Abstract
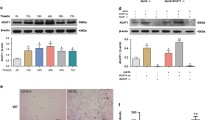
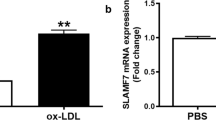
The formation of fat-laden foam cells, contributing to the fatty streaks of the plaques of atheroma, is the critical early process in atherosclerosis. The previous study demonstrated that vascular smooth muscle cells (VSMCs) contain a much larger burden of the excess cholesterol in comparison with monocyte-derived macrophages in human coronary atherosclerosis, as the main origin of foam cells. It is noteworthy that VSMC-derived foam cells are deposited in subintima but not media, where VSMCs normally deposit in. Therefore, migration from media to intima is an indispensable step for a VSMC to accrue neutral lipids and form foam cell. Whether this migration occurs paralleled with or prior to the formation of foam cell is still unclear. Herein, the present study was designed to test the VSMC migratory capability in the process of foam cell formation induced by oxidized low-density lipoprotein (oxLDL). In conclusion, we provide evidence that oxLDL induces the VSMC-derived foam cells formation with increased migration ability and MMP-9 expression, which were partly attributed to the impaired SIRT1 and enhanced nuclear factor-kappa B (NF-κB) activity. As activation of transient receptor potential vanilloid type 1 (TRPV1) has been reported to have anti-atherosclerotic effects, we investigated its role in oxLDL-treated VSMC migration. It is found that activating TRPV1 by capsaicin inhibits VSMC foam cell formation and the accompanied migration through rescuing the SIRT1 and suppressing NF-κB signaling. The present study provides evidence that SIRT1 may be a promising intervention target of atherosclerosis, and raises the prospect of TRPV1 in prevention and treatment of atherosclerosis.





Similar content being viewed by others
References
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389(6653):816–824. doi:10.1038/39807
Chaudhary N, Pfluger PT (2009) Metabolic benefits from Sirt1 and Sirt1 activators. Curr Opin Clin Nutr Metab Care 12(4):431–437. doi:10.1097/MCO.0b013e32832cdaae
Chen YX, Zhang M, Cai Y, Zhao Q, Dai W (2015) The Sirt1 activator SRT1720 attenuates angiotensin II-induced atherosclerosis in apoE(−/−) mice through inhibiting vascular inflammatory response. Biochem Biophys Res Commun 465(4):732–738. doi:10.1016/j.bbrc.2015.08.066
Cho A (2002) Matrix metalloproteinase-9 is necessary for the regulation of smooth muscle cell replication and migration after arterial injury. Circ Res 91(9):845–851. doi:10.1161/01.res.0000040420.17366.2e
Frye RA (2000) Phylogenetic classification of prokaryotic and eukaryotic Sir2-like proteins. Biochem Biophys Res Commun 273(2):793–798. doi:10.1006/bbrc.2000.3000
Gao R, Chen J, Hu Y, Li Z, Wang S, Shetty S, Fu J (2014) Sirt1 deletion leads to enhanced inflammation and aggravates endotoxin-induced acute kidney injury. PLoS One 9(6):e98909. doi:10.1371/journal.pone.0098909
Guan H, Gao L, Zhu L, Yan L, Fu M, Chen C, Dong X, Wang L, Huang K, Jiang H (2012) Apigenin attenuates neointima formation via suppression of vascular smooth muscle cell phenotypic transformation. J Cell Biochem 113(4):1198–1207. doi:10.1002/jcb.23452
Guo L, Ning W, Tan Z, Gong Z, Li X (2014) Mechanism of matrix metalloproteinase axis-induced neointimal growth. J Mol Cell Cardiol 66:116–125. doi:10.1016/j.yjmcc.2013.11.014
Lee GR, Jang SH, Kim CJ, Kim AR, Yoon DJ, Park NH, Han IS (2014) Capsaicin suppresses the migration of cholangiocarcinoma cells by down-regulating matrix metalloproteinase-9 expression via the AMPK-NF-kappaB signaling pathway. Clin Exp Metastasis 31(8):897–907. doi:10.1007/s10585-014-9678-x
