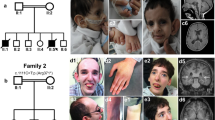
Abstract
We describe a female subject (DGAP100) with a 46,X,t(X;5)(p11.3;q35.3)inv(5)(q35.3q35.1)dn, severe psychomotor retardation with hypotonia, global postnatal growth restriction, microcephaly, globally reduced cerebral volume, seizures, facial dysmorphia and cleft palate. Fluorescence in situ hybridization and whole-genome sequencing demonstrated that the X chromosome breakpoint disrupts KDM6A in the second intron. No genes were directly disrupted on chromosome 5. KDM6A is a histone 3 lysine 27 demethylase and a histone 3 lysine 4 methyltransferase. Expression of KDM6A is significantly reduced in DGAP100 lymphoblastoid cells compared to control samples. We identified nine additional cases with neurodevelopmental delay and various other features consistent with the DGAP100 phenotype with copy number variation encompassing KDM6A from microarray databases. We evaluated haploinsufficiency of kdm6a in a zebrafish model. kdm6a is expressed in the pharyngeal arches and ethmoid plate of the developing zebrafish, while a kdm6a morpholino knockdown exhibited craniofacial defects. We conclude KDM6A dosage regulation is associated with severe and diverse structural defects and developmental abnormalities.





Similar content being viewed by others
References
Agger K, Cloos PAC, Christensen J, Pasini D, Rose S, Rappsilber J, Issaeva I, Canaani E, Salcini AE, Helin K (2007) UTX and JMJD3 are histone H3K27 demethylases involved in HOX gene regulation and development. Nature 449: 731-734. http://www.nature.com/nature/journal/v449/n7163/suppinfo/nature06145_S1.html
Barski A, Cuddapah S, Cui K, Roh T-Y, Schones DE, Wang Z, Wei G, Chepelev I, Zhao K (2007) High-resolution profiling of histone methylations in the human genome. Cell 129:823–837. doi:10.1016/j.cell.2007.05.009
Berger SL (2007) The complex language of chromatin regulation during transcription. Nature 447:407–412
Bernacki SH, Stankovic AK, Williams LO, Beck JC, Herndon JE, Snow-Bailey K, Prior TW, Matteson KJ, Wasserman LM, Cole EC, Stenzel TT (2003) Establishment of stably EBV-transformed cell lines from residual clinical blood samples for use in performance evaluation and quality assurance in molecular genetic testing. J Mol Diagn 5:227–230. doi:10.1016/s1525-1578(10)60478-3
Bernstein BE, Humphrey EL, Erlich RL, Schneider R, Bouman P, Liu JS, Kouzarides T, Schreiber SL (2002) Methylation of histone H3 Lys 4 in coding regions of active genes. Proc Natl Acad Sci 99:8695–8700. doi:10.1073/pnas.082249499
Chaturvedi C-P, Hosey AM, Palii C, Perez-Iratxeta C, Nakatani Y, Ranish JA, Dilworth FJ, Brand M (2009) Dual role for the methyltransferase G9a in the maintenance of β-globin gene transcription in adult erythroid cells. Proc Natl Acad Sci 106:18303–18308. doi:10.1073/pnas.0906769106
