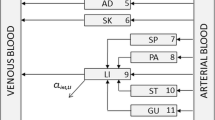
In clinical development stages, an a priori assessment of the sensitivity of the pharmacokinetic behavior with respect to physiological and anthropometric properties of human (sub-) populations is desirable. A physiology-based pharmacokinetic (PBPK) population model was developed that makes use of known distributions of physiological and anthropometric properties obtained from the literature for realistic populations. As input parameters, the simulation model requires race, gender, age, and two parameters out of body weight, height and body mass index. From this data, the parameters relevant for PBPK modeling such as organ volumes and blood flows are determined for each virtual individual. The resulting parameters were compared to those derived using a previously published model (P3M). Mean organ weights and blood flows were highly correlated between the two models, despite the different methods used to generate these parameters. The inter-individual variability differed greatly especially for organs with a log-normal weight distribution (such as fat and spleen). Two exemplary population pharmacokinetic simulations using ciprofloxacin and paclitaxel as model drugs showed good correlation to observed variability. A sensitivity analysis demonstrated that the physiological differences in the virtual individuals and intrinsic clearance variability were equally influential to the pharmacokinetic variability but were not additive. In conclusion, the new population model is well suited to assess the influence of individual physiological variability on the pharmacokinetics of drugs. It is expected that this new tool can be beneficially applied in the planning of clinical studies.
Similar content being viewed by others
References
Ette E.I, Williams P.J. (2004). Population pharmacokinetics I: background, concepts, and models. Ann. Pharmacother. 38(10):1702–1706
Willmann S., Lippert J., Sevestre M., Solodenko J., Fois F., Schmitt W. (2003). PK-Sim®: a physiologically based pharmacokinetic ‘whole-body’ model. Biosilico. 1(4):121–124
Poulin P., Theil F.P. (2000). A priori prediction of tissue: plasma partition coefficients of drugs to facilitate the use of physiologically-based pharmacokinetic models in drug discovery. J. Pharm. Sci. 89(1):16–35
Bjorkman S. (2005). Prediction of drug disposition in infants and children by means of physiologically based pharmacokinetic (PBPK) modelling: theophylline and midazolam as model drugs. Br. J. Clin. Pharmacol. 59(6):691–704
Ginsberg G., Hattis D., Sonawane B. (2004). Incorporating pharmacokinetic differences between children and adults in assessing children’s risks to environmental toxicants. Toxicol. Appl. Pharmacol. 198(2):164–183
Theil F.P, Guentert T.W, Haddad S., Poulin P. (2003). Utility of physiologically based pharmacokinetic models to drug development and rational drug discovery candidate selection. Toxicol. Lett. 138(1–2):29–49
Willmann S., Lippert J., Schmitt W. (2005). From physicochemistry to absorption and distribution: predictive mechanistic modelling and computational tools. Expert Opin. Drug. Meta. Toxicol. 1(1):159–168
Schmitt W., Willmann S. (2005). Physiology-based pharmacokinetic modeling: ready to be used. Drug Discovery Today: Technologies. 2(1):125–132
Price P.S., Conolly R.B., Chaisson C.F., Gross E.A., Young J.S., Mathis E.T., Tedder D.R. (2003). Modeling interindividual variation in physiological factors used in PBPK models of humans. Crit. Rev. Toxicol. 33(5):469–503
Edginton A.N., Willmann S., Schmitt W. (2006). Development and validation of a generic physiology-based pharmacokinetic (PBPK) model for children. Clin. Pharmacokinet. 45:1013–1034
Haerter M.W., Keldenich J., Schmitt W. (2002). Estimation of physicochemical and ADME parameters. In: Nicolaou K.C., Hanki R., Hartwig W. (eds) Handbook of Combinatorial Chemistry: Drugs, Catalysts, Materials, Vol. 2. Wiley-VCH Verlag GmbH, Weinheim, Germany, pp 743–760
Keldenich J., Schmitt W., and Willmann S. A physiological/mechanistical model for predicting organ/plasma partitioning and volume of distribution. LogP2004 – The 3rd Lipophilicity Symposium, Zurich, Switzerland. Feb 29 to Mar 4, (2004).
