Abstract
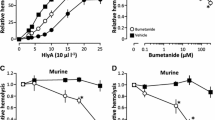
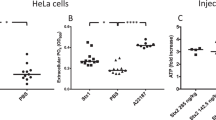
Recently, it was documented that α-haemolysin (HlyA) from Escherichia coli uses erythrocyte P2 receptors cause lysis. This finding was surprising as it appeared firmly established that HlyA-dependent pore formation per se is sufficient for full cell lysis. We discovered that HlyA induced a sequential process of shrinkage and swelling and that the final haemolysis is completely prevented by blockers of P2X receptors and pannexin channels. This finding has potential clinical relevance as it may offer specific pharmacological interference to ameliorate haemolysis inflicted by pore-forming bacterial toxins. In this context, it is essential to know whether this is specific to HlyA-induced cell damage or if other bacterial pore-forming toxins involve purinergic signals to orchestrate haemolysis. Here, we investigate if the haemolysis produced by α-toxin from Staphylococcus aureus involves P2 receptor activation. We observed that α-toxin-induced haemolysis is completely blocked by the unselective P2 receptor antagonist pyridoxalphosphate-6-azophenyl-2′,4′-disulfonic acid. Moreover, several selective blockers of P2X1 and P2X7 ionotropic receptors abolished haemolysis in murine and equine erythrocytes. Inhibitors of pannexin channels partially reduced the α-toxin induced lysis. Thus, we conclude that α-toxin, similar to HlyA from E. coli produces cell damage by specific activation of a purinergic signalling cascade. These data indicate that pore-forming toxins in general require purinergic signalling to elicit their toxicity.









Similar content being viewed by others
References
Arbuthnott JP, Lominski IR, Wright MR (1968) Inhibition of staphylococcal alpha-toxin. The effect of aromatic polysulphonic acids on the lethal effect of alpha-toxin in mice. Biochem J 108:49–55
Arvand M, Bhakdi S, Dahlback B, Preissner KT (1990) Staphylococcus aureus α-toxin attack on human platelets promotes assembly of the prothrombinase complex. J Biol Chem 265:14377–14381
Bantel H, Sinha B, Domschke W, Peters G, Schulze-Osthoff K, Janicke RU (2001) Alpha-toxin is a mediator of Staphylococcus aureus-induced cell death and activates caspases via the intrinsic death pathway independently of death receptor signaling. J Cell Biol 155:637–648
Benz R, Schmid A, Wagner W, Goebel W (1989) Pore formation by the Escherichia coli hemolysin: evidence for an association-dissociation equilibrium of the pore-forming aggregates. Infect Immun 57:887–895
Bezrukov MS, Vodyanoy I, Brutyan RA, Kasianowicz JJ (1996) Dynamics and free energy of polymers partitioning into a nanoscale pore. Macromolecules 29:8517–8522
Bhakdi S, Mackman N, Nicaud JM, Holland IB (1986) Escherichia coli hemolysin may damage target cell membranes by generating transmembrane pores. Infect Immun 52:63–69
Bhakdi S, Muhly M, Korom S, Hugo F (1989) Release of interleukin-1 beta associated with potent cytocidal action of staphylococcal alpha-toxin on human monocytes. Infect Immun 57:3512–3519
Bhakdi S, Weller U, Walev I, Martin E, Jonas D, Palmer M (1993) A guide to the use of pore-forming toxins for controlled permeabilization of cell membranes. Med Microbiol Immunol 182:167–175
Buerke U, Carter JM, Schlitt A, Russ M, Schmidt H, Sibelius U, Grandel U, Grimminger F, Seeger W, Mueller-Werdan U, Werdan K, Buerke M (2008) Apoptosis contributes to septic cardiomyopathy and is improved by simvastatin therapy. Shock 29:497–503
Cassidy PS, Harshman S (1973) The binding of staphylococcal 125I-alpha-toxin (B) to erythrocytes. J Biol Chem 248:5545–5546
