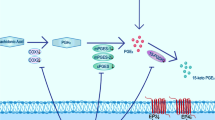
Abstract
Growing lines of evidence suggests that high mobility group box-1 (HMGB1) plays an important role for promoting inflammation and apoptosis in brain ischemia. Previously, we demonstrated that inducers of heme oxygenase-1 (HO-1) significantly reduce HMGB1 release in inflammatory conditions in vitro and in vivo. Thus, we tested our hypothesis that higenamine protects brain injury by inhibition of middle cerebral artery occlusion (MCAO)-mediated HMGB1 release in vivo, and glucose/glucose oxidase (GOX)-induced apoptosis in C6 cells in vitro due to HO-1 induction. Higenamine increased HO-1 expression in C6 cells in both hypoxia and normoxia, in which the former was much more significant than the latter. Higenamine increased Nrf-2 luciferase activity, translocated Nrf-2 to nucleus, and increased phosphorylation of Akt in C6 cells. Consistent with this, LY 294002, a PI3K inhibitor, inhibited HO-1 induction by higenamine and apoptosis induced by glucose/GOX in C6 cells was prevented by higenamine, which effect was reversed by LY 294002. Importantly, administration of higenamine (i.p) significantly reduced brain infarct size, mortality rate, MPO activity and tissue expression of HMGB1 in MCAO rats. In addition, recombinant high mobility group box 1 induced apoptosis in C6 cells by increasing ratio of Bax/bcl-2 and cleaved caspase c, which was inhibited by higenamine, and all of these effects were reversed by co-treatment with ZnPPIX. Therefore, we conclude that higenamine, at least in part, protects brain cells against hypoxic damages by up-regulation of HO-1. Thus, higenamine may be beneficial for the use of ischemic injuries such as stroke.








Similar content being viewed by others
Abbreviations
- ARE:
-
Antioxidant response element
- GFAP:
-
Glial fibrillary acidic protein
- HMGB1:
-
High mobility group box-1
- HO-1:
-
Heme oxygenase-1
- I/R:
-
Ischemia and reperfusion
- LPS:
-
Lipopolysaccharide
- MCAO:
-
Middle cerebral artery occlusion
- MPO:
-
Myeloperoxidase
- Nrf2:
-
Nuclear factor erythroid 2-related factor
- PCNA:
-
Proliferating cell nuclear antigen
- PI3K:
-
Phosphoinositol-3-kinase
- PKA:
-
Protein kinase A
- ROS:
-
Reactive oxygen species
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP-mediated nicked end labeling
- ZnppIX:
-
Zinc protoporphyrin IX
References
Heneka MT, Sharp A, Murphy P, Lyons JA, Dumitrescu L et al (2001) The heat shock response reduces myelin oligodendrocyte glycoprotein-induced experimental autoimmune encephalomyelitis in mice. J Neurochem 77:568–579
Teismann P, Schulz JB (2004) Cellular pathology of Parkinson’s disease: astrocytes, microglia and inflammation. Cell Tissue Res 318:149–161
Perry VH, Nicoll JA, Holmes C (2010) Microglia in neurodegenerative disease. Nat Rev Neurol 6:193–201
Chandel NS, Maltepe E, Goldwasser E, Mathieu CE, Simon MC et al (1998) Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA 95:11715–11720
Chandel NS, Vander Heiden MG, Thompson CB, Schumacker PT (2000) Redox regulation of p53 during hypoxia. Oncogene 19:3840–3848
Bazan NG, Palacios-Pelaez R, Lukiw WJ (2002) Hypoxia signaling to genes: significance in Alzheimer’s disease. Mol Neurobiol 26:283–298
Qiu J, Nishimura M, Wang Y, Sims JR, Qiu S et al (2008) Early release of HMGB-1 from neurons after the onset of brain ischemia. J Cereb Blood Flow Metab 28:927–938
