1. A recently recognized complication of uncontrolled diabetes mellitus is the encephalopathy involving, among other regions, the hippocampus. Since estrogens bring neuroprotection in cases of brain injury and degenerative diseases, we have studied if estradiol (E2) administration counteracts some hippocampal abnormalities of streptozotocin (STZ)-diabetic adult mice.
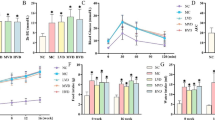
2. We first report the ability of E2 to modulate neurogenesis in the dentate gyrus (DG) and subventricular zone (SVZ) of diabetic mice. Using bromodeoxyuridine (BrdU) to label newly generated cells, a strong reduction in cell proliferation was obtained in DG and SVZ of mice sacrificed 20 days after STZ administration. The reduction was completely relieved by 10 days of E2 pellet implantation, which increased 30-fold the circulating E2 levels.
3. Diabetic mice also showed abnormal expression of astrocyte markers in hippocampus. Thus, increased number of GFAP+ cells, indicative of astrogliosis, and increased number of apolipoprotein-E (Apo-E)+ astrocytes, a marker of ongoing neuronal dysfunction, was found in stratum radiatum below the CA1 hippocampal subfield of diabetic mice. Both parameters were reverted to normal by the E2 regime that upregulated cell proliferation.
4. The studies demonstrated that hippocampal neuropathology of uncontrolled diabetes is a reversible condition and sensitive to estrogen treatment. Studies in animal models may open up new venues for understanding the beneficial role of steroid hormones in diabetic encephalopathy.




Similar content being viewed by others
REFERENCES
Adeghate, E., and Ponery, A. S. (2001). The effect of 17 beta-estradiol on weight, blood glucose and plasma insulin levels in diabetic rats. Gynecol. Endocrinol. 15:433–438.
Anderson, M. F., Aberg, M. A. I., Nilsson, M., and Eriksson, P. S. (2002). Insulin-like growth factor-I and neurogenesis in the adult mammalian brain. Dev. Brain Res. 4:115–122.
Artola, A., Kamal, A., Ramakers, G. M., Gardoni, F., DiLuca, M., Biessels, G. J., Cattabeni, F., and Gispen, W. H. (2002). Synaptic plasticity in the diabetic brain: Advanced aging? Prog. Brain Res. 138:305–314.
Behl, C. (2002). Estrogen as a neuroprotective hormone. Nat. Rev. Neurosci. 3:433–442.
Bestetti, G., and Rossi, G. L. (1980). Hypothalamic lesions in rats with long-term streptozotocin-induced diabetes mellitus. Acta Neurophatol. 52:119–127.
Biessels, G. J., van der Heide, L. P., Kamal, A., Bleys, R. L., and Gispen, W. H. (2002). Ageing and diabetes: Implications for brain function. Eur. J. Pharmacol. 441:1–14.
Boschert, U., Merlo-Pich, E., Higgins, G., Roses, A. D., and Catsicas, S. (1999). Apolipoprotein E expression by neurons surviving excitotoxic stress. Neurobiol. Dis. 6:508–514.
Brannvall, K., Korthonen, L., and Lindholm, D. (2002). Estrogen-receptor dependent regulation of neural stem cell proliferation and differentiation. Mol. Cell. Neurosci. 21:512–520.
Cardona-Gomez, G. P., Mendez, P., DonCarlos, L., Azcoitia, I., and García Segura, L. M. (2001). Interactions of oestrogens and insulin-like growth factor-I in the brain: Implications for neuroprotection. Brain Res. Rev. 37:320–334.
Catalani, A., Sabatini, M., Consoli, C., Cinque, C., Tamassoni, D., Azmitia, E., Angelucci, L., and Amenta, F. (2002). Glial fibrillary acidic protein immunoreactive astrocytes in developing rat hippocampus. Mech. Age Dev. 123:481–490.
Chabot, C. H., Massicotte, G., Milot, M., Trudeau, F., and Gagne, J. (1997). Impaired modulation of AMPA receptors by calcium-dependent processes in streptozotocin-induced diabetic rats. Brain Res. 768:249–256.
De Nicola, A. F., Magariños, A. M., and Foglia, V. G. (1991). Neuroendocrine regulation in experimental diabetes (Houssay Lecture). In Rifkin, H., Colwell, J. A., and Taylor, S. I. (eds.), Diabetes, Elsevier Science, Amsterdam pp. 3–8.
