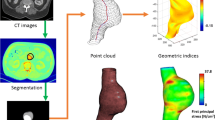
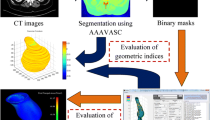
Abstract
An abdominal aortic aneurysm (AAA) is a balloon-like dilation of the aorta, which is potentially fatal in case of rupture. Computational finite element (FE) analysis is a promising approach to a more accurate and patient-specific rupture risk prediction. AAA wall strength and rupture potential index (RPI) calculation are implemented in our FE software. Static structural FE simulations are performed on n = 30 non-ruptured asymptomatic, n = 9 non-ruptured symptomatic, and n = 14 ruptured AAAs. We calculate maximum values for diameter, wall displacement, strain, stress, and RPI as well as minimum wall strength for every AAA. All investigated quantities, except minimum strength, show statistically significant differences between non-ruptured asymptomatic and symptomatic/ruptured AAAs. Maximum wall stress and especially the RPI are notably increased for symptomatic and ruptured AAAs. The biggest difference is found to be the RPI (Δ = 44.9%, p = 8.0e−5). Lowest RPI obtained for symptomatic or ruptured AAAs is 0.3. The RPI of more than 55% of the investigated asymptomatic AAAs falls below this value. Maximum wall stress and maximum RPI criteria enable a reliable rupture risk evaluation for AAAs. Especially in the diameter range where surgical indication is not obvious, the RPI holds great potential for improvement of clinical decisions.







Similar content being viewed by others
References
Ashton, J. H., J. P. Vande Geest, B. R. Simon, and D. G. Haskett. Compressive mechanical properties of the intraluminal thrombus in abdominal aortic aneurysms and fibrin-based thrombus mimics. J. Biomech. 42(3):197–201, 2009.
Cosford, P. A., and G. C. Leng. Screening for abdominal aortic aneurysm. Cochrane Database of Systematic Reviews: Reviews 2007(2), 2007.
Darling, R. C., C. R. Messina, D. C. Brewster, and L. W. Ottinger. Autopsy study of unoperated abdominal aortic aneurysms. Circulation 54:161–164, 1977.
Di Martino, E., S. Mantero, F. Inzoli, G. Melissano, D. Astore, R. Chiesa, and R. Fumero. Biomechanics of abdominal aortic aneurysm in the presence of endoluminal thrombus: Experimental characterisation and structural static computational analysis. Eur. J. Vasc. Endovasc. Surg. 15(4):290–299, 1998.
Doyle, B. J., A. Callanan, P. E. Burke, P. A. Grace, M. T. Walsh, D. A. Vorp, and T. M. McGloughlin. Vessel asymmetry as an additional diagnostic tool in the assessment of abdominal aortic aneurysms. J. Vasc. Surg. 49(2):443–454, 2009.
Fillinger, M. F., S. P. Marra, M. L. Raghavan, and F. E. Kennedy. Prediction of rupture risk in abdominal aortic aneurysm during observation: wall stress versus diameter. J. Vasc. Surg. 37(4):724–732, 2003.
Gasser, T. C., M. Auer, F. Labruto, J. Roy, and J. Swedenborg. Using finite element analysis to assess rupture risk in abdominal aortic aneurysms including the effect of the intraluminal thrombus. J. Vasc. Surg. 49(5):S29, 2009.
Gasser, T. C., G. Görgülü, M. Folkesson, and J. Swedenborg. Failure properties of intraluminal thrombus in abdominal aortic aneurysm under static and pulsating mechanical loads. J. Vasc. Surg. 48(1):179–188, 2008.
Gee, M. W., C. Förster, and W. A. Wall. A computational strategy for prestressing patient specific biomechanical problems under finite deformation. Int. J. Numer. Methods Biomed. Eng. 26(1):52–72, 2010.
Gee, M. W., U. Küttler, and W. A. Wall. Truly monolithic algebraic multigrid for fluid-structure interaction. Int. J. Numer. Methods Biomed. Eng. 2009 (submitted).
Gee, M. W., C. Reeps, H.-H. Eckstein, and W. A. Wall. Prestressing in finite deformation abdominal aortic aneurysm simulation. J. Biomech. 42(11):1732–1739, 2009.
Greenhalgh, R. M. Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysm (evar trial 1), 30-day operative mortality results: randomised controlled trial. Lancet 364(9437):843–848, 2004.
Heider, P., O. Wolf, C. Reeps, M. Hanke, A. Zimmermann, H. Berger, and H.-H. Eckstein. Aneurysmen und Dissektionen der thorakalen und abdominellen Aorta. Der Chirurg 78(7):600–610, 2007.