Cooper LZ, Madoff MA, Weinstein L (1964) Hemolysis of rabbit erythrocytes by purified staphylococcal alpha-toxin. I. kinetics of the lytic reaction. J Bacteriol 87:127–135
Eisen V, Loveday C (1973) Effects of suramin on complement, blood clotting, fibrinolysis and kinin formation. Br J Pharmacol 49:678–687
Essmann F, Bantel H, Totzke G, Engels IH, Sinha B, Schulze-Osthoff K, Janicke RU (2003) Staphylococcus aureus alpha-toxin-induced cell death: predominant necrosis despite apoptotic caspase activation. Cell Death Differ 10:1260–1272
Foller M, Bobbala D, Koka S, Boini KM, Mahmud H, Kasinathan RS, Shumilina E, Amann K, Beranek G, Sausbier U, Ruth P, Sausbier M, Lang F, Huber SM (2010) Functional significance of the intermediate conductance Ca2+-activated K+ channel for the short-term survival of injured erythrocytes. Pflugers Arch 460:1029–1044
Fussle R, Bhakdi S, Sziegoleit A, Tranum-Jensen J, Kranz T, Wellensiek HJ (1981) On the mechanism of membrane damage by Staphylococcus aureus alpha-toxin. J Cell Biol 91:83–94
Hechler B, Eckly A, Ohlmann P, Cazenave JP, Gachet C (1998) The P2Y1 receptor, necessary but not sufficient to support full ADP-induced platelet aggregation, is not the target of the drug clopidogrel. Br J Haematol 103:858–866
Hollopeter G, Jantzen HM, Vincent D, Li G, England L, Ramakrishnan V, Yang RB, Nurden P, Nurden A, Julius D, Conley PB (2001) Identification of the platelet ADP receptor targeted by antithrombotic drugs. Nature 409:202–207
Hyland C, Vuillard L, Hughes C, Koronakis V (2001) Membrane interaction of Escherichia coli hemolysin: flotation and insertion-dependent labeling by phospholipid vesicles. J Bacteriol 183:5364–5370
Jin J, Daniel JL, Kunapuli SP (1998) Molecular basis for ADP-induced platelet activation. II. The P2Y1 receptor mediates ADP-induced intracellular calcium mobilization and shape change in platelets. J Biol Chem 273:2030–2034
Jonas D, Walev I, Berger T, Liebetrau M, Palmer M, Bhakdi S (1994) Novel path to apoptosis: small transmembrane pores created by staphylococcal alpha-toxin in T lymphocytes evoke internucleosomal DNA degradation. Infect Immun 62:1304–1312
Jorgensen SE, Hammer RF, Wu GK (1980) Effects of a single hit from the alpha hemolysin produced by Escherichia coli on the morphology of sheep erythrocytes. Infect Immun 27:988–994
Jorgensen SE, Mulcahy PF, Wu GK, Louis CF (1983) Calcium accumulation in human and sheep erythrocytes that is induced by Escherichia coli hemolysin. Toxicon 21:717–727
Lappin E, Ferguson AJ (2009) Gram-positive toxic shock syndromes. Lancet Infect Dis 9:281–290
Leclercq R (2009) Epidemiological and resistance issues in multidrug-resistant staphylococci and enterococci. Clin Microbiol Infect 15:224–231
Locovei S, Bao L, Dahl G (2006) Pannexin 1 in erythrocytes: function without a gap. Proc Natl Acad Sci USA 103:7655–7659
Locovei S, Wang J, Dahl G (2006) Activation of pannexin 1 channels by ATP through P2Y receptors and by cytoplasmic calcium. FEBS Lett 580:239–244
McGillivray DJ, Valincius G, Heinrich F, Robertson JW, Vanderah DJ, Febo-Ayala W, Ignatjev I, Losche M, Kasianowicz JJ (2009) Structure of functional Staphylococcus aureus alpha-hemolysin channels in tethered bilayer lipid membranes. Biophys J 96:1547–1553
Menestrina G (1986) Ionic channels formed by Staphylococcus aureus alpha-toxin: voltage-dependent inhibition by divalent and trivalent cations. J Membr Biol 90:177–190
Parrillo JE (1989) The cardiovascular pathophysiology of sepsis. Annu Rev Med 40:469–485
Qiu F, Dahl GP (2009) A permeant regulating its permeation pore: inhibition of pannexin 1 channels by ATP. Am J Physiol Cell Physiol 296(2):C250–5.