Diaz-Brinton, R., Chen, S., Montoya, M., Hsieh, D., Minaya, J., Kim, J., and Chu, H. P. (2000). The women's health initiative estrogen replacement therapy is neurotrophic and neuroprotective. Neurobiol. Aging 21:475–496.
Fagan, A. M., Murphy, B. A., Patel, S. N., Kilbridge, J. F., Mobley, W. C., Bu, G., and Holtzman, D. M. (1998). Evidence for normal aging of the septo-hippocampal cholinergic system in apoE (−/−) mice but impaired clearance of axonal degeneration products following injury. Exp. Neurol. 151:314–325.
Ferguson, S. C., Deary, I. J., Evans, J. C., Ellard, S., Hattersley, A. T., and Frier, B. M. (2003). Apolipoprotein-E influences aspects of intellectual ability in type 1 diabetes. Diabetes 52: 145–148.
Ferrini, M., Lima, A., and De Nicola, A. F. (1995). Estradiol abolishes downregulation of glucocorticoids receptor in brain. Life Sci. 57:2403–2412.
Flood, J. F., Mooradian, A. D., and Morley, J. E. (1990). Characteristics of learning and memory in streptozocin-induced diabetic mice. Diabetes 39:1391–1398.
García Segura, L. M., Azcoitia, I., and DonCarlos, L. L. (2001). Neuroprotection by estradiol. Prog. Neurobiol. 63:29–60.
Garcia Segura, L. M., Naftolin, F., Hutchinson, J. B., Azcoitia, I., and Chowen, J. A. (1999). Role of astroglia in estrogen regulation of synaptic plasticity and brain repair. J. Neurobiol. 40:574– 584.
Garris, D. R. (1999). Estrogenic stimulation of hypothalamic-limbic system metabolism in ageing diabetic C57Bl/KsJ mice. Neuroendocrinology 69:424–429.
Gispen, W. H., and Biessels, G. J. (2000). Cognition and synaptic plasticity in diabetes mellitus. Trends Neurosci. 23:542–549.
Gold, A. E., Deary, L. J., Jones, R. W., O’Hare, J. P., Reckless, J. P., and Frier, B. M. (1994). Severe deteriorations in cognitive function and personality in patients with long-standing diabetes: A complication of diabetes or a consequence of treatment? Diabetes Med. 11:499–505.
Gould, E., Tanapat, P., Rydel, T., and Hastings, N. (2000). Regulation of hippocampal neurogenesis in adulthood. Biol. Psychiatry 48:715–720.
Gould, E., Woolley, C. S., and McEwen, B. S. (1991). Naturally occurring cell death in the developing dentate gyrus of the rat. J. Comp. Neurol. 304:408–418.
Grootendorst, J., Mulder, M., Haasdijt, E., de Kloet, R. E., and Jaarsma, D. (2000). Presence of apolipoprotein E immunoreactivity in degenerating neurons of mice is dependent on the severity of kainic acid-induced lesion. Brain Res. 868:165–175.
Hastings, N. B., and Gould, E. (1999). Rapid extension of axons into the CA3 region of adult-generated granule cells. J. Comp. Neurol. 413:146–154.
Ignatius, M. J., Gebicke-Harter, P. J., Pate Skene, J. H., Schilling, J. W., Weisgraber, K. H., Mahley, R. W., and Shooter, M. E. (1986). Expression of apolipoprotein E during nerve degeneration and regeneration. Proc. Natl. Acad. Sci. 83:1125–1129.
Jackson-Guilford, J., Leander, J. D., and Nisenbaum, L. K. (2000). The effect of streptozotocin-induced diabetes on cell proliferation in the rat dentate gyrus. Neurosci. Lett. 293:91–94.
Kempermann, G., Gast, D., Kronenberg, G., Yamaguchi, M., and Gage, F. (2003). Early determination and long-term persistence of adult-generated new neurons in the hippocampus of mice. Development 130:391–399.
Kiessling, M., Auer, R. N., Kleihues, P., and Siesjo, B. K. (1986). Cerebral protein synthesis during long-term recovery from severe hypoglycemia. J. Cereb. Blood Flow Metab. 6:42–51.
Klein, J. P., and Waxman, S. G. (2003). The brain in diabetes: Molecular changes in neurons and their implications for end-organ damage. Lancet Neurol. 2:548–554.
Kuhn, H. G., Dickinson-Anson, H., and Gage, F. H. (1996). Neurogenesis in the dentate gyrus of the adult rat: Age related decrease of neuronal progenitor proliferation. J. Neurosci. 16:2027–2033.