Chiang C, Jacobsen JC, Ernst C, Hanscom C, Heilbut A, Blumenthal I, Mills RE, Kirby A, Lindgren AM, Rudiger SR, McLaughlan CJ, Bawden CS, Reid SJ, Faull RLM, Snell RG, Hall IM, Shen Y, Ohsumi TK, Borowsky ML, Daly MJ, Lee C, Morton CC, MacDonald ME, Gusella JF, Talkowski ME (2012) Complex reorganization and predominant non-homologous repair following chromosomal breakage in karyotypically balanced germline rearrangements and transgenic integration. Nat Genet 44: 390–397. http://www.nature.com/ng/journal/v44/n4/abs/ng.2202.html (supplementary-information)
Cole TR, Hughes HE (1994) Sotos syndrome: a study of the diagnostic criteria and natural history. J Med Genet 31:20–32
Cooper GM, Coe BP, Girirajan S, Rosenfeld JA, Vu TH, Baker C, Williams C, Stalker H, Hamid R, Hannig V, Abdel-Hamid H, Bader P, McCracken E, Niyazov D, Leppig K, Thiese H, Hummel M, Alexander N, Gorski J, Kussmann J, Shashi V, Johnson K, Rehder C, Ballif BC, Shaffer LG, Eichler EE (2011) A copy number variation morbidity map of developmental delay. Nat Genet 43:838–846. http://www.nature.com/ng/journal/v43/n9/abs/ng.909.html#supplementary-information
Corpas M, Bragin E, Clayton S, Bevan P, Firth HV (2001) Interpretation of genomic copy number variants using DECIPHER. Current protocols in human genetics. Wiley, New York
Dougherty M, Kamel G, Shubinets V, Hickey G, Grimaldi M, Liao EC (2012) Embryonic fate map of first pharyngeal arch structures in the sox10: kaede zebrafish transgenic model. J Craniofac Surg 23:1333–1337. doi:10.1097/SCS.0b013e318260f20b
Ferreira MA, O’Donovan MC, Meng YA, Jones IR, Ruderfer DM, Jones L, Fan J, Kirov G, Perlis RH, Green EK, Smoller JW, Grozeva D, Stone J, Nikolov I, Chambert K, Hamshere ML, Nimgaonkar VL, Moskvina V, Thase ME, Caesar S, Sachs GS, Franklin J, Gordon-Smith K, Ardlie KG, Gabriel SB, Fraser C, Blumenstiel B, Defelice M, Breen G, Gill M, Morris DW, Elkin A, Muir WJ, McGhee KA, Williamson R, MacIntyre DJ, MacLean AW, St CD, Robinson M, Van Beck M, Pereira AC, Kandaswamy R, McQuillin A, Collier DA, Bass NJ, Young AH, Lawrence J, Ferrier IN, Anjorin A, Farmer A, Curtis D, Scolnick EM, McGuffin P, Daly MJ, Corvin AP, Holmans PA, Blackwood DH, Gurling HM, Owen MJ, Purcell SM, Sklar P, Craddock N (2008) Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 40:1056–1058. doi:10.1038/ng.209
Greenfield A, Carrel L, Pennisi D, Philippe C, Quaderi N, Siggers P, Steiner K, Tam PPL, Monaco AP, Willard HF, Koopman P (1998) The UTX gene escapes X inactivation in mice and humans. Hum Mol Genet 7:737–742. doi:10.1093/hmg/7.4.737
Hannibal MC, Buckingham KJ, Ng SB, Ming JE, Beck AE, McMillin MJ, Gildersleeve HI, Bigham AW, Tabor HK, Mefford HC, Cook J, K-i Yoshiura, Matsumoto T, Matsumoto N, Miyake N, Tonoki H, Naritomi K, Kaname T, Nagai T, Ohashi H, Kurosawa K, Hou J-W, Ohta T, Liang D, Sudo A, Morris CA, Banka S, Black GC, Clayton-Smith J, Nickerson DA, Zackai EH, Shaikh TH, Donnai D, Niikawa N, Shendure J, Bamshad MJ (2011) Spectrum of MLL2 (ALR) mutations in 110 cases of Kabuki syndrome. Am J Med Genet Part A 155:1511–1516. doi:10.1002/ajmg.a.34074
Herz H-M, Madden LD, Chen Z, Bolduc C, Buff E, Gupta R, Davuluri R, Shilatifard A, Hariharan IK, Bergmann A (2010) The H3K27me3 demethylase dUTX is a suppressor of notch- and Rb-dependent tumors in Drosophila. Mol Cell Biol 30:2485–2497. doi:10.1128/mcb.01633-09
