Abstract
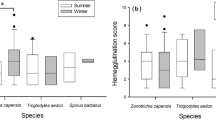
It is widely accepted that stable isotope ratios in inert tissues such as feather keratin reflect the dietary isotopic signature at the time of the tissue synthesis. However, some elements such as stable nitrogen isotopes can be affected by individual physiological state and nutritional stress. Using malaria infection experiment protocols, we estimated the possible effect of malaria parasite infections on feather carbon (δ13C) and nitrogen (δ15N) isotope signatures in juvenile common crossbills Loxia curvirostra. The birds were experimentally infected with Plasmodium relictum (lineage SGS1) and P. ashfordi (GRW2), two widespread parasites of passerines. Experimental birds developed heavy parasitemia of both parasites and maintained high levels throughout the experiment (33 days). We found no significant difference between experimental and control birds in both δ13C and δ15N values of feathers re-grown. The study shows that even heavy primary infections of malaria parasites do not affect feather δ13C and δ15N isotopic signatures. The results of this experiment demonstrate that feather isotope values of wild-caught birds accurately reflect the dietary isotopic sources at the time of tissue synthesis even when the animal’s immune system might be challenged due to parasitic infection.


Similar content being viewed by others
References
Atkinson CT, Dusek RJ, Lease JK (2001) Serological responses and immunity to superinfection with avian malaria in experimentally-infected Hawaii Amakihi. J Wildl Dis 37:20–27
Boag BR, Neilson D, Robinson CM, Csrimgeou R, Handley LL (1998) Wild rabbit host and some parasites show trophic level relationships for a first report. Isot Environ Health Stud 34:81–85
Coop RL, Holmes PH (1996) Nutrition and parasite interaction. Int J Parasitol 26:951–962
Doucett RR, Giberson DJ, Power G (1999) Parasitic association of Nanocladius (Diptera: Chironomidae) and Pteronarcys biloba (Plecoptera: Pteronarcyidae): insights from stable-isotope analysis. J North Am Benthol Soc 18:514–523
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for social, behavioral, and biometrical sciences. Behav Res Meth 39:175–191
Garnham PCC (1966) Malaria parasites and other Haemosporidia. Blackwell, Oxford
Hellgren O, Waldenström J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802
Hobson KA (1999) Tracing origins and migration of wildlife using stable isotopes: a review. Oecologia 120:314–326
Hobson KA (2003) Making migratory connections with stable isotopes. In: Berthold P, Gwinner E, Sonnenschein E (eds) Avian migration. Springer, Berlin, pp 379–391
Kelly JF (2000) Stable isotopes of carbon and nitrogen in the study of avian and mammalian trophic ecology. Can J Zool 78:1–27
Kempster B, Zanette L, Longstaffe FJ, MacDougal-Shackleton SA, Wingfield JC, Clinchy M (2007) Do stable isotopes reflect nutritional stress? results from a laboratory experiment on song sparrows. Oecologia 151:365–371
Lajtha K, Michener RH (1994) Stable isotopes in ecology and environmental science. Blackwell, Oxford
Minagawa M, Wada E (1984) Stepwise enrichment of 15N along food chains: further evidence and the relation between 15N and animal age. Geochim Cosmochim Acta 48:1135–1140
Miura O, Kuris AM, Torchin ME, Hechinger RF, Chiba S (2006) Parasites alter host phenotype and may create a new ecological niche for snail hosts. Proc R Soc Lond B 273:1323–1328
Moore J (1995) The behavior of parasitized animals. Bioscience 45:89–96
Moret Y, Schmid-Hempel P (2000) Survival for immunity: the price of immune system activation for bumblebee workers. Science 290:1166–1168
Newton I (2008) The migration ecology of birds. Academic, London
O’Grady SP, Dearing MD (2006) Isotopic insight into host- endosymbiont relationships in liolaemid lizards. Oecologia 150:355–361
Palinauskas V, Valkiūnas G, Bolshakov CV, Bensch S (2008) Plasmodium relictum (lineage P-SGS1): effects on experimentally infected passerine birds. Exp Parasitol 120:372–380
Palinauskas V, Valkiūnas G, Bolshakov CV, Bensch S (2011) Plasmodium relictum (lineage SGS1) and Plasmodium ashfordi (lineage GRW2): the effects of the co-infections on experimentally infected passerine birds. Exp Parasitol 127:527–533
Pearson SF, Levey DJ, Greenberg CH, Martínez del Rio C (2003) Effects of elemental composition on the incorporation of dietary nitrogen and carbon isotopic signatures in an omnivorous songbird. Oecologia 135:516–523
Power M, Klein GM (2004) Fish host–cestode parasite stable isotope enrichment patterns in marine, estuarine and freshwater fishes from northern Canada. Isot Environ Health S 40:257–266
Rubenstein DR, Hobson KA (2004) From birds to butterflies: animal movement patterns and stable isotopes. Trends Ecol Evol 19:256–263
Sorensen RE, Minchella DJ (1998) Parasite influences on host life history: Echinostoma revolutum parasitism of Lymnaea elodes snails. Oecologia 115:188–195
Thompson SN (1990) Physiological alterations during parasitism and their effects on host behaviour. In: Barnard J, Behnke JM (eds) Parasitism and host behavior. Taylor and Francis, London, pp 64–94
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC Press, Boca Raton
Valkiūnas G, Iezhova TA, Krizanauskiene A, Palinauskas V, Sehgal RNM, Bensch S (2008) A comparative analysis of microscopy and PCR-based detection methods for blood parasites. J Parasitol 94:1395–1401
Voigt CC, Matt F (2004) Nitrogen stress causes unpredictable enrichments of 15N in two bat species. J Exp Biol 207:1741–1748
Webster MS, Marra PP (2005) The importance of understanding migratory connectivity and seasonal interactions. In: Greenberg R, Marra PP (eds) Birds of Two Worlds: the ecology and evolution of migration. John Hopkins University Press, Baltimore, pp 199–209
Webster MS, Marra PP, Haig SM, Bensch S, Holmes RT (2002) Links between worlds; unraveling migratory connectivity. Trends Ecol Evol 17:76–83
Yohannes E, Hansson B, Lee RW, Waldenström J, Westerdahl H, Åkesson M, Hasselquist D, Bensch S (2008) Isotope signatures in winter moulted feathers predict malaria prevalence in a breeding avian host. Oecologia 158:299–306
Zehtindjiev P, Ilieva M, Westerdahl H, Hansson B, Valkiūnas G, Bensch S (2008) Dynamics of parasitemia of malaria parasites in a naturally and experimentally infected migratory songbird, the great reed warbler Acrocephalus arundinaceus. Exp Parasitol 119:99–110
Acknowledgments
The present study was supported by the Swedish Research Council (621–2007–5193), the Lithuanian State Science and Studies Foundation, NSF Grant DBI-011620 and Max-Planck Society. The experiments described herein comply with the current laws of Sweden, Russia and Lithuania. The authors are grateful to the staff of the Biological Station “Rybachy” of the Zoological Institute of the Russian Academy of Sciences for assistance in the field. We thank Scott McWilliams, Petr Prochazka and an anonymous reviewer for valuable comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Scott McWilliams.
Rights and permissions
About this article
Cite this article
Yohannes, E., Palinauskas, V., Valkiūnas, G. et al. Does avian malaria infection affect feather stable isotope signatures?. Oecologia 167, 937–942 (2011). https://doi.org/10.1007/s00442-011-2041-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-011-2041-x