Lambert, K. G., Gerecke, K. M., Quadros, P. S., Doudera, E., Jasnow, A. J., and Kinsley, C. H. (2000). Activity-stress increases density of GFAP-immunoreactive astrocytes in the rat hippocampus. Stress 4:275–284.
Lannert, H., Wirtz, P., Schuhmann, V., and Galmbacher, R. (1998). Effects of estradiol (17β) on learning, memory and cerebral energy metabolism in male rats after intracerebroventricular administration of streptozotocin. J. Neural Transm. 105:1045–1063.
Launer, L. J., Rodriguez, B. L., and Peila, R. (2002). Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies. Diabetes 51:1256–1262.
Lehmann, A. (1974). Atlas stereotaxique du cerveau de la souris, Editions du Centre National de la Recherche Scientifique, Paris.
Li, Z. G., Zhang, W., Grunberger, G., and Sima, A. A. F. (2002). Hippocampal neuronal apoptosis in type I diabetes. Brain Res. 946:221–231.
Liu, Z., Gastard, M., Verina, T., Bora, S., Mouton, P. R., and Koliatsos, V. E. (2001). Estrogens modulate experimentally induced apoptosis of granule cells in the adult hippocampus. J. Comp. Neurol. 441: 1–8.
Lux-Lantos, V., Hockl, O., Tesone, M., and Libertun, C. (1994). Anterior pituitary estradiol receptors associated with the reinstatement of ovulatory cycles after lactation interruption in the rat. Neuroendocrinology 59:265–270.
Magariños, A. M., and McEwen, B. S. (2000). Experimental diabetes in rats causes hippocampal dendritic and synaptic reorganization and increased glucocorticoid reactivity to stress. Proc. Natl. Acad. Sci. 97:10056–11061.
Makimattila, S., Malmberg-Ceder, K., Hakkinen, A. M., Vuori, K., Salonen, O., Summanen, P.,Yki-Jarvinen, H., Kaste, M., Heikkinen, S., Lundbom, N., and Roine, R. O. (2004). Brain metabolic alterations in patients with type 1 diabetes-hyperglycemia-induced injury. J Cereb. Blood Flow Metab. 24:1393–1399.
McCall, A. L. (1992). The impact of diabetes on the CNS. Diabetes 41:557–570.
McEwen, B. S., Akama, K., Alves, S., Brake, S. G., Bulloch, K., Lee, S., Li, C., Yuen, G., and Milner, T. A. (2001). Tracking the estrogen receptor in neurons: Implications for estrogen-induced synapse formation. Proc. Natl. Acad. Sci. 98:7093–7100.
McEwen, B. S., Magariños, A. M., and Reagan, L. P. (2002). Studies of hormone action in the hippocampal formation. Possible relevance to depression and diabetes. J. Psychsom. Res. 53:883–890.
McMahon, S. S., and McDermott, K. W. (2001). Proliferation and migration of glial precursor cells in the developing rat spinal cord. J. Neurocytol. 30:821–828.
Mukai, N., Hori, S., and Pomeroy, M. (1980). Cerebral lesions in rats with streptozotocin-induced diabetes. Acta Neuropathol. (Berl.) 51:79–84.
Muller, H. W., Matthiese, H. P., Schmalenbach, C., and Schroeder, W. O. (1991). Glial support of CNS neuronal survival, neurite growth and regeneration. Restorative Neurol. Neurosci. 2:229– 232.
Nitta, A., Murai, R., Suzuki, N., Ito, H., Nomoto, H., katoh, G., Furukawa, Y., and Furukawa, S. (2002). Diabetic neuropathies in brain are induced by a deficiency of BDNF. Neurotoxicol. Teratol. 24:695–701.
Norenberg, M. D. (1994). Astrocyte responses to CNS injury. J. Neuropathol. Exp. Neurol. 53:213–220.
Ormerod, B. K., Lee, T. T., and Galea, L. A. M. (2003). Estradiol initially enhances but subsequently suppresses (via adrenal steroids) granule cell proliferation in the dentate gyrus of adult female rats. J. Neurobiol. 55:247–260.
Perez-Martin, M., Azcoitia, I., Trejo, J. L., Sierra, A., and Garcia-Segura, L. M. (2003). An antagonist of estrogen receptors blocks the induction of adult neurogenesis by insulin-like growth factor-I in the dentate gyrus of adult female rat. Eur. J. Neurosci. 18:923–930.
Piotrowski, P., Wierzbicka, K., and Smiatek, M. (2001). Neuronal death in the rat hippocampus in experimental diabetes and cerebral ischemia treated with antioxidants. Folia Neuropathol. 39:147–154.
