Abstract
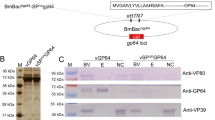
IE-2 of Bombyx mori nucleopolyhedrovirus (BmNPV) has been shown to play important roles in baculovirus infection, which are involved in gene expression and viral replication. However, the mechanism remains unknown. In this paper, by TargetP software, four genes, i.e.-2, odv-e26, odv-e56 and BmNPV-gp101 (Ac-orf116) of BmNPV and Autographa californica multiple NPV (AcMNPV) were predicted to be located in mitochondria. By BLAST tool using BmNPV IE-2 protein sequence, 14 NPVs were found to have IE-2 homologues in GenBank, and most of them were predicted to be located in mitochondria, except for that of Antheraea pernyi NPV (AnpeNPV) and Anticarsia gemmatalis NPV (AngeNPV). To observe the subcellular localization of BmNPV IE-2, a recombinant virus overexpressed the IE-2 and eGFP fusion protein was constructed. In infected BmN cells, the fluorescence specifically enriched in the cellular mitochondria. This evidence was accordant with the prediction. Further, Pull-down assay was used to select protein candidates interacting with IE-2 in B. mori cells infected with BmNPV. Of several isolated protein components, sixteen candidates were identified by MALDI-TOF mass-spectrometry, eight baculoviral proteins (ALK-EXO, F protein, IAP-1, LEF-3, LEF-9, ODV-NC42, TLP, and VP39), and eight proteins from B. mori (Actin, ADP/ATP translocase, ATP synthase subunit beta, Beta-tubulin, DNA topoisomerase 2, Histone H4, Soluble guanylyl cyclae alpha-1 subunit, Transketolase). From the functional point of view, most of these proteins were generally divided into two groups, mitochondrial interaction proteins and viral DNA replication proteins. These results implied that the IE-2 had multiple functions involved in regulating viral gene expression, viral replication and also as a component of mitochondrial factors to regulate the cellular energy supply and apoptosis.




Similar content being viewed by others
References
Herniou EA et al (2003) The genome sequence and evolution of baculoviruses. Annu Rev Entomol 48:211–234
Jehle JA et al (2006) On the classification and nomenclature of baculoviruses: a proposal for revision. Arch Virol 151(7):1257–1266
Guarino LA, Summers MD (1986) Functional mapping of a trans-activating gene required for expression of a baculovirus delayed-early gene. J Virol 57(2):563–571
Friesen PD, Miller LK (1986) The regulation of baculovirus gene expression. Curr Top Microbiol Immunol 131:31–49
Krappa R, Roncarati R, Knebel-Morsdorf D (1995) Expression of PE38 and IE2, viral members of the C3HC4 finger family, during baculovirus infection: PE38 and IE2 localize to distinct nuclear regions. J Virol 69(9):5287–5293
Imai N et al (2000) Analysis of interaction between molecules of Bombyx mori nucleopolyhedrovirus IE-2 using a yeast two-hybrid system. Acta Virol 44(3):199–202
Ayres MD et al (1994) The complete DNA sequence of Autographa californica nuclear polyhedrosis virus. Virology 202(2):586–605
Birnbaum MJ, Clem RJ, Miller LK (1994) An apoptosis-inhibiting gene from a nuclear polyhedrosis virus encoding a polypeptide with Cys/his sequence motifs. J Virol 68(4):2521–2528
Crook NE, Clem RJ, Miller LK (1993) An apoptosis-inhibiting baculovirus gene with a zinc finger-like motif. J Virol 67(4):2168–2174
Krappa R, Knebel-Morsdorf D (1991) Identification of the very early transcribed baculovirus gene PE-38. J Virol 65(2):805–812
Theilmann DA, Stewart S (1992) Molecular analysis of the trans-activating IE-2 gene of Orgyia pseudotsugata multicapsid nuclear polyhedrosis virus. Virology 187(1):84–96
Thiem SM, Miller LK (1989) A baculovirus gene with a novel transcription pattern encodes a polypeptide with a zinc finger and a leucine zipper. J Virol 63(11):4489–4497
Gomi S et al (1997) Deletion analysis of four of eighteen late gene expression factor gene homologues of the baculovirus, BmNPV. Virology 230(1):35–47
Yoo S, Guarino LA (1994) The Autographa californica nuclear polyhedrosis virus ie2 gene encodes a transcriptional regulator. Virology 202(2):746–753
Kool M et al (1994) Identification of genes involved in DNA replication of the Autographa californica baculovirus. Proc Natl Acad Sci USA 91(23):11212–11216
Passarelli AL, Miller LK (1993) Three baculovirus genes involved in late and very late gene expression: ie-1, ie-n, and lef-2. J Virol 67(4):2149–2158