Lee YS, Kang YJ, Kim HJ, Park MK, Seo HG et al (2006) Higenamine reduces apoptotic cell death by induction of heme oxygenase-1 in rat myocardial ischemia-reperfusion injury. Apoptosis 11:1091–1100
Park MK, Kang YJ, Lee HS, Kim HJ, Seo HG et al (2008) The obligatory role of COX-2 expression for induction of HO-1 in ischemic preconditioned rat brain. Biochem Biophys Res Commun 377:1191–1194
Maines MD (1997) The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol 37:517–554
Abraham NG, Tsenovoy PL, McClung J, Drummond GS (2008) Heme oxygenase: a target gene for anti-diabetic and obesity. Curr Pharm Des 14:412–421
Keyse SM, Tyrrell RM (1989) Heme oxygenase is the major 32-kDa stress protein induced in human skin fibroblasts by UVA radiation, hydrogen peroxide, and sodium arsenite. Proc Natl Acad Sci USA 86:99–103
Ryter SW, Alam J, Choi AM (2006) Heme oxygenase-1/carbon monoxide: from basic science to therapeutic applications. Physiol Rev 86:583–650
Park JE, Kang YJ, Park MK, Lee YS, Kim HJ et al (2006) Enantiomers of higenamine inhibit LPS-induced iNOS in a macrophage cell line and improve the survival of mice with experimental endotoxemia. Int Immunopharmacol 6:226–233
Geremia E, Baratta D, Zafarana S, Giordano R, Pinizzotto MR et al (1990) Antioxidant enzymatic systems in neuronal and glial cell-enriched fractions of rat brain during aging. Neurochem Res 15:719–723
Claes P, Van Kolen K, Roymans D, Blero D, Vissenberg K et al (2004) Reactive blue 2 inhibition of cyclic AMP-dependent differentiation of rat C6 glioma cells by purinergic receptor-independent inactivation of phosphatidylinositol 3-kinase. Biochem Pharmacol 67:1489–1498
Pita I, Jelaso AM, Ide CF (1999) IL-1beta increases intracellular calcium through an IL-1 type 1 receptor mediated mechanism in C6 astrocytic cells. Int J Dev Neurosci 17:813–820
Mead C, Pentreath VW (1998) Evaluation of toxicity indicators in rat primary astrocytes, C6 glioma and human 1321N1 astrocytoma cells: can gliotoxicity be distinguished from cytotoxicity? Arch Toxicol 72:372–380
Tsoyi K, Jang HJ, Nizamutdinova IT, Kim YM, Lee YS et al (2011) Metformin inhibits HMGB1 release in LPS-treated RAW 264.7 cells and increases survival rate of endotoxaemic mice. Br J Pharmacol 162:1498–1508
Park MK, Kim CH, Kim YM, Kang YJ, Kim HJ et al (2007) Akt-dependent heme oxygenase-1 induction by NS-398 in C6 glial cells: a potential role for CO in prevention of oxidative damage from hypoxia. Neuropharmacology 53:542–551
Park MK, Kang YJ, Ha YM, Jeong JJ, Kim HJ et al (2009) EP2 receptor activation by prostaglandin E2 leads to induction of HO-1 via PKA and PI3K pathways in C6 cells. Biochem Biophys Res Commun 379:1043–1047
Hamdulay SS, Wang B, Birdsey GM, Ali F, Dumont O et al (2010) Celecoxib activates PI-3K/Akt and mitochondrial redox signaling to enhance heme oxygenase-1-mediated anti-inflammatory activity in vascular endothelium. Free Radic Biol Med 48:1013–1023
Liu K, Mori S, Takahashi HK, Tomono Y, Wake H et al (2007) Anti-high mobility group box 1 monoclonal antibody ameliorates brain infarction induced by transient ischemia in rats. FASEB J 21:3904–3916
Kim JB, Lim CM, Yu YM, Lee JK (2008) Induction and subcellular localization of high-mobility group box-1 (HMGB1) in the postischemic rat brain. J Neurosci Res 86:1125–1131
