Abstract
Background
Vonoprazan is a novel potassium-competitive acid blocker (P-CAB) recently approved for Helicobacter pylori eradication therapy in Japan.
Aims
To compare PPI- and P-CAP-containing triple therapy and vonoprazan-based triple therapy.
Methods
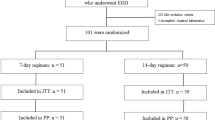
Two hundred ninety-five initial subjects received a PPI-containing triple therapy; the next 125 subjects received vonoprazan-containing triple therapy. Two sequential groups received 7-day eradication regimens consisting of amoxicillin 750 mg, clarithromycin 200 mg both twice a day with standard dose PPI or vonoprazan (20 mg) each twice daily. H. pylori eradication was confirmed by a 13C-UBT. Clarithromycin susceptibility was evaluated by 23S rRNA PCR.
Results
Population cure rates with clarithromycin susceptible strains were 89.6 versus 100 % for PPI and vonoprazan therapies, respectively. Cure rates with resistant strains were 40.2 % with PPI therapy versus 76.1 % with vonoprazan triple therapy. There was no difference in side effects.
Conclusions
Although 7-day P-CAB triple therapy was superior to 7-day PPI triple therapy, neither was highly effective, or can be recommended, in the presence of clarithromycin-resistant infections.


Similar content being viewed by others
References
Asaka M, Mabe K. Strategies for eliminating death from gastric cancer in Japan. Proc Jpn Acad Ser B Phys Biol Sci. 2014;90:251–258.
Maeda S, Yoshida H, Ogura K, Kanai F, Shiratori Y, Omata M. Helicobacter pylori specific nested PCR assay for the detection of 23S rRNA mutation associated with clarithromycin resistance. Gut. 1998;43:317–321.
Melake NA, Shaker GH, Salama MA. Incidence of Helicobacter pylori infection and their clarithromycin-resistant strains in otitis media with effusion regarding phenotypic and genotypic studies. Saudi Pharm J: SPJ.. 2012;20:345–353.
Graham DY, Fischbach L. Helicobacter pylori treatment in the era of increasing antibiotic resistance. Gut. 2010;59:1143–1153.
Kim SY, Choi DJ, Chung JW. Antibiotic treatment for Helicobacter pylori: is the end coming? World J Gastrointest Pharmacol Ther. 2015;6:183–198.
Megraud F, Benejat L, Benejat L, Ontsira Ngoyi EN, Lehours P. Molecular approaches to identify Helicobacter pylori antimicrobial resistance. Gastroenterol Clin North Am. 2015;44:577–596.
Shin JM, Inatomi N, Munson K, et al. Characterization of a novel potassium-competitive acid blocker of the gastric H,K-ATPase, 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438). J Pharmacol Exp Ther. 2011;339:412–420.
Hori Y, Imanishi A, Matsukawa J, et al. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther. 2010;335:231–238.
Matsukawa J, Hori Y, Nishida H, Kajino M, Inatomi N. A comparative study on the modes of action of TAK-438, a novel potassium-competitive acid blocker, and lansoprazole in primary cultured rabbit gastric glands. Biochem Pharmacol. 2011;81:1145–1151.
Murakami K, Sakurai Y, Shiino M, Funao N, Nishimura A, Asaka M. Vonoprazan, a novel potassium-competitive acid blocker, as a component of first-line and second-line triple therapy for Helicobacter pylori eradication: a phase III, randomised, double-blind study. Gut. 2016;65:1439–1446.
Matsumoto H, Shiotani A, Nishibayashi H, et al. Molecular detection of H. pylori using adherent gastric mucous to biopsy forceps. Helicobacter. 2016. doi:10.1111/hel.12310.
Uotani T, Graham DY. Diagnosis of Helicobacter pylori using the rapid urease test. Ann Transl Med. 2015;3:9.
Graham DY. Vonoprazan Helicobacter pylori eradication therapy: ethical and interpretation issues. Gut 2016. doi:10.1136/gutjnl-2016-311796.
Kirchheiner J, Glatt S, Fuhr U, et al. Relative potency of proton-pump inhibitors-comparison of effects on intragastric pH. Eur J Clin Pharmacol. 2009;65:19–31.
Yuan Y, Ford AC, Khan KJ, et al. Optimum duration of regimens for Helicobacter pylori eradication. Cochrane Database Syst Rev. 2013;12:Cd008337.