Höglund P, Kurotaki N, Kytölä S, Miyake N, Somer M, Matsumoto N (2003) Familial Sotos syndrome is caused by a novel 1 bp deletion of the NSD1 gene. J Med Genet 40:51–54. doi:10.1136/jmg.40.1.51
Hong S, Cho Y-W, Yu L-R, Yu H, Veenstra TD, Ge K (2007) Identification of JmjC domain-containing UTX and JMJD3 as histone H3 lysine 27 demethylases. Proc Natl Acad Sci 104:18439–18444. doi:10.1073/pnas.0707292104
Huang N, Lee I, Marcotte EM, Hurles ME (2010) Characterising and predicting haploinsufficiency in the human genome. PLoS Genet 6:e1001154. doi:10.1371/journal.pgen.1001154
ISC (2008) Rare chromosomal deletions and duplications increase risk of schizophrenia. Nature 455:237–241
Issaeva I, Zonis Y, Rozovskaia T, Orlovsky K, Croce CM, Nakamura T, Mazo A, Eisenbach L, Canaani E (2007) Knockdown of ALR (MLL2) reveals ALR target genes and leads to alterations in cell adhesion and growth. Mol Cell Biol 27:1889–1903. doi:10.1128/mcb.01506-06
Jiang W, Wang J, Zhang Y (2012) Histone H3K27me3 demethylases KDM6A and KDM6B modulate definitive endoderm differentiation from human ESCs by regulating WNT signaling pathway. Cell Res. http://www.nature.com/cr/journal/vaop/ncurrent/suppinfo/cr2012119s1.html
Kasnauskiene J, Cimbalistiene L, Ciuladaite Z, Preiksaitiene E, Kucinskiene ZA, Hettinger JA, Sismani C, Patsalis PC, Kucinskas V (2011) De novo 5q35.5 duplication with clinical presentation of Sotos syndrome. Am J Med Genet A 155A:2501–2507
Kim E, Song J–J (2011) Diverse ways to be specific: a novel Zn-binding domain confers substrate specificity to UTX/KDM6A histone H3 Lys 27 demethylase. Genes Dev 25:2223–2226. doi:10.1101/gad.179473.111
Kouzarides T (2007) Chromatin Modifications and Their Function. Cell 128:693–705. doi:10.1016/j.cell.2007.02.005
Kuroki Y, Suzuki Y, Chyo H, Hata A, Matsui I (1981) A new malformation syndrome of long palpebral fissures, large ears, depressed nasal tip, and skeletal anomalies associated with postnatal dwarfism and mental retardation. J Pediatr 99:570–573. doi:10.1016/s0022-3476(81)80256-9
Kurotaki N, Imaizumi K, Harada N, Masuno M, Kondoh T, Nagai T, Ohashi H, Naritomi K, Tsukahara M, Makita Y, Sugimoto T, Sonoda T, Hasegawa T, Chinen Y, Tomita H-A, Kinoshita A, Mizuguchi T, Yoshiura K-i, Ohta T, Kishino T, Fukushima Y, Niikawa N, Matsumoto N (2002) Haploinsufficiency of NSD1 causes Sotos syndrome. Nat Genet 30:365–366
Kurotaki N, Harada N, Shimokawa O, Miyake N, Kawame H, Uetake K, Makita Y, Kondoh T, Ogata T, Hasegawa T, Nagai T, Ozaki T, Touyama M, Shenhav R, Ohashi H, Medne L, Shiihara T, Ohtsu S, Kato Z-i, Okamoto N, Nishimoto J, Lev D, Miyoshi Y, Ishikiriyama S, Sonoda T, Sakazume S, Fukushima Y, Kurosawa K, Cheng J-F, Yoshiura K-i, Ohta T, Kishino T, Niikawa N, Matsumoto N (2003) Fifty microdeletions among 112 cases of Sotos syndrome: low copy repeats possibly mediate the common deletion. Hum Mutat 22:378–387. doi:10.1002/humu.10270
