Abstract
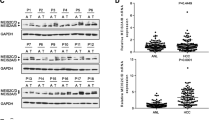
Hepatocellular carcinoma (HCC) is considered as a disease of dysfunction of the stem cells. Studies on stem cells have demonstrated that Oct4 plays a pivotal role in embryo regulation. In order to understand the role of Oct4 in HCC and the relationship among Oct4 and wnt/β-catenin and TGF-β signal pathways, we have detected the expression of Oct4, Nanog, Sox2, STAT3 as well as the genes in wnt/β-catenin, and TGF-β families in HCC cell lines and in tumor specimens from HCC patients. The authors found that Oct4 was expressed in all of the four HCC cell lines and the tumor specimens from HCC patients. Some other genes were also expressed in them with different level including Nanog, Sox2, STAT3 and TCF3, wnt10b, β-catenin, ELF, Smad3 and Smad4. The ability of the clone formation and migration of the HepG2 decreased after Oct4 was knockdowned. Silencing of Oct4 and TCF3 in HCC cell line HepG2 revealed that there were complicated relationships among Oct4, wnt/β-catenin family and TGF-β family genes. Knockdowning Oct4 reduced the expression of TGF-β family genes ELF, Smad3, Smad4 and wnt/β-catenin family genes, wnt10b, and β-catenin but increased TCF3. In reverse, knockdowning TCF3 led to the increased expression of Oct4 and TGF-β family genes. In conclusion, the expression of Oct4 in HCC may play an important role as in stem cell. Because Oct4 improves not only the function of wnt/β-catenin, but also the TGF-β signal pathways, the significance of its expression in HCC might be more complicated than we evinced before.





Similar content being viewed by others
References
Thomas MB, Zhu AX (2005) Hepatocellular carcinoma: the need for progress. J clinic Oncol 23:2892–2899
Yuan FJ, Zhou WB, Zhang JF, Zhang ZY, Zou C, Huang L, Zhang YS, Dai ZQ (2008) Anti-cancer drugs are synergistic with freezing in induction of apoptosis in HCC cells. Cryobiology 57:60–65
Chiba T, Kita K, Zheng YW, Yokosuka O, Saisho H, Iwama A, Nakauchi H, Taniguchi H (2006) Side population purified from hepatocellular carcinoma cells harbors cancer stem cell-like properties. Hepatology 44:240–251
Clarke MF, Fuller M (2006) Stem cells and cancer: two faces of eve. Cell 124:1111–1115
Freberg CT, Dahl JA, Timoskainen S, Collas P (2007) Epigenetic reprogramming of oct4 and nanog regulatory regions by embryonal carcinoma cell extract. Mol Biol Cell 18:1543–1553
Babaie Y, Herwig R, Greber B, Brink TC, Wruck W, Groth D, Lehrach H, Burdon T, Adjaye J (2007) Analysis of oct4-dependent transcriptional networks regulating self-renewal and pluripotency in human embryonic stem cells. Stem Cell 25:500–510
Hay DC, Sutherland L, Clark J, Burdon T (2004) Oct-4 knockdown induces similar patterns of endoderm and trophoblast differentiation markers in human and mouse embryonic stem cells. Stem Cell 22:225–235
Boyer LA, Lee TI, Cole MF, Johnstone SE, Levine SS, Zucker JP, Guenther MG, Kumar RM, Murray HL, Jenner RG, Gifford DK, Melton DA, Jaenisch R, Young RA (2005) Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122:947–956
Ezeh UI, Turek PJ, Reijo RA, Clark AT (2005) Human embryonic stem cell genes OCT4, NANOG, STELLAR, and GDF3 are expressed in both seminoma and breast carcinoma. Cancer 104:2255–2265
Gu G, Yuan J, Wills M, Kasper S (2007) Prostate cancer cells with stem cell characteristics reconstitute the original human tumor in vivo. Cancer Res 67:4807–4815
Chen YC, Hsu HS, Chen YW, Tsai TH, How CK, Wang CY, Hung SC, Chang YL, Tsai ML, Lee YY, Ku HH, Chiou SH (2008) Oct-4 expression maintained cancer stem-like properties in lung cancer-derived cd133-positive cells. PLoS ONE 3:1–14