Poirier, J. (1994). Apolipoprotein E in animal models of CNS injury and in Alzheimer's disease. Trends Neurosci. 17:525–530.
Reagan, L. P., Magariños, A. M., and McEwen, B. S. (1999). Neurological changes induced by stress in streptozotocin diabetic rats. Ann. N.Y. Acad. Sci. 893:126–137.
Revsin, Y., Saravia, F., Roig, P., Lima, A., de Kloet, E. R., Homo-Delarche, F., and De Nicola, A. F. (2005). Neuronal and astroglial alterations in the hippocampus of a mouse model for type 1 diabetes. Brain Res. 1038:22–31.
Ristow, M. (2004). Neurodegenerative disorders associated with diabetes mellitus. J. Mol. Med. 82:510–529.
Russel, J. W., Sullivan, K. A., Windebank, A. J., Herrmann, D. N., and Feldman, E. L. (1999). Neurons undergo apoptosis in animal and cell culture models of diabetes. Neurobiol. Dis. 6:347–363.
Saravia, F., Gonzalez, S., Roig, P., Alves, V., Homo-Delarche, F., and De Nicola, A. F. (2001). Diabetes increases the expression of hypothalamic neuropeptides in a spontaneous model of type I diabetes, the nonobese diabetic (NOD) mouse. Cell Mol. Neurobiol. 21:15–27.
Saravia, F., Revsin, Y., Gonzalez Deniselle, M. C., Gonzalez, S., Roig, P., Lima, A., Homo-Delarche, F., and De Nicola, A. F. (2002). Increased astrocyte reactivity in the hippocampus of murine models of type I diabetes: The nonobese diabetic (NOD) and streptozotocin-treated mice. Brain Res. 957:345–353.
Saravia, F., Revsin, Y., Lux-Lantos, V., Beauquis, J., Homo-Delarche, F., and De Nicola, A. F. (2004). Oestradiol restores cell proliferation in dentate gryus and subventricular zone of streptozotocin-diabetic rats. J. Neuroendocrinol. 16:704–710.
Scharfman, H. E., and Maclusky, N. J. (2005). Similarities between actions of estrogen and BDNF in the hippocampus: Coincidence or clue? Trends Neurosci. 28:79–85.
Seaberg, R. M., and van der Kooy, D. (2003). Stem and progenitor cells: The premature desertion of rigorous definitions. Trends Neurosci. 26:125–131.
Shors, T. J., Townsend, D. A., Zhao, M., Kozorovitsky, Y., and Gould, E. (2002). Neurogenesis may relate to some but not all types of hippocampal-dependent learning. Hippocampus 12:578–584.
Tanapat, P., Hastings, N. B., Reeves, A. J., and Gould, E. (1999). Estrogen stimulates a transient increase in the number of new neurons in the dentate gyrus of the adult female rat. J. Neurosci. 19:5792–5801.
Taupin, P., and Gage, F. (2002). Adult neurogenesis and neural stem cells of the central nervous system in mammals. J. Neurosci. Res. 69:745–749.
Toung, T. K., Hurn, P. D., Traysman, R. J., and Sieber, F. E. (2000). Estrogen decreases infarct size after temporary focal ischemia in a genetic model of type I diabetes mellitus. Stroke 31:2701–2706.
Veinbergs, I., Jung, M. W., Young, S. J., Van Uden, E., Groves, P. M., and Masliah, E. (1998). Altered long-term potentiation in the hippocampus of apolipoprotein E-deficient mice. Neurosci. Lett. 249:71–74.
Weeber, E. J., Beffert, U., Jones, C., Christian, J. M., Forster, E., Sweat, J. D., and Herz, J. (2002). Reelin and ApoE receptors cooperate to enhance hippocampal synaptic plasticity and learning. J. Biol. Chem. 277:39944–39952.
ACKNOWLEDGMENTS
The authors acknowledge the technical assistance of Analia Lima and Paulina Roig. This study was supported by an International agreement between CONICET-INSERM and the WOTRO Project (The Netherlands) and grants from the University of Buenos Aires (M022 and M094), CONICET (PIP 2007), FONCYT (PICT 05-08663), and Beca Carrillo-Oñativia 2005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saravia, F.E., Beauquis, J., Revsin, Y. et al. Hippocampal Neuropathology of Diabetes Mellitus is Relieved by Estrogen Treatment. Cell Mol Neurobiol 26, 941–955 (2006). https://doi.org/10.1007/s10571-006-9096-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-006-9096-y