Lan F, Bayliss PE, Rinn JL, Whetstine JR, Wang JK, Chen S, Iwase S, Alpatov R, Issaeva I, Canaani E, Roberts TM, Chang HY, Shi Y (2007) A histone H3 lysine 27 demethylase regulates animal posterior development. Nature 449: 689–694. http://www.nature.com/nature/journal/v449/n7163/suppinfo/nature06192_S1.html
Lederer D, Grisart B, Digilio Maria C, Benoit V, Crespin M, Ghariani Sophie C, Maystadt I, Dallapiccola B, Verellen-Dumoulin C (2012) Deletion of KDM6A, a histone demethylase interacting with MLL2, in three patients with Kabuki syndrome. Am J Hum Genet 90:119–124. doi:10.1016/j.ajhg.2011.11.021
Lee MG, Villa R, Trojer P, Norman J, Yan K-P, Reinberg D, Di Croce L, Shiekhattar R (2007) Demethylation of H3K27 regulates polycomb recruitment and H2A ubiquitination. Science 318:447–450. doi:10.1126/science.1149042
Lee S, Lee Jae W, Lee S-K (2012) UTX, a histone H3-lysine 27 demethylase, acts as a critical switch to activate the Cardiac Developmental Program. Dev Cell 22:25–37. doi:10.1016/j.devcel.2011.11.009
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25:1754–1760. doi:10.1093/bioinformatics/btp324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R, Subgroup GPDP (2009) The sequence alignment/Map format and SAMtools. Bioinformatics 25:2078–2079. doi:10.1093/bioinformatics/btp352
Li Y, Bögershausen N, Alanay Y, Simsek Kiper P, Plume N, Keupp K, Pohl E, Pawlik B, Rachwalski M, Milz E, Thoenes M, Albrecht B, Prott E-C, Lehmkühler M, Demuth S, Utine G, Boduroglu K, Frankenbusch K, Borck G, Gillessen-Kaesbach G, Yigit G, Wieczorek D, Wollnik B (2011) A mutation screen in patients with Kabuki syndrome. Hum Genet 130:715–724. doi:10.1007/s00439-011-1004-y
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. doi:10.1006/meth.2001.1262
Mansour AA, Gafni O, Weinberger L, Zviran A, Ayyash M, Rais Y, Krupalnik V, Zerbib M, Amann-Zalcenstein D, Maza I, Geula S, Viukov S, Holtzman L, Pribluda A, Canaani E, Horn-Saban S, Amit I, Novershtern N, Hanna JH (2012) The H3K27 demethylase Utx regulates somatic and germ cell epigenetic reprogramming. Nature 488: 409–413. http://www.nature.com/nature/journal/v488/n7411/abs/nature11272.html (supplementary-information)
Martin C, Zhang Y (2005) The diverse functions of histone lysine methylation. Nat Rev Mol Cell Biol 6:838–849
Micale L, Augello B, Fusco C, Selicorni A, Loviglio M, Silengo M, Reymond A, Gumiero B, Zucchetti F, D’Addetta E, Belligni E, Calcagni A, Digilio M, Dallapiccola B, Faravelli F, Forzano F, Accadia M, Bonfante A, Clementi M, Daolio C, Douzgou S, Ferrari P, Fischetto R, Garavelli L, Lapi E, Mattina T, Melis D, Patricelli M, Priolo M, Prontera P (2011) Mutation spectrum of MLL2 in a cohort of kabuki syndrome patients. Orphanet J Rare Dis 6:38
Miller SA, Mohn SE, Weinmann AS (2010) Jmjd3 and UTX play a demethylase-independent role in chromatin remodeling to regulate T-box family member-dependent gene expression. Mol Cell 40:594–605. doi:10.1016/j.molcel.2010.10.028
Miyake N, Mizuno S, Okamoto N, Ohashi H, Shiina M, Ogata K, Tsurusaki Y, Nakashima M, Saitsu H, Niikawa N, Matsumoto N (2013) KDM6A point mutations cause Kabuki syndrome. Hum Mutat 34:108–110. doi:10.1002/humu.22229