Wang J, Wynshaw-Boris A (2004) The canonical Wnt pathway in early mammalian embryogenesis and stem cell maintenance/differentiation. Curr Opin Genet Dev 14:533–539
Thompson MD, Monga SP (2007) WNT/β-catenin signaling in liver health and disease. Hepatology 45:1298–1305
Reya T, Clevers H (2005) Wnt signalling in stem cells and cancer. Nature 434:843–850
Glasgow E, Mishra L (2008) Transforming growth factor-b signaling and ubiquitinators in cancer. Endocr Relat Cancer 15:59–72
Amin R, Mishra L (2008) Liver stem cells and TGF-β in hepatic carcinogenesis. Gastrointest Cancer Res 2(4 Suppl):S27–S30
Mishra L, Banker T, Murray J, Byers S, Thenappan A, He AR, Shetty K, Johnson L, Reddy EP (2009) Liver stem cells and hepatocellular carcinoma. Hepatology 49:318–329
Ouyang L, Shen LY, Li T, Liu J (2006) Inhibition effedt of Oncostatin M on metastatic homan lumg cancer cells 95-D in vitro and on mutine melanoma cells B16BL6 in vitro. Biomed Res 27:197–202
Hamill KJ, Langbein L, Jones JR, McLean W (2009) Identification of a novel family of laminin N-terminal alternate splice isoforms: structural and functional characterization. JBC 284:35588–35596
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676
Takahashi K, Tanabe K, Ohnuki M (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131:861–872
Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R (2007) In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448:318–324
Lensch MW, West JA (2008) Looking into the future of cell-based therapy. South Med J 101:79–82
Gil J, Stembalska A, Pesz KA, Sasiadek MM (2008) Cancer stem cells: the theory and perspectives in cancer therapy. J App Genet 49:193–199
Chang CC, Shieh GS, Wu P, Lin CC, Shiau AL, Wu CL (2008) Oct-3/4 expression reflects tumor progression and regulates motility of bladder cancer cells. Cancer Res 68:6281–6291
Tang Y, Kitisin K, Jogunoori W, Mueller SC, Ressom HW, Rashid A, He AR, Mendelson JS, Jessup JM, Shetty K, Zasloff M, Mishra B, Reddy EP, Johnson L, Mishra L (2008) Progenitor/stem cells give rise to liver cancer due to aberrant TGF-beta and IL-6 signaling. Proc Natl Acad Sci USA 105:2445–2450
Palma I, Pen RY, Contreras A, Ceballos-Reyes G, Coyote N, Eran L, Kofman-Alfaro S, Queipo G (2008) Participation of OCT3/4 and b-catenin during dysgenetic gonadal malignant transformation. Cancer Lett 263:204–211
Cole MF, Johnstone SE, Newman JJ, Kagey MH, Young RA (2008) Tcf3 is an integral component of the core regulatory circuitry of embryonic stem cells. Genes Dev 22:746–755
Tam WL, Lim CY, Han JY, Zhang JQ, Ang YS, Ng HH, Yang H, Lim B (2008) T-Cell factor 3 regulates embryonic stem cell pluripotency and self-renewal by the transcriptional control of multiple lineage pathways. Stem Cells 26:2019–2031
Yi F, Pereira L, Merrill BJ (2008) Tcf3 functions as a steady-state limiter of transcriptional programs of mouse embryonic stem cell self-renewal. Stem Cell 26:1951–1960
Hahn WC, Polyak K (2005) Roots and stems: stem cells in cancer. Nat Med 11:296–300
Millet C, Zhang YE (2007) Roles of smad3 in TGF-β signaling during carcinogenesis. Crit Rev Eukaryot Gene Expr 17:281–293
Shackel NA, McCaughan GW, Warner FJ (2008) Hepatocellular carcinoma development requires hepatic stem cells with altered transforming growth factor and interleukin-6 signaling. Hepatology 47:2134–2136
Acknowledgments
This study was supported by The Special Fund for The promotion of Science, Hubei Provincial Department of Education and Bureau of Science, Shiyan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuan, F., Zhou, W., Zou, C. et al. Expression of Oct4 in HCC and modulation to wnt/β-catenin and TGF-β signal pathways. Mol Cell Biochem 343, 155–162 (2010). https://doi.org/10.1007/s11010-010-0509-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0509-3