Abstract
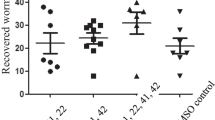
Methanolic extracts of Anogeissus leiocarpus has been considered locally to have the same antimalarial activities as artemisinin derivatives. This work studied the in vivo antiplasmodial activity of methanolic extracts of A. leiocarpus and its effect on oxidative stress and lipid profile in mice infected with Plasmodium bergheii. Mice used for this study were divided into five groups; four of the groups were infected with P. bergheii. The first group was not infected with the parasite. The second group was infected with parasite but not treated with antimalarial drugs (negative control). The third group was infected and treated with artesunat at 5 mg/kg body weight (positive control), while the fourth and fifth groups were infected and treated with 100 and 200 mg/kg body weight of extract of stem bark of A. leiocarpus, respectively. The rate of parasite clearance was higher in the group treated with 200 mg/kg body weight of extract of A. leiocarpus when compared with the groups treated with artesunat. Malondialdehyde (MDA) level was significantly higher (P < 0.05) in the serum of negative control as compared with other groups which have received treatment. MDA level was moderately higher in the liver homogenates of infected mice treated with artesunat than in other groups. There were significant increases (P < 0.05) in the levels of serum and liver superoxide dismutase of infected mice treated with 200 mg/kg body weight of A. leiocarpus when compared with other groups. Serum low density lipoprotein, total triglyceride, and total cholesterol were moderately higher in the group treated with artesunat than other groups, while high density lipoprotein (HDL) level was higher in the two groups treated with A. leiocarpus as compared with the group treated with artesunat. This study shows that the methanolic extract of A. leiocarpus has high antimalarial activities, high antioxidant property, and capable of boosting HDL level in malaria-infected organisms.






Similar content being viewed by others
References
Ademowo OG (2000) Malaria—Africa’s health challenge of the millennium. J Transfig Math 6:29–38
Akanbi OM, Odaibo AB, Ademowo OG (2009) Anti-MSP1(19) antibody (IgG) and reactive oxygen species (ROS) response against malaria infection in pregnancy in south western Nigeria. Asian Pac J Trop Med 2:9–15
Asres K, Bucar F, Edelsbrunner S, Kartnig T, Hoger G, Theil W (2001) Investigation on antimycobacterial activity of some Ethiopian medicinal plant. Phytother Res 15:323–326
Breeman JG, Alilio MS, Mills A (2004) Conquering the intolerable burden of malaria: what’s new, what’s needed: a summary. Am J Trop Med Hyg 71:1–15
De Clercq E (2005) Recent highlights in the development of new antiviral drugs. Curr Opin Microbiol 8(5):552–560
Faucher JF, Ngou-Milama E, Missinou MA, Ngomo R, Kombila M, Kremsner PG (2002) The impact of malaria on common lipid parameters. Parasitol Res 88:1040–1043
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–505
Gansané A, Sanon S, Ouattara LP, Traoré A, Hutter S, Ollivier E, Azas N et al (2010) Antiplasmodial activity and toxicity of crude extracts from alternatives parts of plants widely used for the treatment of malaria in Burkina Faso: contribution for their preservation. Parasitol Res 106:335–340
Gessler MC, Nkunya MH, Mwasumbi LB, Heinrich M, Tanner M (1994) Screening Tanzanian medical for antimalarial activity. Acta Trop 56:65–77
Greenwood BM, Bojang K, Whitty CJ, Targett GA (2005) Malaria. Lancet 365:1487–1498
Hale V, Keasling JD, Regnniner N, Diagana TT (2007) Microbilly derived artemisinin: a biotechnology solution to the global problem of access to affordable antimalarial drugs. Am J Trop Med Hyg 77:198–202