Ney PA, Andrews NC, Jane SM, Safer B, Purucker ME, Weremowicz S, Morton CC, Goff SC, Orkin SH, Nienhuis AW (1993) Purification of the human NF-E2 complex: cDNA cloning of the hematopoietic cell-specific subunit and evidence for an associated partner. Mol Cell Biol 13:5604–5612. doi:10.1128/mcb.13.9.5604
Ng SB, Bigham AW, Buckingham KJ, Hannibal MC, McMillin MJ, Gildersleeve HI, Beck AE, Tabor HK, Cooper GM, Mefford HC, Lee C, Turner EH, Smith JD, Rieder MJ, Yoshiura K-i, Matsumoto N, Ohta T, Niikawa N, Nickerson DA, Bamshad MJ, Shendure J (2010) Exome sequencing identifies MLL2 mutations as a cause of Kabuki syndrome. Nat Genet 42: 790–793. http://www.nature.com/ng/journal/v42/n9/abs/ng.646.html (supplementary-information)
Niikawa N, Matsuura N, Fukushima Y, Ohsawa T, Kajii T (1981) Kabuki make-up syndrome: a syndrome of mental retardation, unusual facies, large and protruding ears, and postnatal growth deficiency. J Pediatr 99:565–569. doi:10.1016/s0022-3476(81)80255-7
O’Donovan MC, Craddock N, Norton N, Williams H, Peirce T, Moskvina V, Nikolov I, Hamshere M, Carroll L, Georgieva L, Dwyer S, Holmans P, Marchini JL, Spencer CC, Howie B, Leung HT, Hartmann AM, Moller HJ, Morris DW, Shi Y, Feng G, Hoffmann P, Propping P, Vasilescu C, Maier W, Rietschel M, Zammit S, Schumacher J, Quinn EM, Schulze TG, Williams NM, Giegling I, Iwata N, Ikeda M, Darvasi A, Shifman S, He L, Duan J, Sanders AR, Levinson DF, Gejman PV, Cichon S, Nothen MM, Gill M, Corvin A, Rujescu D, Kirov G, Owen MJ, Buccola NG, Mowry BJ, Freedman R, Amin F, Black DW, Silverman JM, Byerley WF, Cloninger CR (2008) Identification of loci associated with schizophrenia by genome-wide association and follow-up. Nat Genet 40:1053–1055. doi:10.1038/ng.201
Paulussen ADC, Stegmann APA, Blok MJ, Tserpelis D, Posma-Velter C, Detisch Y, Smeets EEJGL, Wagemans A, Schrander JJP, van den Boogaard M-JH, van der Smagt J, van Haeringen A, Stolte-Dijkstra I, Kerstjens-Frederikse WS, Mancini GM, Wessels MW, Hennekam RCM, Vreeburg M, Geraedts J, de Ravel T, Fryns J-P, Smeets HJ, Devriendt K, Schrander-Stumpel CTRM (2011) MLL2 mutation spectrum in 45 patients with Kabuki syndrome. Hum Mutat 32:E2018–E2025. doi:10.1002/humu.21416
Purcell SM, Wray NR, Stone JL, Visscher PM, O’Donovan MC, Sullivan PF, Sklar P (2009) Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature 460:748–752. doi:10.1038/nature08185
Santos-Rosa H, Schneider R, Bannister AJ, Sherriff J, Bernstein BE, Emre NCT, Schreiber SL, Mellor J, Kouzarides T (2002) Active genes are tri-methylated at K4 of histone H3. Nature 419: 407–411. http://www.nature.com/nature/journal/v419/n6905/suppinfo/nature01080_S1.html
Schilling TF, Kimmel CB (1997) Musculoskeletal patterning in the pharyngeal segments of the zebrafish embryo. Development 124:2945–2960
Schuettengruber B, Chourrout D, Vervoort M, Leblanc B, Cavalli G (2007) Genome regulation by polycomb and trithorax proteins. Cell 128:735–745. doi:10.1016/j.cell.2007.02.009
