Abstract
Objective
To prove that 1.0 M gadobutrol provides superior contrast enhancement in suspicion of osteomyelitis of the feet compared with 0.5 M gadoterate.
Methods
MRI of feet was performed on 2 separate occasions. Independent injections of 1.0 M gadobutrol and 0.5 M gadoterate at doses of 0.1 mmol Gd/kg body weight were administered per patient. The interval between the two MR examinations was between 24 h and 7 days. Evaluation was performed in an off-site blinded read.
Results
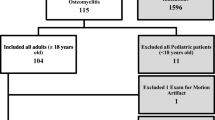
41 patients were eligible for efficacy analysis. Results of secondary efficacy variables did not show statistically significant differences. For the primary efficacy variable, a trend in favour of gadobutrol was seen in the full analysis set (ITT) population resulting in at least non-inferiority. In the per protocol (PP) analysis set gadobutrol had better contrast than gadoterate (Wilcoxon signed rank test, p = 0.0466).
Conclusion
Imaging of the distal lower limb in this special patient population requires a large number of patients to obtain enough comparative images where non-contrast-agent-dependent factors do not disturb contrast agent efficacy. The ITT analysis showed at least non-inferiority of gadobutrol in comparison to gadoterate. The avoidance of imaging artefacts demonstrates a better outcome for gadobutrol.



Similar content being viewed by others
References
Grayson ML, Gibbons GW, Balogh K, Levin E, Karchmer AW (1995) Probing to bone in infected pedal ulcers. A clinical sign of underlying osteomyelitis in diabetic patients. JAMA 273:721–723
Meyers SP, Wiener SN (1991) Diagnosis of hematogenous pyogenic vertebral osteomyelitis by magnetic resonance imaging. Arch Intern Med 151:683–687
Schmid MR, Hodler J, Vienne P, Binkert CA, Zanetti M (2002) Bone marrow abnormalities of foot and ankle: STIR versus T1-weighted contrast-enhanced fat-suppressed spin-echo MR imaging. Radiology 224:463–469
Maley JE, Yuh WT (1998) Contrast dosage in the neuroimaging of brain tumors. Principles and indications. Magn Reson Imaging Clin N Am 6:113–124
Morcos SK (2008) Extracellular gadolinium contrast agents: differences in stability. Eur J Radiol 66:175–179
Morcos SK, Thomsen HS (2008) Nephrogenic systemic fibrosis: more questions and some answers. Nephron Clin Pract 110:c24–31, discussion c32
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann HJ (2005) Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Invest Radiol 40:715–724
Huppertz A, Rohrer M (2004) Gadobutrol, a highly concentrated MR-imaging contrast agent: its physicochemical characteristics and the basis for its use in contrast-enhanced MR angiography and perfusion imaging. Eur Radiol 14(Suppl 5):M12–18
Lemke AJ, Sander B, Balzer T, Geens V, Hosten N, Felix R (1997) Safety and use of gadobutrol in patients with brain tumors (phase III trial). Rofo 167:591–598
Mohrs OK, Oberholzer K, Krummenauer F, Bernhard S, Kalden P, Neufang A, Thelen M, Kreitner KF (2004) Comparison of contrast-enhanced MR angiography of the aortoiliac vessels using a 1.0 molar contrast agent at 1.0T with intra-arterial digital subtraction angiography. Rofo 176:985–991
Schaefer FK, Schaefer PJ, Altjohann C, Bourne M, Decobelli F, Goyen M, Griffiths PD, Kopka L, Kreitner KF, Link J, Oberholzer K, Pering C, Poeckler-Schoeninger C, Ruehm SG, Sachoran MR, Schulte-Altedorneburg G, Springer OS, Steiner P, Wall A, Winterer J, Tombach B (2007) A multicenter, site-independent, blinded study to compare the diagnostic accuracy of contrast-enhanced magnetic resonance angiography using 1.0 M gadobutrol (Gadovist) to intraarterial digital subtraction angiography in body arteries. Eur J Radiol 61:315–323
Tombach B (2004) Whole-body CE-MRA with Gadovist. Eur Radiol 14(Suppl 5):M26–27
Tombach B, Bremer C, Reimer P, Matzkies F, Schaefer RM, Ebert W, Geens V, Eisele J, Heindel W (2002) Using highly concentrated gadobutrol as an MR contrast agent in patients also requiring hemodialysis: safety and dialysability. AJR Am J Roentgenol 178:105–109
Tombach B, Heindel W (2002) Value of 1.0- M gadolinium chelates: review of preclinical and clinical data on gadobutrol. Eur Radiol 12:1550–1556
Vogler H, Platzek J, Schuhmann-Giampieri G, Frenzel T, Weinmann HJ, Raduchel B, Press WR (1995) Pre-clinical evaluation of gadobutrol: a new, neutral, extracellular contrast agent for magnetic resonance imaging. Eur J Radiol 21:1–10
Goyen M, Lauenstein TC, Herborn CU, Debatin JF, Bosk S, Ruehm SG (2001) 0.5 M Gd chelate (Magnevist) versus 1.0 M Gd chelate (Gadovist): dose-independent effect on image quality of pelvic three-dimensional MR-angiography. J Magn Reson Imaging 14:602–607