Li X, Zhang S, Blander G, Tse JG, Krieger M, Guarente L (2007) SIRT1 deacetylates and positively regulates the nuclear receptor LXR. Mol Cell 28(1):91–106. doi:10.1016/j.molcel.2007.07.032
Li L, Zhang H-N, Chen H-Z, Gao P, Zhu L-H, Li H-L, Lv X, Zhang Q-J, Zhang R, Wang Z, She Z-G, Zhang R, Wei Y-S, Du G-H, Liu D-P, Liang C-C (2011) SIRT1 acts as a modulator of neointima formation following vascular injury in mice. Circ Res 108(10):1180–1189. doi:10.1161/circresaha.110.237875
Li G, Liu D, Zhang Y, Qian Y, Zhang H, Guo S, Sunagawa M, Hisamitsu T, Liu Y (2013) Celastrol inhibits lipopolysaccharide-stimulated rheumatoid fibroblast-like synoviocyte invasion through suppression of TLR4/NF-kappaB-mediated matrix metalloproteinase-9 expression. PLoS One 8(7):e68905. doi:10.1371/journal.pone.0068905
Li BH, Yin YW, Liu Y, Pi Y, Guo L, Cao XJ, Gao CY, Zhang LL, Li JC (2014) TRPV1 activation impedes foam cell formation by inducing autophagy in oxLDL-treated vascular smooth muscle cells. Cell Death Dis 5:e1182. doi:10.1038/cddis.2014.146
Liu J, Ren Y, Kang L, Zhang L (2014) Oxidized low-density lipoprotein increases the proliferation and migration of human coronary artery smooth muscle cells through the upregulation of osteopontin. Int J Mol Med 33(5):1341–1347. doi:10.3892/ijmm.2014.1681
Liu Y, Li J, Yin Y, Li B, Guo L, Liao S, Cao X, Zhang M, Zhou Y, Chen L, Long C, Zhang L (2015) Effect of TRPV1 activation on proliferation of vascular smooth muscle cells in spontaneously hypertensive rats. J Third Mil Med Univ 37(8):762–766. doi:10.16016/j.1000-5404.201411015
Loftus IM, Naylor AR, Goodall S, Crowther M, Jones L, Bell PRF, Thompson MM (2000) Increased matrix metalloproteinase-9 activity in unstable carotid plaques: a potential role in acute plaque disruption. Stroke 31(1):40–47. doi:10.1161/01.str.31.1.40
Ma L, Zhong J, Zhao Z, Luo Z, Ma S, Sun J, He H, Zhu T, Liu D, Zhu Z, Tepel M (2011) Activation of TRPV1 reduces vascular lipid accumulation and attenuates atherosclerosis. Cardiovasc Res 92(3):504–513. doi:10.1093/cvr/cvr245
Mason DP, Kenagy RD, Hasenstab D, Bowen-Pope DF, Seifert RA, Coats S, Hawkins SM, Clowes AW (1999) Matrix metalloproteinase-9 overexpression enhances vascular smooth muscle cell migration and alters remodeling in the injured rat carotid artery. Circ Res 85(12):1179–1185. doi:10.1161/01.res.85.12.1179
McMurray HF, Parrott DP, Bowyer DE (1991) A standardised method of culturing aortic explants, suitable for the study of factors affecting the phenotypic modulation, migration and proliferation of aortic smooth muscle cells. Atherosclerosis 86(2–3):227–237. doi:10.1016/0021-9150(91)90219-S
Moon S-K, Cha B-Y, Kim C-H (2004) ERK1/2 mediates TNF-?-induced matrix metalloproteinase-9 expression in human vascular smooth muscle cells via the regulation of NF-?B and AP-1: involvement of the ras dependent pathway. J Cell Physiol 198(3):417–427. doi:10.1002/jcp.10435
Ota H, Akishita M, Eto M, Iijima K, Kaneki M, Ouchi Y (2007) Sirt1 modulates premature senescence-like phenotype in human endothelial cells. J Mol Cell Cardiol 43(5):571–579. doi:10.1016/j.yjmcc.2007.08.008
Rosenfeld ME, Ross R (1990) Macrophage and smooth muscle cell proliferation in atherosclerotic lesions of WHHL and comparably hypercholesterolemic fat-fed rabbits. Arterioscler Thromb Vasc Biol 10(5):680–687. doi:10.1161/01.atv.10.5.680
Saneipour M, Ghatreh-Samani K, Heydarian E, Farrokhi E, Abdian N (2015) Adiponectin inhibits oxidized low density lipoprotein-induced increase in matrix metalloproteinase 9 expression in vascular smooth muscle cells. ARYA Atheroscler 11(3):191–195