Seenundun S, Rampalli S, Liu Q-C, Aziz A, Palii C, Hong S, Blais A, Brand M, Ge K, Dilworth FJ (2010) UTX mediates demethylation of H3K27me3 at muscle-specific genes during myogenesis. EMBO J 29: 1401–1411. http://www.nature.com/emboj/journal/v29/n8/suppinfo/emboj201037a_S1.html
Sengoku T, Yokoyama S (2011) Structural basis for histone H3 Lys 27 demethylation by UTX/KDM6A. Genes Dev 25:2266–2277. doi:10.1101/gad.172296.111
Shpargel KB, Sengoku T, Yokoyama S, Magnuson T (2012) UTX and UTY demonstrate histone demethylase-independent function in mouse embryonic development. PLoS Genet 8:e1002964. doi:10.1371/journal.pgen.1002964
Sims Iii RJ, Nishioka K, Reinberg D (2003) Histone lysine methylation: a signature for chromatin function. Trends Genet 19:629–639. doi:10.1016/j.tig.2003.09.007
Smith EN, Bloss CS, Badner JA, Barrett T, Belmonte PL, Berrettini W, Byerley W, Coryell W, Craig D, Edenberg HJ, Eskin E, Foroud T, Gershon E, Greenwood TA, Hipolito M, Koller DL, Lawson WB, Liu C, Lohoff F, McInnis MG, McMahon FJ, Mirel DB, Murray SS, Nievergelt C, Nurnberger J, Nwulia EA, Paschall J, Potash JB, Rice J, Schulze TG, Scheftner W, Panganiban C, Zaitlen N, Zandi PP, Zollner S, Schork NJ, Kelsoe JR (2009) Genome-wide association study of bipolar disorder in European American and African American individuals. Mol Psychiatry 14:755–763. doi:10.1038/mp.2009.43
Sotos JF, Dodge PR, Muirhead D, Crawford JD, Talbot NB (1964) Cerebral gigantism in childhood. N Engl J Med 271:109–116. doi:10.1056/NEJM196407162710301
Sullivan PF, Lin D, Tzeng JY, van den Oord E, Perkins D, Stroup TS, Wagner M, Lee S, Wright FA, Zou F, Liu W, Downing AM, Lieberman J, Close SL (2008) Genomewide association for schizophrenia in the CATIE study: results of stage 1. Mol Psychiatry 13:570–584. http://www.nature.com/mp/journal/v13/n6/suppinfo/mp200825s1.html
Talkowski ME, Ernst C, Heilbut A, Chiang C, Hanscom C, Lindgren A, Kirby A, Liu S, Muddukrishna B, Ohsumi Toshiro K, Shen Y, Borowsky M, Daly Mark J, Morton Cynthia C, Gusella James F (2011a) Next-generation sequencing strategies enable routine detection of balanced chromosome rearrangements for Clinical Diagnostics and Genetic Research. Am J Hum Genet 88:469–481. doi:10.1016/j.ajhg.2011.03.013
Talkowski ME, Mullegama Sureni V, Rosenfeld Jill A, van Bon Bregje WM, Shen Y, Repnikova Elena A, Gastier-Foster J, Thrush Devon L, Kathiresan S, Ruderfer Douglas M, Chiang C, Hanscom C, Ernst C, Lindgren Amelia M, Morton Cynthia C, An Y, Astbury C, Brueton Louise A, Lichtenbelt Klaske D, Ades Lesley C, Fichera M, Romano C, Innis Jeffrey W, Williams Charles A, Bartholomew D, Van Allen Margot I, Parikh A, Zhang L, Wu B-L, Pyatt Robert E, Schwartz S, Shaffer Lisa G, de Vries Bert BA, Gusella James F, Elsea Sarah H (2011b) Assessment of 2q23.1 microdeletion syndrome implicates MBD5 as a single causal locus of intellectual disability, epilepsy, and autism spectrum disorder. Am J Hum Genet 89:551–563. doi:10.1016/j.ajhg.2011.09.011
