation of epilepsies-what do pathological spectra stand for with regard to current results and what answers do they give to common clinical questions concerning the treatment of epilepsies? Acta Neurol Scand 108:223–238
Hajek M, Komarek V, Dezortova M, Hlavnicka P, Smejkalova M, Faladova L, Hovorka J (1995) Determination of epileptogenic focus using 1H MR spectroscopy. (Cz) Ces Slov Neurol Neurochir 3:103–107
Hetherington HP, Pan JW, Spencer DD (2000) 1H and 31P spectroscopy and bioenergetics in the lateralization of seizures in temporal lobe epilepsy. J Magn Reson Imaging 16:477–483
Bernasconi A, Tasch E, Cendes F, Li LM, Arnold DL (2002) Proton magnetic resonance spectroscopic imaging suggests progressive neuronal damage in human temporal lobe epilepsy. Prog Brain Res 135:297–304
Klose U, Jiru F (2005) Principles of MR spectroscopy and chemical shift imaging. In: Landini L (ed) Advanced image processing in magnetic resonance imaging (signal processing and communications). CRC, Boca Raton, pp 369–409
Garcia PA, Laxer KD, van der Grond J, Hugg JW, Matson GB, Weiner MW (1995) Proton magnetic resonance spectroscopic imaging in patients with frontal lobe epilepsy. Ann Neurol 37:279–281
Stanley JA, Cendes F, Dubeau F, Andermann F, Arnold DL (1998) Proton magnetic resonance spectroscopic imaging in patients with extratemporal epilepsy. Epilepsia 39:267–273
Lundbom N, Gaily E, Vuori K, Paetau R, Liukkonen E, Rajapakse JC, Valanne L, Hakkinen AM, Granstrom ML (2001) Proton spectroscopic imaging shows abnormalities in glial and neuronal cell pools in frontal lobe epilepsy. Epilepsia 42:1507–1514
Guye M, Ranjeva JP, Le Fur Y, Bartolomei F, Confort-Gouny S, Regis J, Chauvel P, Cozzone PJ (2005) 1H-MRS imaging in intractable epilepsies characterized by depth electrode recording. Neuroimage 26:1174–1183
Jiru F, Skoch A, Hajek M (2003) LCModel for quantitative single voxel spectroscopy and spectroscopic imaging: a comparison. Magn Reson Mater Phy 16 (Suppl 1):S211. Abstract
Jiru F, Skoch A, Klose U, Grodd W, Hajek M (2006) Error images for spectroscopic imaging by LCModel using Cramer-Rao bounds. Magn Reson Mater Phy 19:1–14
Jiru F, Dezortova M, Burian M, Hajek M (2003) The role of relaxation time corrections for the evaluation of long and short echo time 1H MR spectra of the hippocampus by NUMARIS and LCModel techniques. Magn Reson Mater Phy 16:135–143
Palmini A, Najm I, Avanzini G, Babb T, Guerrini R, Foldvary-Schaefer N et al (2004) Terminology and classification of the cortical dysplasias. Neurology 62 (Suppl 3):S2–S8
Nagae-Poetscher LM, Bonekamp D, Barker PB, Brant LJ, Kaufmann WE, Horska A (2004) Asymmetry and gender effect in functionally lateralized cortical regions: a proton MRS imaging study. J Magn Reson Imaging 19:27–33
Siegel AM, Jobst BC, Thadani VM, Rhodes CH, Lewis PJ, Roberts DW, Williamson PD (2000) Medically intractable, localization-related epilepsy with normal MRI: presurgical evaluation and surgical outcome in 43 patients. Epilepsia 42:883–888
Chapman K, Wyllie E, Najm I, Ruggieri P, Bingaman W, Lüders J, Kotagal P, Lachhwani D, Dinner D, Lüders HO (2005) Seizure outcome after epilepsy surgery in patients with normal preoperative MRI. J Neurol Neurosurg Psychiatry 76:710–713
Blume WT, Ganapathy GR, Munoz D, Lee DH (2004) Indices of resective surgery effectiveness for intractable nonlesional focal epilepsy. Epilepsia 45:46–53
Tassi L, Colombo N, Garbelli R, Francione S, Lo Russo G, Mai R et al (2002) Focal cortical dysplasia: neuropathological subtypes, EEG, neuroimaging and surgical outcome. Brain 125:1719–1732