Lee GH, Kim HK, Chae SW, Kim DS, Ha KC et al (2007) Bax inhibitor-1 regulates endoplasmic reticulum stress-associated reactive oxygen species and heme oxygenase-1 expression. J Biol Chem 282:21618–21628
Lee PJ, Jiang BH, Chin BY, Iyer NV, Alam J et al (1997) Hypoxia-inducible factor-1 mediates transcriptional activation of the heme oxygenase-1 gene in response to hypoxia. J Biol Chem 272:5375–5381
Li RC, Guo SZ, Lee SK, Gozal D (2010) Neuroglobin protects neurons against oxidative stress in global ischemia. J Cereb Blood Flow Metab 30:1874–1882
Noshita N, Lewen A, Sugawara T, Chan PH (2001) Evidence of phosphorylation of Akt and neuronal survival after transient focal cerebral ischemia in mice. J Cereb Blood Flow Metab 21:1442–1450
Wang JK, Yu LN, Zhang FJ, Yang MJ, Yu J et al (2010) Postconditioning with sevoflurane protects against focal cerebral ischemia and reperfusion injury via PI3K/Akt pathway. Brain Res 1357:142–151
Chang KC, Chong WS, Lee IJ (1994) Different pharmacological characteristics of structurally similar benzylisoquinoline analogs, papaverine, higenamine, and GS 389, on isolated rat aorta and heart. Can J Physiol Pharmacol 72:327–334
Zhang JJ, Chen WW (1985) Protection of the synovial fluid by higenamine. Yao Xue Xue Bao 20:423–426
D’Agnillo F, Alayash AI (2002) A role for the myoglobin redox cycle in the induction of endothelial cell apoptosis. Free Radic Biol Med 33:1153–1164
Kikuchi K, Kawahara K, Biswas KK, Ito T, Tancharoen S et al (2009) Minocycline attenuates both OGD-induced HMGB1 release and HMGB1-induced cell death in ischemic neuronal injury in PC12 cells. Biochem Biophys Res Commun 385:132–136
Jin YC, Lee YS, Kim YM, Seo HG, Lee JH et al (2009) (S)-1-(alpha-naphthylmethyl)-6,7-dihydroxy-1,2,3,4-tetrahydroisoquinoline (CKD712) reduces rat myocardial apoptosis against ischemia and reperfusion injury by activation of phosphatidylinositol 3-kinase/Akt signaling and anti-inflammatory action in vivo. J Pharmacol Exp Ther 330:440–448
Tsoyi K, Lee TY, Lee YS, Kim HJ, Seo HG et al (2009) Heme-oxygenase-1 induction and carbon monoxide-releasing molecule inhibit lipopolysaccharide (LPS)-induced high-mobility group box 1 release in vitro and improve survival of mice in LPS- and cecal ligation and puncture-induced sepsis model in vivo. Mol Pharmacol 76:173–182
Takamiya R, Hung CC, Hall SR, Fukunaga K, Nagaishi T et al (2009) High-mobility group box 1 contributes to lethality of endotoxemia in heme oxygenase-1-deficient mice. Am J Respir Cell Mol Biol 41:129–135
Pyo MK, Kim JM, Jin JL, Chang KC, Lee DH et al (2007) Effects of higenamine and its 1-naphthyl analogs, YS-49 and YS-51, on platelet TXA2 synthesis and aggregation. Thromb Res 120:81–86
Yun-Choi HS, Pyo MK, Park KM, Chang KC, Lee DH (2001) Anti-thrombotic effects of higenamine. Planta Med 67:619–622
Acknowledgment
This work was supported by grant from NRF (03-2010-0298) and MRC program from MOST/KOSEF (R13-2005-012-01003-0).
Conflict of interest
There is no report to declare the conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yu Mi Ha and Min Young Kim have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ha, Y.M., Kim, M.Y., Park, M.K. et al. Higenamine reduces HMGB1 during hypoxia-induced brain injury by induction of heme oxygenase-1 through PI3K/Akt/Nrf-2 signal pathways. Apoptosis 17, 463–474 (2012). https://doi.org/10.1007/s10495-011-0688-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0688-8