Humphrey, J. D., and C. A. Taylor. Intracranial and abdominal aortic aneurysms: Similarities, differences, and need for a new class of computational models. Annu. Rev. Biomed. Eng. 10(1):221–246, 2008.
Kazi, M., J. Thyberg, P. Religa, J. Roy, P. Eriksson, U. Hedin, and J. Swedenborg. Influence of intraluminal thrombus on structural and cellular composition of abdominal aortic aneurysm wall. J. Vasc. Surg. 38(6):1283–1292, 2003.
Leung, J., A. Wright, N. Cheshire, J. Crane, S. Thom, A. Hughes, and Y. Xu. Fluid structure interaction of patient specific abdominal aortic aneurysms: a comparison with solid stress models. BioMed. Eng. OnLine 5(1):33, 2006.
Li, Z.-Y., J. U-King-Im, T. Y. Tang, E. Soh, T. C. See, and J. H. Gillard. Impact of calcification and intraluminal thrombus on the computed wall stresses of abdominal aortic aneurysm. J. Vasc. Surg. 47(5):928–935, 2008.
Lu, J., X. Zhou, and M. Raghavan. Inverse method of stress analysis for cerebral aneurysms. J. Biomech. Model. Mechanobiol. 7:477–486, 2008.
Lu, J., X. Zhou, and M. L. Raghavan. Inverse elastostatic stress analysis in pre-deformed biological structures: demonstration using abdominal aortic aneurysms. J. Biomech. 40(3):693–696, 2007.
Maier, A., M. W. Gee, C. Reeps, H.-H. Eckstein, and W. A. Wall. Impact of model complexity on patient specific wall stress analyses of abdominal aortic aneurysms. In: IFMBE Proceedings of World Congress on Medical Physics and Biomedical Engineering, September 7–12, 2009, Munich, Germany, vol. 25/IV, edited by O. Dössel, and W. C. Schlegel. Springer, 2009, pp. 510–513.
Maier, A., M. W. Gee, C. Reeps, H.-H. Eckstein, and W. A. Wall. Impact of calcifications on patient-specific wall stress analysis of abdominal aortic aneurysms. Biomech. Model. Mechanobiol. doi:10.1007/s10237-010-0191-0, 2010 (accepted).
Müller, M. Chirurgie für Studium un Praxis, chapter Gefäßchirurgie - arterielle Aneurysmen. Breisach: Medizinische Verlags- und Informationsdienste, 2004/05, pp. 58–61.
Ockert, S., D. Boeckler, J. Allenberg, and H. Schumacher. Rupturiertes abdominelles Aortenaneurysma. Gefaesschirurgie 12(5):379–391, 2007.
R Development Core Team. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, 2008.
Raghavan, M. L., J. Kratzberg, E. M. Castro de Tolosa, M. M. Hanaoka, P. Walker, and E. S. da Silva. Regional distribution of wall thickness and failure properties of human abdominal aortic aneurysm. J. Biomech. 39(16):3010–3016, 2006.
Raghavan, M. L., and D. A. Vorp. Toward a biomechanical tool to evaluate rupture potential of abdominal aortic aneurysm: identification of a finite strain constitutive model and evaluation of its applicability. J. Biomech. 33(4):475–482, 2000.
Raghavan, M. L., M. W. Webster, and D. A. Vorp. Ex vivo biomechanical behavior of abdominal aortic aneurysm: Assessment using a new mathematical model. Ann. Biomed. Eng. 24(5):573–582, 1996.
Reeps, C., M. Gee, A. Maier, M. Gurdan, H.-H. Eckstein, and W. A. Wall. The impact of model assumptions on results of computational mechanics in abdominal aortic aneurysm. J. Vasc. Surg. 51(3):679–688, 2010.
Reeps, C., M. W. Gee, A. Maier, J. Pelisek, M. Gurdan, W. A. Wall, J. Mariss, H.-H. Eckstein, and M. Essler. Glucose metabolism in the vessel wall correlates with mechanical instability and inflammatory changes in a patient with a growing aneurysm of the abdominal aorta. Circ. Cardiovasc. Imaging 2(6):507–509, 2009.
Reeps, C., A. Maier, M. Gee, M. Baust, A. Zimmermann, S. Ockert, J. Pongratz, N. Navab, W. Wall, H.-H. Eckstein, and M. Essler. Correlation of biomechanics to tissue reaction in aortic aneurysm assessed by finite elements and 18F-fluorodeoxyglucose–pet/ct. Biomech. Model. Mechanobiol. 2010 (submitted).