Third National Health and Nutrition Examination Survey (NHANES III). 1997. National Center for Health Statistics Hyattsville, MD 20782 USA. http://www.cdc.gov/nchs/nhanes.htm. (1997).
International Commission on Radiological Protection (ICRP). Basic Anatomical and Physiological Data for Use in Radiological Protection: Reference Values. ICRP Publication 89. Elsevier Science, Amsterdam, The Netherlands (2002).
Sachs L. (2003). Angewandte Statistik, 11 Auflage. Springer Verlag GmbH, Heidelberg, Deutschland
de la Grandmaison G.L., Clairand I., Durigon M. (2001). Organ weight in 684 adult autopsies: new tables for a Caucasoid population. Forensic. Sci. Int. 119(2):149–154
DuBois D., DuBois E.F. (1916). A formula to estimate the approximate surface area if height and weight be known. Arch. Int. Med. 17:863–871
Haycock G.B., Schwartz G.J., Wisotsky D.H. (1978). Geometric method for measuring body surface area: a height weight formula validated in infants, children and adults. J. Pediatr. 93:1:62–66
Gehan E.A., George S.L. (1970). Estimation of human body surface area from height and weight. Cancer. Chemother. Rep. 54:225–35
Mosteller R.D. (1987). Simplified Calculation of Body Surface Area. N. Engl. J. Med. 317(17):1098(letter)
Hense H.W., Gneiting B., Muscholl M., Broeckel U., Kuch B., Doering A., G.A. Riegger, Schunkert H. (1998). The associations of body size and body composition with left ventricular mass: impacts for indexation in adults. J. Am. Coll. Cardiol. 32(2):451–457
Lauer M.S., Anderson K.M., Larson M.G., Levy D. (1994). A new method for indexing left ventricular mass for differences in body size. Am. J. Cardiol. 74(5):487–491
Stanforth P.R., Jackson A.S., Green J.S., Gagnon J., Rankinen T., Despres J.P., Bouchard C., Leon A.S., Rao D.C., Skinner J.S., Wilmore J.H. (2004). Generalized abdominal visceral fat prediction models for black and white adults aged 17–65 y: the HERITAGE Family Study. Int. J. Obes. Relat. Metab. Disord. 28(7):925–932
Gimondo P., Mirk P., La B.A., Messina G., Pizzi C. (1995). Sonographic estimation of fetal liver weight: an additional biometric parameter for assessment of fetal growth. J. Ultrasound. Med. 14(5):327–333
Maroun L.L., Graem N. (2005). Autopsy standards of body parameters and fresh organ weights in nonmacerated and macerated human fetuses. Pediatr. Dev. Pathol. 8(2):204–217
Bergmann K.E., Bergmann R.L., Von K.R., Bohm O., Richter R., Dudenhausen J.W., Wahn U. (2003). Early determinants of childhood overweight and adiposity in a birth cohort study: role of breast-feeding. Int. J. Obes. Relat. Metab. Disord. 27(2):162–172
Urbina E.M., Gidding S.S., Bao W., Pickoff A.S., Berdusis K., Berenson G.S. (1995). Effect of body size, ponderosity, and blood pressure on left ventricular growth in children and young adults in the Bogalusa Heart Study. Circulation 91(9):2400–2406
Frankenfield D.C., Rowe W.A., Cooney R.N., Smith J.S., Becker D. (2001). Limits of body mass index to detect obesity and predict body composition. Nutrition 17(1):26–30
Clarys J.P., Provyn S., Marfell-Jones M. J. (2005). Cadaver studies and their impact on the understanding of human adiposity. Ergonomics 48(11–14):1445–1461
Willmann S., Schmitt W., Keldenich J., Lippert J., Dressman J.B. (2004). A physiological model for the estimation of the fraction dose absorbed in humans. J. Med. Chem. 47(15):4022–4031
Hardman J.G., Limbird L.E., Gilman A. (2001). Goodman and Gilman’s: The Pharmacological Basis of Therapeutics, 10th ed. McGraw Hill, New York
Shah A., Lettieri J., Heller A., Kaiser L., Collins S., and Birkett J. Pharmacokinetics of IV ciprofloxacin in subjects with normal renal function and with various degrees of renal impairment. Internal Report No. R6098 (1993).