Stein S, Matter CM (2011) Protective roles of SIRT1 in atherosclerosis. Cell Cycle 10(4):640–647. doi:10.4161/cc.10.4.14863
Visse R, Nagase H (2003) Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res 92(8):827–839. doi:10.1161/01.RES.0000070112.80711.3D
Xu X, Wang P, Zhao Z, Cao T, He H, Luo Z, Zhong J, Gao F, Zhu Z, Li L, Yan Z, Chen J, Ni Y, Liu D, Zhu Z (2011) Activation of transient receptor potential vanilloid 1 by dietary capsaicin delays the onset of stroke in stroke-prone spontaneously hypertensive rats. Stroke 42(11):3245–3251. doi:10.1161/strokeaha.111.618306
Yang D, Luo Z, Ma S, Wong WT, Ma L, Zhong J, He H, Zhao Z, Cao T, Yan Z, Liu D, Arendshorst WJ, Huang Y, Tepel M, Zhu Z (2010) Activation of TRPV1 by dietary capsaicin improves endothelium-dependent vasorelaxation and prevents hypertension. Cell Metab 12(2):130–141. doi:10.1016/j.cmet.2010.05.015
Yu W, Fu YC, Chen CJ, Wang X, Wang W (2009) SIRT1: a novel target to prevent atherosclerosis. J Cell Biochem 108(1):10–13. doi:10.1002/jcb.22240
Zhang LL, Liu DY, Ma LQ, Luo ZD, Cao TB, Zhong J, Yan ZC, Wang LJ, Zhao ZG, Zhu SJ, Schrader M, Thilo F, Zhu ZM, Tepel M (2007) Activation of transient receptor potential vanilloid type-1 channel prevents adipogenesis and obesity. Circ Res 100(7):1063–1070. doi:10.1161/01.RES.0000262653.84850.8b
Zhang QJ, Wang Z, Chen HZ, Zhou S, Zheng W, Liu G, Wei YS, Cai H, Liu DP, Liang CC (2008) Endothelium-specific overexpression of class III deacetylase SIRT1 decreases atherosclerosis in apolipoprotein E-deficient mice. Cardiovasc Res 80(2):191–199. doi:10.1093/cvr/cvn224
Zhang H, Wang ZW, Wu HB, Li Z, Li LC, Hu XP, Ren ZL, Li BJ, Hu ZP (2013) Transforming growth factor-beta1 induces matrix metalloproteinase-9 expression in rat vascular smooth muscle cells via ROS-dependent ERK-NF-kappaB pathways. Mol Cell Biochem 375(1–2):11–21. doi:10.1007/s11010-012-1512-7
Zhang M-J, Yin Y-W, Li B-H, Liu Y, Liao S-Q, Gao C-Y, Li J-C, Zhang L-L (2015) The role of TRPV1 in improving VSMC function and attenuating hypertension. Prog Biophys Mol Biol 117(2–3):212–216. doi:10.1016/j.pbiomolbio.2015.03.004
Zordoky BN, Robertson IM, Dyck JR (2015) Preclinical and clinical evidence for the role of resveratrol in the treatment of cardiovascular diseases. Biochim Biophys Acta 1852(6):1155–1177. doi:10.1016/j.bbadis.2014.10.016
Acknowledgments
This work was supported by National Natural Science Foundation of China (NSFC 81471193 to Li-Li Zhang, 81271282 to Jing-Cheng Li and 81400967 to Yan Pi).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Electronic supplementary material
Below is the link to the electronic supplementary material.

Figure S1
Identification of primary VSMCs from WT mice by immunofluorescence. Primary VSMCs from WT mice were stained with α-SMA (red fluorescence). Cell nuclei were counterstained with DAPI (blue fluorescence). Scale bar = 40μm. (JPEG 187 kb)

Figure S2
Identification of primary VSMCs from WT mice by flow cytometry. Overlay histogram showed primary VSMCs from WT mice stained with α-SMA (orange line). Isotype control antibody (blue line) was rabbit IgG (monoclonal). Unlabeled control (red line) was cells without incubation with primary antibody and secondary antibody. Over 96% of primary cultured cells were α-SMA positive cells, namely VSMCs, under our experimental conditions. (JPEG 835 kb)
Rights and permissions
About this article
Cite this article
Zhang, MJ., Zhou, Y., Chen, L. et al. Impaired SIRT1 promotes the migration of vascular smooth muscle cell-derived foam cells. Histochem Cell Biol 146, 33–43 (2016). https://doi.org/10.1007/s00418-016-1408-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-016-1408-9