Vosshenrich R, Engeroff B, Obenauer S, Grabbe E (2003) Contrast-enhanced MR angiography of the arterial and portovenous system of the liver with varying concentrations of contrast medium. Rofo 175:1239–1243
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Bellin MF, Van Der Molen AJ (2008) Extracellular gadolinium-based contrast media: an overview. Eur J Radiol 66:160–167
Hadizadeh DR, Von Falkenhausen M, Kukuk GM, Schoneseiffen K, Gieseke J, Schild HH, Willinek WA Contrast material for abdominal dynamic contrast-enhanced 3D MR angiography with parallel imaging: intraindividual equimolar comparison of a macrocyclic 1.0 M gadolinium chelate and a linear ionic 0.5 M gadolinium chelate. AJR Am J Roentgenol 194:821–829
Hammerstingl R, Adam G, Ayuso JR, Van Beers B, Belfiore G, Bellin MF, Bongartz G, Ernst O, Frericks B, Giuseppetti G, Heinz-Peer G, Laghi A, Martin J, Pering C, Reimer P, Richter GM, Roemer FW, Schafer FK, Vilgrain V, Vogl TJ, Weishaupt D, Wall A, Zech C, Tombach B (2009) Comparison of 1.0 M Gadobutrol and 0.5 M Gadopentetate Dimeglumine-Enhanced Magnetic Resonance Imaging in Five Hundred Seventy-Two Patients With Known or Suspected Liver Lesions: Results of a Multicenter, Double-Blind, Interindividual, Randomized Clinical Phase-III Trial. Invest Radiol
Tombach B, Bohndorf K, Brodtrager W, Claussen CD, Duber C, Galanski M, Grabbe E, Gortenuti G, Kuhn M, Gross-Fengels W, Hammerstingl R, Happel B, Heinz-Peer G, Jung G, Kittner T, Lagalla R, Lengsfeld P, Loose R, Oyen RH, Pavlica P, Pering C, Pozzi-Mucelli R, Persigehl T, Reimer P, Renken NS, Richter GM, Rummeny EJ, Schafer F, Szczerbo-Trojanowska M, Urbanik A, Vogl TJ, Hajek P (2008) Comparison of 1.0 M gadobutrol and 0.5 M gadopentate dimeglumine-enhanced MRI in 471 patients with known or suspected renal lesions: results of a multicenter, single-blind, interindividual, randomized clinical phase III trial. Eur Radiol 18:2610–2619
Idee JM, Port M, Raynal I, Schaefer M, Le Greneur S, Corot C (2006) Clinical and biological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundam Clin Pharmacol 20:563–576
Frenzel T, Lengsfeld P, Schirmer H, Hutter J, Weinmann HJ (2008) Stability of gadolinium-based magnetic resonance imaging contrast agents in human serum at 37 degrees C. Invest Radiol 43:817–828
Anzalone N, Gerevini S, Scotti R, Vezzulli P, Picozzi P (2009) Detection of cerebral metastases on magnetic resonance imaging: intraindividual comparison of gadobutrol with gadopentetate dimeglumine. Acta Radiol 50:933–940
Attenberger UI, Runge VM, Jackson CB, Baumann S, Birkemeier K, Michaely HJ, Schoenberg SO, Reiser MF, Wintersperger BJ (2009) Comparative evaluation of lesion enhancement using 1 M gadobutrol vs. 2 conventional gadolinium chelates, all at a dose of 0.1 mmol/kg, in a rat brain tumor model at 3 T. Invest Radiol 44:251–256
Hentsch A, Aschauer MA, Balzer JO, Brossmann J, Busch HP, Davis K, Douek P, Ebner F, van Engelshoven JM, Gregor M, Kersting C, Knusel PR, Leen E, Leiner T, Loewe C, McPherson S, Reimer P, Schafer FK, Taupitz M, Thurnher SA, Tombach B, Wegener R, Weishaupt D, Meaney JF (2003) Gadobutrol-enhanced moving-table magnetic resonance angiography in patients with peripheral vascular disease: a prospective, multi-centre blinded comparison with digital subtraction angiography. Eur Radiol 13:2103–2114
Huppertz A, Balzer T, Blakeborough A, Breuer J, Giovagnoni A, Heinz-Peer G, Laniado M, Manfredi RM, Mathieu DG, Mueller D, Reimer P, Robinson PJ, Strotzer M, Taupitz M, Vogl TJ (2004) Improved detection of focal liver lesions at MR imaging: multicenter comparison of gadoxetic acid-enhanced MR images with intraoperative findings. Radiology 230:266–275
Reimer P, Schneider G, Schima W (2004) Hepatobiliary contrast agents for contrast-enhanced MRI of the liver: properties, clinical development and applications. Eur Radiol 14:559–578
Acknowledgements
Many thanks to all colleagues and the clinical team.
Special thanks to Dr. med. Thomas Wels – Medical Consulting & Key Account Management, for his continuous intellectual contributions and support in study administration.
This study was supported by Bayer Schering Pharma AG, Berlin. The authors are solely responsible for the content of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pennekamp, W., Roggenland, D., Hering, S. et al. Intra-individual, randomised comparison of the MRI contrast agents gadobutrol and gadoterate in imaging the distal lower limb of patients with known or suspected osteomyelitis, evaluated in an off-site blinded read. Eur Radiol 21, 1058–1067 (2011). https://doi.org/10.1007/s00330-010-2008-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-010-2008-z