Abstract
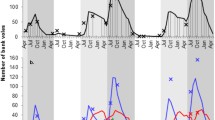
The effect of intermittently occurring, non-reservoir host species on pathogen transmission and prevalence in a reservoir population is poorly understood. We investigated whether voles, Microtus spp., which occur intermittently, influenced estimated standing antibody prevalence (ESAP) to Sin Nombre hantavirus (SNV, Bunyaviridae: Hantavirus) among deer mice, Peromyscus maniculatus, whose populations are persistent. We used 14 years of data from central Montana to investigate whether ESAP among deer mice was related to vole presence or abundance while controlling for the relationship between deer mouse abundance and ESAP. We found a reduction in deer mouse ESAP associated with the presence of voles, independent of vole abundance. A number of studies have documented that geographic locations which support a higher host diversity can be associated with reductions in pathogen prevalence by a hypothesized dilution effect. We suggest a dilution effect may also occur in a temporal dimension at sites where host richness fluctuates. Preservation of host diversity and optimization of environmental conditions which promote occurrence of ephemeral species, such as voles, may result in a decreased ESAP to hantaviruses among reservoir hosts. Our results may extend to other zoonotic infectious diseases.


Similar content being viewed by others
References
Altizer S, Dobson A, Hosseini P, Hudson P, Pascual M, Rohani P (2006) Seasonality and the dynamics of infectious diseases. Ecol Lett 9:467–484
Anderson RM, May RM (1992) Infectious diseases of humans: dynamics and control. Oxford University Press, New York
Bennett SG, Webb JP, Madon MB, Childs JE, Ksiazek TG, Torrez-Martinez N, Hjelle B (1999) Hantavirus (Bunyaviridae) infections in rodents from orange and San Diego Counties, California. Am J Trop Med Hyg 60:75–84
Bowker LS, Pearson PG (1975) Habitat orientation and interspecific interaction of Microtus pennsylvanicus and Peromyscus leucopus. Am Midl Nat 94:491–496
Calisher CH, Root JJ, Mills JN, Beaty BJ (2002) Assessment of ecologic and biologic factors leading to hantavirus pulmonary syndrome, Colorado, USA. Croat Med J 43:330–337
Calisher CH, Root JJ, Mills JN, Rowe JE, Reeder SA, Jentes ES, Wagoner K, Beaty BJ (2005) Epizootiology of Sin Nombre and El Moro Canyon hantaviruses, southeastern Colorado, 1995–2000. J Wildl Dis 41:1–11
Calisher CH, Wagoner KD, Amman BR, Root JJ, Douglass RJ, Kuenzi AJ, Abbott KD, Parmenter C, Yates TL, Ksiazek TG, Beaty BJ, Mills JN (2007) Demographic factors associated with prevalence of antibody to Sin Nombre Virus in deer mice in the Western United States. J Wildl Dis 43:1–11
Carver S, Sakalidis V, Weinstein P (2008) House mouse abundance and Ross River virus notifications in Victoria, Australia. Int J Infect Dis 12:528–533
Carver S, Mills JN, Kuenzi A, Flietstra T, Douglass R (2010) Sampling frequency differentially influences interpretation of zoonotic pathogen and host dynamics: Sin Nombre virus and deer mice. Vector-Borne Zoonot Dis 10:575–583
Cavanagh RD, Lambin X, Ergon T, Bennett M, Graham IM, van Soolingen D, Begon M (2004) Disease dynamics in cyclic populations of field voles (Microtus agrestis): cowpox virus and vole tuberculosis (Mycobacterium microti). Proc R Soc Lond B 271:859–867
Clay CA, Lehmer EM, Jeor SS, Dearing MD (2009a) Sin Nombre virus and rodent species diversity: a test of the dilution and amplification hypotheses. PLoS One 4:e6467
Clay CA, Lehmer EM, Jeor SS, Dearing MD (2009b) Testing mechanisms of the dilution effect: deer mice encounter rates, Sin Nombre virus prevalence and species diversity. Ecohealth 6:250–259
Clay CA, Lehmer EM, Previtali A, St. Jeor S, Dearing MD (2009c) Contact heterogeneity in deer mice: implications for Sin Nombre virus transmission. Proc R Soc Lond B 276:1305–1312
Dickman CR, Mahon PS, Masters P, Gibson DF (1999) Long-term dynamics of rodent populations in arid Australia: the influence of rainfall. Wildl Res 26:389–403
Douglass RJ, VanHorn R, Coffin KW, Zanto SN (1996) Hantavirus in Montana deer mouse populations: preliminary results. J Wildl Dis 32:527–530
Douglass RJ, Wilson T, Semmens WJ, Zanto SN, Bond CW, Van Horn RC, Mills JN (2001) Longitudinal studies of Sin Nombre virus in deer mouse-dominated ecosystems of Montana. Am J Trop Med Hyg 65:33–41