Shah A., Lettieri J., Blum R., Millikin S., Sica D., Heller A.H. (1996). Pharmacokinetics of intravenous ciprofloxacin in normal and renally impaired subjects. J. Antimicrob. Chemother. 38(1):103–116
Sorgel F., Naber K.G., Jaehde U., Reiter A., Seelmann R., Sigl G. (1989). Gastrointestinal secretion of ciprofloxacin. Evaluation of the charcoal model for investigations in healthy volunteers. Am. J. Med. 87(5A):62S–65S
Granfors M.T., Backman J.T., Neuvonen M., Neuvonen P.J. (2004). Ciprofloxacin greatly increases concentrations and hypotensive effect of tizanidine by inhibiting its cytochrome P450 1A2-mediated presystemic metabolism. Clin. Pharmacol. Ther. 76(6):598–606
Dorne J.L., Walton K., Renwick A.G. (2001). Uncertainty factors for chemical risk assessment: human variability in the pharmacokinetics of CYP1A2 probe substrates. Food. Chem. Toxicol. 39(7):681–696
Dorne J.L., Walton K., Renwick A.G. (2004). Human variability in the renal elimination of foreign compounds and renal excretion-related uncertainty factors for risk assessment. Food. Chem. Toxicol. 42:275–298
Panday V.R., ten Bokkel Huinink W.W., Vermorken J.B., Rosing H., Koopman F.J., Swart M., Schellens J.H., Beijnen J.H. (1999). Pharmacokinetics of paclitaxel administered as a 3-hour or 96-hour infusion. Pharmacol. Res. 40(1):67–74
van den Bongard H.J. , Kemper E.M., van T.O., Rosing H., Mathot R.A., Schellens J.H., Beijnen J.H. (2004). Development and validation of a method to determine the unbound paclitaxel fraction in human plasma. Anal. Biochem. 324(1):11–15
Callies S., de Alwis D.P., Harris A., Vasey P., Beijnen J.H., Schellens J.H., Burgess M., Aarons L. (2003). A population pharmacokinetic model for paclitaxel in the presence of a novel P-gp modulator, Zosuquidar Trihydrochloride (LY335979). Br. J Clin. Pharmacol. 56(1):46–56
Vaclavikova R., Horsky S., Simek P., Gut I. (2003). Paclitaxel metabolism in rat and human liver microsomes is inhibited by phenolic antioxidants. Naunyn Schmiedebergs Arch Pharmacol. 368(3):200–209
Dorne J.L., Walton K., Renwick A.G. (2003). Human variability in CYP3A4 metabolism and CYP3A4-related uncertainty factors for risk assessment. Food. Chem. Toxicol. 41(2):201–224
MacDonald A.J., Rostami-Hodjegan A., Tucker G.T., Linkens D.A. (2002). Analysis of solvent central nervous system toxicity and ethanol interactions using a human population physiologically based kinetic and dynamic model. Regul. Toxicol. Pharmacol. 35(2 Pt 1):165–76
Jonsson F., Johanson G. (2002). Physiologically based modeling of the inhalation kinetics of styrene in humans using a bayesian population approach. Toxicol. Appl. Pharmacol. 15(1):35–49
Jonsson F., Bois F.Y., Johanson G. (2001). Assessing the reliability of PBPK models using data from methyl chloride-exposed, non-conjugating human subjects. Arch. Toxicol. 75(4):189–99
Jonsson F., Johanson G. (2001). Bayesian estimation of variability in adipose tissue blood flow in man by physiologically based pharmacokinetic modeling of inhalation exposure to toluene. Toxicology. 157(3):177–93