Rodriguez, J. F., C. Ruiz, M. Doblare, and G. A. Holzapfel. Mechanical stresses in abdominal aortic aneurysms: influence of diameter, asymmetry, and material anisotropy. J. Biomech. Eng. 130(2):021023-1–021023-10, 2008.
Sakalihasan, N., and J. B. Michel. Functional imaging of atherosclerosis to advance vascular biology. Eur. J. Vasc. Endovasc. Surg. 37(6):728–734, 2009.
Speelman, L., A. Bohra, E. M. H. Bosboom, G. W. H. Schurink, F. N. van de Vosse, M. S. Makaroun, and D. A. Vorp. Effects of wall calcifications in patient-specific wall stress analyses of abdominal aortic aneurysms. J. Biomech. Eng. 129(1):105–109, 2007.
Speelman, L., E. Bosboom, G. Schurink, J. Buth, M. Breeuwer, M. Jacobs, and F. van de Vosse. Initial stress and nonlinear material behavior in patient-specific AAA wall stress analysis. J. Biomech. 42(11):1713–1719, 2009.
Speelman, L., E. M. H. Bosboom, G. W. H. Schurink, F. A. M. V. I. Hellenthal, J. Buth, M. Breeuwer, M. J. Jacobs, and F. N. van de Vosse. Patient-specific aaa wall stress analysis: 99-percentile versus peak stress. Eur. J. Vasc. Endovasc. Surg. 36(6):668–676, 2008.
Truijers, M., J. A. Pol, L. J. SchultzeKool, S. M. van Sterkenburg, M. F. Fillinger, and J. D. Blankensteijn. Wall stress analysis in small asymptomatic, symptomatic and ruptured abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 33(4):401–407, 2007.
Vande Geest, J., E. Di Martino, A. Bohra, M. Makaroun, and D. Vorp. A biomechanics-based rupture potential index for abdominal aortic aneurysm risk assessment. Ann. N. Y. Acad. Sci. 1085:11–21, 2006.
Vande Geest, J. P., M. S. Sacks, and D. A. Vorp. The effects of aneurysm on the biaxial mechanical behavior of human abdominal aorta. J. Biomech. 39(7):1324–1334, 2006.
Vande Geest, J. P., M. S. Sacks, and D. A. Vorp. A planar biaxial constitutive relation for the luminal layer of intra-luminal thrombus in abdominal aortic aneurysms. J. Biomech. 39(13):2347–2354, 2006.
Vande Geest, J. P., D. Schmidt, M. Sacks, and D. A. Vorp. The effects of anisotropy on the stress analyses of patient-specific abdominal aortic aneurysms. Ann. of Biomed. Eng. 36(6):921–932, 2008.
Vande Geest, J. P., D. H. J. Wang, S. Wisniewski, M. Makaroun, and D. Vorp. Towards a noninvasive method for determination of patient-specific wall strength distribution in abdominal aortic aneurysms. Ann. Biomed. Eng. 34(7):1098–1106, 2006.
Vorp, D. A. Biomechanics of abdominal aortic aneurysm. J. Biomech. 40(9):1887–1902, 2007.
Vorp, D. A., P. C. Lee, D. H. J. Wang, M. S. Makaroun, E. M. Nemoto, S. Ogawa, and M. W. Webster. Association of intraluminal thrombus in abdominal aortic aneurysm with local hypoxia and wall weakening. J. Vasc. Surg. 34(2):291–299, 2001.
Vorp, D. A., S. Shah, and M. S. Makaroun. Changes of biomechanics–based indices for patient–specific abdominal aortic aneurysms over time. In: Proceedings of the ASME 2009 Summer Bioengineering Conference. 2009.
Wall, W. A., and M. W. Gee. Baci: a parallel multiphysics finite element environment. Technical report, Institute for Computational Mechanics, Technische Universität München, 2010.
Wang, D. H. J., M. S. Makaroun, M. W. Webster, and D. A. Vorp. Effect of intraluminal thrombus on wall stress in patient-specific models of abdominal aortic aneurysm. J. Vasc. Surg. 36(3):598–604, 2002.
Acknowledgments
The authors gratefully acknowledge the support through the International Graduate School of Science and Engineering (IGSSE) of the Technische Universität München, Germany under projects 2–11 and 3–07.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Jane Grande-Allen oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Maier, A., Gee, M.W., Reeps, C. et al. A Comparison of Diameter, Wall Stress, and Rupture Potential Index for Abdominal Aortic Aneurysm Rupture Risk Prediction. Ann Biomed Eng 38, 3124–3134 (2010). https://doi.org/10.1007/s10439-010-0067-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-010-0067-6