Talkowski ME, Rosenfeld Jill A, Blumenthal I, Pillalamarri V, Chiang C, Heilbut A, Ernst C, Hanscom C, Rossin E, Lindgren AM, Pereira S, Ruderfer D, Kirby A, Ripke S, Harris DJ, Lee J-H, Ha K, Kim H-G, Solomon Benjamin D, Gropman Andrea L, Lucente D, Sims K, Ohsumi Toshiro K, Borowsky Mark L, Loranger S, Quade B, Lage K, Miles J, Wu B-L, Shen Y, Neale B, Shaffer Lisa G, Daly Mark J, Morton Cynthia C, Gusella James F (2012) Sequencing chromosomal abnormalities reveals neurodevelopmental loci that confer risk across diagnostic boundaries. Cell 149:525–537. doi:10.1016/j.cell.2012.03.028
Tatton-Brown K, Douglas J, Coleman K, Baujat G, Cole TRP, Das S, Horn D, Hughes HE, Temple IK, Faravelli F, Waggoner D, Türkmen S, Cormier-Daire V, Irrthum A, Rahman N (2005) Genotype-phenotype associations in Sotos syndrome: an analysis of 266 individuals with NSD1 aberrations. Am J Hum Genet 77:193–204. doi:10.1086/432082
Thisse C, Thisse B (2008) High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat Protoc 3:59–69. doi:10.1038/nprot.2007.514
Walker M, Kimmel C (2007) A two-color acid-free cartilage and bone stain for zebrafish larvae. Biotech Histochem 82:23–28. doi:10.1080/10520290701333558
Wang JK, Tsai M-C, Poulin G, Adler AS, Chen S, Liu H, Shi Y, Chang HY (2010) The histone demethylase UTX enables RB-dependent cell fate control. Genes Dev 24:327–332. doi:10.1101/gad.1882610
Westerfield M (1993) The zebrafish book: a guide for the laboratory use of zebrafish (Brachydanio rerio). University of Oregon Press, Eugene
Wysocka J, Swigut T, Milne TA, Dou Y, Zhang X, Burlingame AL, Roeder RG, Brivanlou AH, Allis CD (2005) WDR5 associates with histone H3 methylated at K4 and is essential for H3 K4 methylation and vertebrate development. Cell 121:859–872. doi:10.1016/j.cell.2005.03.036
Xu J, Burgoyne PS, Arnold AP (2002) Sex differences in sex chromosome gene expression in mouse brain. Hum Mol Genet 11:1409–1419. doi:10.1093/hmg/11.12.1409
Xu J, Deng X, Watkins R, Disteche CM (2008) Sex-specific differences in expression of histone demethylases Utx and Uty in mouse brain and neurons. J Neurosci 28:4521–4527. doi:10.1523/jneurosci.5382-07.2008
Acknowledgments
We would like to thank DGAP100 and her family for participating in our research study and all of the healthcare professionals that contributed to this effort. We would also like to thank Mary Anne Anderson for her technical assistance at the Massachusetts General Hospital Genomics Core Facility in the Center for Human Genetic Research, Tammy Gillis and the MGH NextGen Sequencing Core, and the Cytogenetics Core of Dana Farber Harvard Cancer Center (P30 CA006516). This work was supported by PO1GM061354 (CCM), HD065286 (JFG) and MH095867 (MET) from the National Institutes of Health and the National Institute of General Medical Sciences, and March of Dimes and Shriners Hospitals for Children (ECL).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lindgren, A.M., Hoyos, T., Talkowski, M.E. et al. Haploinsufficiency of KDM6A is associated with severe psychomotor retardation, global growth restriction, seizures and cleft palate. Hum Genet 132, 537–552 (2013). https://doi.org/10.1007/s00439-013-1263-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-013-1263-x