Ezenwa VO, Godsey MS, King RJ, Guptill SC (2006) Avian diversity and West Nile virus: testing associations between biodiversity and infectious disease risk. Proc R Soc Lond B 273:109–117
Feldmann H, Sanchez A, Morzunov S, Spiropoulou CF, Rollin PE, Ksiazek TG, Peters CJ, Nichol ST (1993) Utilization of autopsy RNA for the synthesis of the nucleocapsid antigen of a newly recognized virus associated with hantavirus pulmonary syndrome. Virus Res 30:351–367
Hall ER, Kelson KR (1959) Mammals of North America. Ronald, New York
Hjelle B, Jenison SA, Goade DE, Green WB, Feddersen RM, Scott AA (1995) Hantaviruses: clinical, microbiologic, and epidemiologic aspects. Crit Rev Clin Lab Sci 32:469–508
Keeling MJ, Rohani P (2008) Modeling infectious diseases in humans and animals. Princeton University Press, Princeton
Keesing F, Holt RD, Ostfeld R (2006) Effects of species diversity on disease risk. Ecol Lett 9:485–498
Korpimaki E, Brown PR, Jacob J, Pech RP (2004) The puzzles of population cycles and outbreaks of small mammals solved? Bioscience 54:1071–1079
Kozakiewicz A, Boniecki P (1994) Intra-specific and interspecific behaviors in bank vole and striped field-mouse under enclosure conditions. Acta Theriol 39:29–36
Krebs CJ (1966) Demographic changes in fluctuating populations of Microtus californicus. Ecol Monogr 36:239–273
Krebs CJ (1996) Population cycles revisited. J Mammal 77:8–24
Krebs CJ, Myers JM (1974) Population cycles in small mammals. Adv Ecol Res 8:267–399
LoGiudice K, Ostfeld RS, Schmidt KA, Keesing F (2003) The ecology of infectious disease: effects of host diversity and community composition on Lyme disease risk. Proc Natl Acad Sci USA 100:567–571
Luis AD, Douglass RJ, Mills JN, Bjørnstad ON (2010) The effect of seasonality, density and climate on the population dynamics of Montana deer mice, important reservoir hosts for Sin Nombre hantavirus. J Anim Ecol 79:462–470
Matuschka FR, Spielman A (1992) Loss of Lyme disease spirochetes from Ixodes ricinus ticks feeding on European blackbirds. Exp Parasitol 74:151–158
Mills JN (2005) Regulation of rodent-borne viruses in the natural host: implications for human disease. Arch Virol Suppl 19:45–57
Mills JN (2006) Biodiversity loss and emerging infectious disease: an example from the rodent-borne hemorrhagic fevers. Biodiversity 7:9–17
Mills JN, Yates TL, Childs JE, Parmenter RR, Ksiazek TG, Rollin PE, Peters CJ (1995) Guidelines for working with rodents potentially infected with hantavirus. J Mammal 76:716–722
Mills JN, Ksiazek TG, Ellis BA, Rollin PE, Nichol ST, Yates TL, Gannon WL, Levy CE, Engelthaler DM, Davis T, Tanda DT, Frampton JW, Nichols CR, Peters CJ, Childs JE (1997) Patterns of association with host and habitat: antibody reactive with Sin Nombre virus in small mammals in the major biotic communities of the southwestern United States. Am J Trop Med Hyg 56:273–284
Mills JN, Ksiazek TG, Peters CJ, Childs JE (1999) Long-term studies of hantavirus reservoir populations in the southwestern United States: a synthesis. Emerg Infect Dis 5:135–142
Morris RD, Grant PR (1972) Experimental studies of competitive interaction in a two-species system. J Anim Ecol 41:275–290
Nichol ST, Spiropoulou CF, Morzunov S, Rollin PE, Ksiazek TG, Feldmann H, Sanchez A, Childs J, Zaki S, Peters CJ (1993) Genetic identification of a hantavirus associated with an outbreak of acute respiratory illness. Science 262:914–917
Norman R, Bowers RG, Begon M, Hudson PJ (1999) Persistence of tick-horne virus in the presence of multiple host species: tick reservoirs and parasite mediated competition. J Theor Biol 200:111–118
Ostfeld RS (1985) Limiting resources and territoriality in microtine rodents. Am Nat 126:1–15
Ostfeld RS, Keesing F (2000a) Biodiversity and disease risk: the case of Lyme disease. Conserv Biol 14:722–728
Ostfeld RS, Keesing F (2000b) The function of biodiversity in the ecology of vector-borne zoonotic diseases. Can J Zool 78:2061–2078
Ovadia O, Abramsky Z, Kotler BP, Pinshow B (2005) Inter-specific competitors reduce inter-gender competition in Negev Desert gerbils. Oecologia 142:480–488
Pearce J, Venier L (2005) Small mammals as bioindicators of sustainable boreal forest management. For Ecol Manag 208:153–175
Rawlings JA, TorrezMartinez N, Neill SU, Moore GM, Hicks BN, Pichuantes S, Nguyen A, Bharadwaj M, Hjelle B (1996) Cocirculation of multiple hantaviruses in Texas, with characterization of the small (S) genome of a previously undescribed virus of cotton rats (Sigmodon hispidus). Am J Trop Med Hyg 55:672–679