Sweeney L.M., Tyler T.R., Kirman C.R., Corley R.A., Reitz R.H., Paustenbach D.J., Holson J.F., Whorton M.D., Thompson K.M., Gargas M.L. (2001). Proposed occupational exposure limits for select ethylene glycol ethers using PBPK models and Monte Carlo simulations. Toxicol. Sci. 62(1):124–39
Isukapalli S.S., Roy A., Georgopoulos P.G. (1998). Stochastic response surface methods (SRSMs) for uncertainty propagation: application to environmental and biological systems. Risk Anal. 18(3):351–63
Thomas R.S., Lytle W.E., Keefe T.J., Constan A.A., Yang R.S. (1996). Incorporating Monte Carlo simulation into physiologically based pharmacokinetic models using advanced continuous simulation language (ACSL): a computational method. Fundam. Appl. Toxicol. 31(1):19–28
Clewell H.J. 3rd and Andersen M.E. Use of physiologically based pharmacokinetic modeling to investigate individual versus population risk. Toxicology 111(1–3):315–29 (1996).
Beck B.D., Mattuck R.L., Bowers T.S., Cohen J.T., O’Flaherty E. (2001). The development of a stochastic physiologically-based pharmacokinetic model for lead. Sci. Total Environ. 274(1–3):15–9
Gueorguieva I., Aarons L., Rowland M. (2006). Diazepam pharmacokinetics from preclinical to phase I using a Bayesian population physiologically based pharmacokinetic model with informative prior distributions in Winbugs. J. Pharmacokin. Pharmacodyn. 33(5):571–594
Cheymol G. (2000). Effects of obesity on pharmacokinetics. Clin Pharmacokinet. 39(3):215–231
La S.F., Patti R., Sciarrino E., Valenza F., Costanzo G.S., Tese L., Lagalla R. (2004). Ultrasound, spleen and portal hypertension. Radiol. Med (Torino). 107(4):332–343
Abramson N., Melton B. (2000). Leukocytosis: basics of clinical assessment. Am. Fam. Physician 62(9):2053–2060
Crandall D.L., Hausman G.J., Kral J.G. (1997). A review of the microcirculation of adipose tissue: anatomic, metabolic, and angiogenic perspectives. Microcirculation 4(2):211–232
Engfeldt P., Linde B. (1992). Subcutaneous adipose tissue blood flow in the abdominal and femoral regions in obese women: effect of fasting. Int. J. Obes. Relat. Metab. Disord. 16(11):875–879
Lesser G., Deutsch S. (1967). Measurement of adipose tissue blood flow and perfusion in man by uptake of 85Kr. J. Appl. Physiol. 23:621–623
Alcorn J., McNamara P.J. (2002). Ontogeny of hepatic and renal systemic clearance pathways in infants: part I. Clin. Pharmacokinet. 41(12):959–998
Cusack B.J. (2004). Pharmacokinetics in older persons. Am. J. Geriatr. Pharmacother. 2(4):274–302
Johnson J.A. (2000). Predictability of the effects of race or ethnicity on pharmacokinetics of drugs. Int. J. Clin. Pharmacol. Ther. 38(2):53–60
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Willmann, S., Höhn, K., Edginton, A. et al. Development of a Physiology-Based Whole-Body Population Model for Assessing the Influence of Individual Variability on the Pharmacokinetics of Drugs. J Pharmacokinet Pharmacodyn 34, 401–431 (2007). https://doi.org/10.1007/s10928-007-9053-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-007-9053-5