Khovidhunkit V, Memon RA, Feingold KR, Gruafeld C (2000) Infection and inflammation-induced proatherogenic changes of lipoproteins. J Infect Dis 181:8462–8472
Krishna AP, Chandrika SK, Manasa A, Shrikant LP (2009) Variation in common lipid parameters in malaria infected patients. Indian J Pharmacol 53:27–274
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Muregi FW, Chhabra SC, Njagi EN, Langat-Thoruwa CC, Njue WM, Orago AS, Omar SA, Ndiege IO (2003) In vivo antiplasmodial activity of some plants used in Kisii, Kenya against malaria and their chloroquine potentiation effects. J Ethnopharmacol 84:235–239
National Institutes of Health Consensus Development Conference Statement (NIHCDCS) (1992) Triglycerides, high density lipoprotein and coronary heart disease. Washington D.C., 26–28
Nilsson-Ehle I, Nilsson-Ehle P (1990) Changes in plasma lipoproteins in acute malaria. J Intern Med 227:151–155
Omosun YO, Anumudu CI, Adoro S, Odaibo AB, Sodeinde O, Holder AA et al (2005) Variation in the relationship between anti-MSP1 (19) antibody response and age in children infected with P. falciparum during the dry and rainy seasons. Acta Trop 95:233–247
Okeola VO, Adaramoye OA, Nneji CM, Falade CO, Farombi EO, Ademowo OG (2010) Antimalarial and antioxidant activities of methanolic extract of Nigella sativa seeds (black cumin) in mice infected with Plasmodium yoelli nigeriensis. Parasitol Res. doi:10.1007/s00436-010-2204-4
Shuaibu MN, Wuyep PA, Yanagi T, Tanaka T (2008) The use of microflourometric method from plant extract. Parasitol Res 10:436–450
Sibmooh N, Yamanont P, Krudsood S (2004) Increased fluidity and oxidation of malarial lipoproteins: relation with severity and induction of endothelial expression of adhesion molecules. Lipids Health Dis 3:15. doi:10.1186(1476):1–11.
Sirima SB, Gansane A (2007) Artesunate-amodiaquine for the treatment of uncomplicated malaria. Expert Opin Investig Drugs 16:1079–1085
Tietze NW (1990) Clinical guide to laboratory tests, 2nd edn. Saunders, Philadelphia, pp 554–556
Titanji VPK, Zofou D, Ngemenga MN (2008) The antimalarial potential of medicinal plants used for the treatment of malaria in Cameroonian folk medicine. Afr J Trad CAM 5:302–321
Trinder P (1969) Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem 6:24
Varshney R, Kale RK (1990) Effects of Calmodulin Antagonist on Radiation Induced Lipid Peroxidationin Microsomes. Int J Rad Biol 58:733–43
Vonthron-Sénécheau C, Weniger B, Ouattara M, Tra Bi F, Kamenan A, Lobstein A, Brun R, Anton R (2003) In vitro antiplasmodial activity and cytotoxicity of ethnobotanically selected Ivorian plants. J Ethnopharmacol 87(2–3):221–225
Woerdenberg HJ, Lugt CB, Prans N (1990) Artemisia annua: a source of novel antimalarial drugs. Pharm Wk Sci 12:169–181
World Health Organization (2003) Fact sheet no 134. Traditional medicine
Yakubu IM et al (1999) Biomass, productivity, chemical composition and plant physiognomy in lame burra game reserve. Bull Sci Assoc Niger 22:167–175
Acknowledgment
We acknowledge Dr. O. Aina of the Department of Parasitology, National Institute for Medical Research (NIMR), Yaba, Lagos, Nigeria who donated the malaria parasite. We are also grateful to Mr. B. F. Ologunorisa, a laboratory attendant, for assisting in harvesting the plant and Dr. A. O. Obembe, both of the Department of Plant Science and Biotechnology, Adekunle Ajasin University, for identifying the plant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akanbi, O.M., Omonkhua, A.A., Cyril-Olutayo, C.M. et al. The antiplasmodial activity of Anogeissus leiocarpus and its effect on oxidative stress and lipid profile in mice infected with Plasmodium bergheii . Parasitol Res 110, 219–226 (2012). https://doi.org/10.1007/s00436-011-2472-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2472-7