Rowe JE, Stjeor SC, Riolo J, Otteson EW, Monroe MC, Henderson WW, Ksiazek TG, Rollin PE, Nichol ST (1995) Coexistence of several novel hantaviruses in rodents indigenous to North America. Virology 213:122–130
Safronetz D, Drebot MA, Artsob H, Cote T, Makowski K, Lindsay LR (2008) Sin Nombre virus shedding patterns in naturally infected deer mice (Peromyscus maniculatus) in relation to duration of infection. Vector-Borne Zoonotic Dis 8:97–100
Scharninghausen JJ, Pitts RM, Bickham JW, Davis DS, Mills JN (1999) Evidence of hantavirus infection in Microtus orchrogaster in St. Louis County, Missouri. Trans Mo Acad Sci 33–34:23–25
Schountz T, Calisher CH, Richens TR, Rich AA, Doty JB, Hughes MT, Beaty BJ (2007) Rapid field immunoassay for detecting antibody to Sin Nombre virus in deer mice. Emerg Infect Dis 13:1604–1607
Schulte-Hostedde AI, Brooks RJ (1997) An experimental test of habitat selection by rodents of Algonquin Park. Can J Zool 75:1989–1993
Singleton GR (1985) Population dynamics of Mus musculus and its parasites in Mallee wheatlands in Victoria Australia during and after a drought. Aust Wildl Res 12:437–446
Singleton G (1989) Population dynamics of an outbreak of house mouse (Mus domesticus) in the Mallee wheatlands of Australia—hypothesis of plague formation. J Zool 219:495–515
Song WM, Torrezmartinez N, Irwin W, Harrison FJ, Davis R, Ascher M, Jay M, Hjelle B (1995) Isla Vista virus—a genetically novel hantavirus of the California vole Microtus californicus. J Gen Virol 76:3195–3199
Telfer S, Bown KJ, Sekules R, Begon I, Hayden T, Birtles R (2005) Disruption of a host-parasite system following the introduction of an exotic host species. Parasitology 130:661–668
Telfer S, Begon M, Bennett M, Bown KJ, Burthe S, Lambin X, Telford G, Birtles R (2007) Contrasting dynamics of Bartonella spp. in cyclic field vole populations: the impact of vector and host dynamics. Parasitology 134:413–425
Tersago K, Schreurs A, Linard C, Verhagen R, van Dongen S, Leirs H (2008) Population, environmental, and community effects on local bank vole (Myodes glareolus) Puumala virus infection in an area with low human incidence. Vector-Borne Zoonotic Dis 8:235–244
Van Buskirk J, Ostfeld RS (1995) Controlling Lyme disease by modifying the density and species composition of tick hosts. Ecol Appl 5:1133–1140
Wilson BA, Bourne AR, Jessop RE (1986) Ecology of small mammals in coastal heath at Anglesea, Victoria. Aust Wildl Res 13:397–406
Yanagihara R, Daum CA, Lee PW, Baek LJ, Amyx HL, Gajdusek DC, Gibbs CJ (1987) Serological survey of Prospect Hill virus infection in indigenous wild rodents in the USA. Trans R Soc Trop Med Hyg 81:42–45
Yates TL, Mills JN, Parmenter CA, Ksiazek TG, Parmenter RR, Vande Castle JR, Calisher CH, Nichol ST, Abbott KD, Young JC, Morrison ML, Beaty BJ, Dunnum JL, Baker RJ, Salazar-Bravo J, Peters CJ (2002) The ecology and evolutionary history of an emergent disease: Hantavirus pulmonary syndrome. Bioscience 52:989–998
Acknowledgments
We thank the private ranch owner at Cascade for allowing us access to his property. Numerous individuals provided valuable assistance in the field including K. Coffin, R. Van Horn, C. Rognli, T. Wilson, W. Semmens, K. Hughes, A. Skypala, D. Waltee, B. Lonner, J. Wilson, A. Leary, A. Alvarado, J. Bertoglio and F. Arneson. K. Wagoner provided database support, encouragement and general advice. R. Ostfeld and two anonymous reviewers provided constructive feedback, which greatly improved this paper. Financial support was provided by NIH grant P20 RR16455-06-07,08 from the INBRE-BRIN program of the National Center for Research Resources and the U.S. Centers for Disease Control and Prevention, Atlanta, GA, through cooperative agreement. The authors declare that the study described herein complies with the laws of the United States of America. The findings and conclusions presented here are those of the author(s) and do not necessarily represent the views of the funding agencies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Herwig Leirs.
Rights and permissions
About this article
Cite this article
Carver, S., Kuenzi, A., Bagamian, K.H. et al. A temporal dilution effect: hantavirus infection in deer mice and the intermittent presence of voles in Montana. Oecologia 166, 713–721 (2011). https://doi.org/10.1007/s00442-010-1882-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-010-1882-z