Nishizawa T, Maekawa T, Watanabe N, et al. Clarithromycin versus metronidazole as first-line Helicobacter pylori eradication: a multicenter, prospective, randomized controlled study in Japan. J Clin Gastroenterol. 2015;49:468–471.
Hori K, Miwa H, Matsumoto T. Efficacy of 2-week, second-line Helicobacter pylori eradication therapy using rabeprazole, amoxicillin, and metronidazole for the Japanese population. Helicobacter. 2011;16:234–240.
Savarino V, Sandro Mela G, Zentilin P, Vigneri S, Celle G. Acid inhibition and amoxicillin activity against Helicobacter pylori. Am J Gastroenterol. 1993;88:1975–1976.
Furuta T, Shirai N, Kodaira M, et al. Pharmacogenomics-based tailored versus standard therapeutic regimen for eradication of H. pylori. Clin Pharmacol Ther. 2007;81:521–528.
Marcus EA, Inatomi N, Nagami GT, Sachs G, Scott DR. The effects of varying acidity on Helicobacter pylori growth and the bactericidal efficacy of ampicillin. Aliment Pharmacol Ther. 2012;36:972–979.
Sachs G, Shin JM, Munson K, et al. Review article: the control of gastric acid and Helicobacter pylori eradication. Aliment Pharmacol Ther. 2000;14:1383–1401.
Scott D, Weeks D, Melchers K, Sachs G. The life and death of Helicobacter pylori. Gut. 1998;43:S56–S60.
Graham DY, Shiotani A. New concepts of resistance in the treatment of Helicobacter pylori infections. Nat Clin Pract Gastroenterol Hepatol. 2008;5:321–331.
Yang JC, Lin CJ, Wang HL, et al. High-dose dual therapy is superior to standard first-line or rescue therapy for Helicobacter pylori infection. Clin Gastroenterol Hepatol. 2015;13:895.e895–905.e895.
Shin JM, Sachs G. Long lasting inhibitors of the gastric H,K-ATPase. Expert Rev Clin Pharmacol. 2009;2:461–468.
Hori Y, Matsukawa J, Takeuchi T, et al. A study comparing the antisecretory effect of TAK-438, a novel potassium-competitive acid blocker, with lansoprazole in animals. J Pharmacol Exp Ther. 2011;337:797–804.
Graham DY. Hp-normogram (normo-graham) for assessing the outcome of H. pylori therapy: effect of resistance, duration, and CYP2C19 genotype. Helicobacter. 2016;21:85–90.
Dore MP, Lu H, Graham DY. Role of bismuth in improving Helicobacter pylori eradication with triple therapy. Gut. 2016;65:870–888.
Furuta T, Graham DY. Pharmacologic aspects of eradication therapy for Helicobacter pylori infection. Gastroenterol Clin North Am. 2010;39:465–480.
Miehlke S, Kirsch C, Schneider-Brachert W, et al. A prospective, randomized study of quadruple therapy and high-dose dual therapy for treatment of Helicobacter pylori resistant to both metronidazole and clarithromycin. Helicobacter. 2003;8:310–319.
Graham DY, Dore MP. Helicobacter pylori therapy: a paradigm shift. Expert Rev Anti Infect Ther. 2016;14:577–585.
Midolo PD, Turnidge JD, Munckhof WJ. Is bactericidal activity of amoxicillin against Helicobacter pylori concentration dependent? Antimicrob Agents Chemother. 1996;40:1327–1328.
Ren L, Lu H, Li HY, et al. New dual therapy for primary treatment of Helicobacter pylori infection: a prospective randomized study in Shanghai, China. J Dig Dis. 2014;15:622–627.
Yang JC, Wang HL, Chern HD, et al. Role of omeprazole dosage and cytochrome P450 2C19 genotype in patients receiving omeprazole-amoxicillin dual therapy for Helicobacter pylori eradication. Pharmacotherapy. 2011;31:227–238.
Smith SI, Fowora MA, Lesi OA, et al. Application of stool-PCR for the diagnosis of Helicobacter pylori from stool in Nigeria—a pilot study. SpringerPlus. 2012;1:78.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors state that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Matsumoto, H., Shiotani, A., Katsumata, R. et al. Helicobacter pylori Eradication with Proton Pump Inhibitors or Potassium-Competitive Acid Blockers: The Effect of Clarithromycin Resistance. Dig Dis Sci 61, 3215–3220 (2016). https://doi.org/10.1007/s10620-016-4305-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4305-0