Rolf MG, Brearley CA, Mahaut-Smith MP (2001) Platelet shape change evoked by selective activation of P2X1 purinoceptors with alpha, beta-methylene ATP. Thromb Haemost 85:303–308
Sarri E, Bockmann I, Kempter U, Valeva A, von Eichel-Streiber C, Weichel O, Klein J (1998) Regulation of phospholipase D activity in synaptosomes permeabilized with Staphylococcus aureus alpha-toxin. FEBS Lett 440:287–290
Savi P, Labouret C, Delesque N, Guette F, Lupker J, Herbert JM (2001) P2y12, a new platelet ADP receptor, target of clopidogrel. Biochem Biophys Res Commun 283:379–383
Savi P, Beauverger P, Labouret C, Delfaud M, Salel V, Kaghad M, Herbert JM (1998) Role of P2Y1 purinoceptor in ADP-induced platelet activation. FEBS Lett 422:291–295
Sibelius U, Grandel U, Buerke M, Mueller D, Kiss L, Kraemer HJ, Braun-Dullaeus R, Haberbosch W, Seeger W, Grimminger F (2000) Staphylococcal alpha-toxin provokes coronary vasoconstriction and loss in myocardial contractility in perfused rat hearts: role of thromboxane generation. Circulation 101:78–85
Sim JA, Young MT, Sung HY, North RA, Surprenant A (2004) Reanalysis of P2X7 receptor expression in rodent brain. J Neurosci 24:6307–6314
Skals M, Jensen UB, Ousingsawat J, Kunzelmann K, Leipziger J, Praetorius HA (2010) Escherichia coli alpha-hemolysin triggers shrinkage of erythrocytes via KCa3.1 and TMEM16A channels with subsequent phosphatidylserine exposure. J Biol Chem 285:15557–15565
Skals MG, Jorgensen NR, Leipziger J, Praetorius HA (2009) Alpha-hemolysin from Escherichia coli uses endogenous amplification through P2X receptor activation to induce hemolysis. Proc Natl Acad Sci USA 106:4030–4035
Song L, Hobaugh MR, Shustak C, Cheley S, Bayley H, Gouaux JE (1996) Structure of staphylococcal alpha-hemolysin, a heptameric transmembrane pore. Science 274:1859–1866
Walev I, Martin E, Jonas D, Mohamadzadeh M, Muller-Klieser W, Kunz L, Bhakdi S (1993) Staphylococcal alpha-toxin kills human keratinocytes by permeabilizing the plasma membrane for monovalent ions. Infect Immun 61:4972–4979
Wright MR, Arbuthnott JP, Lominski IR (1968) Inhibition of staphylococcal alpha-toxin. A kinetic evaluation of aromatic polysulphonic acids as inhibitors of haemolysis. Biochem J 108:41–48
Yarovinsky TO, Monick MM, Husmann M, Hunninghake GW (2008) Interferons increase cell resistance to Staphylococcal alpha-toxin. Infect Immun 76:571–577
Acknowledgements
We thank Edith Bjoern Moeller for the skilled technical assistance and Niklas R. Jorgensen for the collaboration with regards the P2X7−/− mice. The project is financially supported by the Danish Medical Research Council, Danish National Research Foundation and the Aarhus University Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Suppl. Fig. 1
(PDF 264 kb)
Suppl. Fig.2
(PDF 266 kb)
Rights and permissions
About this article
Cite this article
Skals, M., Leipziger, J. & Praetorius, H.A. Haemolysis induced by α-toxin from Staphylococcus aureus requires P2X receptor activation. Pflugers Arch - Eur J Physiol 462, 669–679 (2011). https://doi.org/10.1007/s00424-011-1010-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-011-1010-x