Bernasconi A (2003) Advanced MRI analysis methods for detection of focal cortical dysplasia. Epileptic Disord 5:81–84
Kuzniecky R, Hetherington H, Pan J, Hugg J, Palmer C, Gilliam F et al (1997) Proton spectroscopic imaging at 4.1 Tesla in patients with malformations of cortical development and epilepsy. Neurology 48:1018–1024
Li LM, Cendes F, Bastos AC, Andermann F, Dubeau F, Arnold DL (1998) Neuronal metabolic dysfunction in patients with cortical developmental malformations: a proton magnetic resonance spectroscopic imaging study. Neurology 50:755–759
Kaminaga T, Kobayashi M, Abe T (2001) Proton magnetic resonance spectroscopy in disturbances of cortical development. Neuroradiology 43:575–580
Simone IL, Federico F, Tortorella C, De Blasi R, Bellomo R, Lucivero V et al (1999) Metabolic changes in neuronal migration disorders: evaluation by combined MRI and proton MR spectroscopy. Epilepsia 40:872–879
Woermann FG, McLean MA, Bartlett PA, Barker GJ, Duncan JS (2001) Quantitative short echo time proton magnetic resonance spectroscopic imaging study of malformations of cortical development causing epilepsy. Brain 124:427–436
Palmini A, Andermann F, Olivier A, Tampieri D, Robitaille Y, Andermann E, Wright G (1991) Focal neuronal migration disorders and intractable partial epilepsy: a study of 30 patients. Ann Neurol 30:741–749
Sisodiya SM (2004) Surgery for focal cortical dysplasia. Brain 127:2383–2384
Urbach H (2005) Imaging of the epilepsies. Eur Radiol 15(3):494–500
Duncan JS (2002) Neuroimaging methods to evaluate the etiology and consequences of epilepsy. Epilepsy Res 50:131–140
Henry TR, Van Heertum RL (2003) Positron emission tomography and single photon emission computed tomography in epilepsy care. Semin Nucl Med 33:88–104
Petroff OAC, Errante LD, Kim JH (2003) N-acetyl-aspartate, total creatine, and myo-inositol in the epileptogenic human hippocampus. Neurology 60:1646–1651
Boulanger Y, Labell M, Khiat A (2000) Role of phospholipase A2 on the variation of the choline signal intensity observed by 1H magnetic resonance spectroscopy in brain diseases. Brain Res Rev 33:380–389
Yegin A, Akbas SH, Ozben T, Korgun DK (2002) Secretory phospholipase A2 and phospholipids in neural membranes in an experimental epilepsy model. Acta Neurol Scand 106:258–262
Lawson JA, Birchansky S, Pacheco E, Jayakar P, Resnick TJ, Dean P, Duchowny MS (2005) Distinct clinicopathologic subtypes of cortical dysplasia of Taylor. Neurology 64:55–61
Colombo N, Citterio A, Galli C, Tassi L, Lo Russo G, Scialfa G, Spreafico R (2003) Neuroimaging of focal cortical dysplasia: neuropathological correlations. Epileptic Disord 5 (Suppl 2):S67–72
Acknowledgements
Supported by grants from the Grant Agency of Czech Republic, no. 309/02/D076, Internal Grant Agency of Ministry of Health, Czech Republic, no. NF/7411-3 and NR/8843-4, Ministry of Education, Youth and Sport, Czech Republic LC554, and research projects no. 00000064203-4, 111300003, 111300004 and MZO 00023001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Pavel Krsek and Milan Hajek contributed equally to this work.
Rights and permissions
About this article
Cite this article
Krsek, P., Hajek, M., Dezortova, M. et al. 1H MR spectroscopic imaging in patients with MRI-negative extratemporal epilepsy: correlation with ictal onset zone and histopathology. Eur Radiol 17, 2126–2135 (2007). https://doi.org/10.1007/s00330-007-0594-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-007-0594-1