Gomi S, Majima K, Maeda S (1999) Sequence analysis of the genome of Bombyx mori nucleopolyhedrovirus. J Gen Virol 80(5):1323–1337
Lu A, Miller LK (1995) The roles of eighteen baculovirus late expression factor genes in transcription and DNA replication. J Virol 69(2):975–982
Evans JT, Rohrmann GF (1997) The baculovirus single-stranded DNA binding protein, LEF-3, forms a homotrimer in solution. J Virol 71(5):3574–3579
Hang X, Dong W, Guarino LA (1995) The lef-3 gene of Autographa californica nuclear polyhedrosis virus encodes a single-stranded DNA-binding protein. J Virol 69(6):3924–3928
McDougal VV, Guarino LA (1999) Autographa californica nuclear polyhedrosis virus DNA polymerase: measurements of processivity and strand displacement. J Virol 73(6):4908–4918
Evans JT et al (1999) Characterization of the interaction between the baculovirus ssDNA-binding protein (LEF-3) and putative helicase (P143). J Gen Virol 80(Pt 2):493–500
Wu Y, Carstens EB (1998) A baculovirus single-stranded DNA binding protein, LEF-3, mediates the nuclear localization of the putative helicase P143. Virology 247(1):32–40
Mainz D, Quadt I, Knebel-Morsdorf D (2002) Nuclear IE2 structures are related to viral DNA replication sites during baculovirus infection. J Virol 76(10):5198–5207
Shevchenko A et al (1996) Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal Chem 68(5):850–858
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Tamura K et al (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24(8):1596–1599
Tamura K et al (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Emanuelsson O et al (2007) Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc 2(4):953–971
Bork P, Koonin EV (1996) Protein sequence motifs. Curr Opin Struct Biol 6(3):366–376
Deveraux QL, Reed JC (1999) IAP family proteins—suppressors of apoptosis. Genes Dev 13(3):239–252
Vucic D et al (1997) Inhibition of reaper-induced apoptosis by interaction with inhibitor of apoptosis proteins (IAPs). Proc Natl Acad Sci USA 94(19):10183–10188
Bryant B et al (2009) A lepidopteran orthologue of reaper reveals functional conservation and evolution of IAP antagonists. Insect Mol Biol 18(3):341–351
Thomenius M et al (2011) Mitochondrial fusion is regulated by Reaper to modulate Drosophila programmed cell death. Cell Death Differ 18(10):1640–1650
McDougal VV, Guarino LA (2000) The Autographa californica nuclear polyhedrosis virus p143 gene encodes a DNA helicase. J Virol 74(11):5273–5279
Liu CY et al (2009) RING and coiled-coil domains of baculovirus IE2 are critical in strong activation of the cytomegalovirus major immediate-early promoter in mammalian cells. J Virol 83(8):3604–3616
Charlton CA, Volkman LE (1993) Penetration of Autographa californica nuclear polyhedrosis virus nucleocapsids into IPLB Sf 21 cells induces actin cable formation. Virology 197(1):245–254
Lanier LM, Volkman LE (1998) Actin binding and nucleation by Autographa california M nucleopolyhedrovirus. Virology 243(1):167–177
Slack JM, Kuzio J, Faulkner P (1995) Characterization of v-cath, a cathepsin L-like proteinase expressed by the baculovirus Autographa californica multiple nuclear polyhedrosis virus. J Gen Virol 76(Pt 5):1091–1098
Mikhailov VS, Okano K, Rohrmann GF (2003) Baculovirus alkaline nuclease possesses a 5′ → 3′ exonuclease activity and associates with the DNA-binding protein LEF-3. J Virol 77(4):2436–2444
Fan W et al (1998) Identification of altered expression of ADP/ATP translocase during cellular senescence in vitro. Exp Gerontol 33(5):457–465
Acknowledgments
We thank Prof. Weide Shen and Dr. Bing Li from Soochow University, China for suggestions regarding the study’s experiments. We also thank Chenrui Hou (Shanghai applied protein technology co. Ltd) for providing support with MALDI analysis, and Dr. Jun Wu and Prof. Rongfang Wang (DiaSys Diagnostic Systems, Shanghai, Co. Ltd) for help in the pull-down assay. This paper was supported by Natural Science Foundation (31272499).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Y., Wu, Y., Wu, Y. et al. Screening of candidate proteins interacting with IE-2 of Bombyx mori nucleopolyhedrovirus. Mol Biol Rep 40, 5797–5804 (2013). https://doi.org/10.1007/s11033-013-2683-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-013-2683-3