Abstract
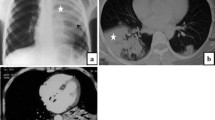
We retrospectively analyzed the medical records and clinical characteristics of 15 patients diagnosed with Penicilliosis marneffei (PSM) between January 1, 1993, and December 31, 2012, at the Third Affiliated Hospital of Sun Yat-sen University. The most common symptoms of PSM were fever (14/15, 93 %), cough (13/15, 87 %), and sputum production (6/15, 40 %), weight loss (14/15, 60 %), lymph node enlargement (9/15, 60 %), hepatosplenomegaly (7/15, 47 %), anemia (7/15, 47 %), and hemoptysis (4/15, 26 %). The most common underlying diseases in patients diagnosed with PSM were AIDS (9/15, 60 %), post-organ transplantation (3/15, 20 %), rheumatic autoimmune disease (2/15, 13 %), and hematological malignancy (1/15, 7 %). All patients, except those with AIDS, were treated with immunosuppressant drugs. White blood cell counts were increased in 10/15 (67 %) patients, while hemoglobin concentrations were decreased in 8/15 (53 %) patients. The ratios of CD4+/total T lymphocytes and CD4+/CD8+ T lymphocytes declined in all the 11 test cases. Nodular lesions or masses were the most common anomalies detected during computed tomography scans, but disseminated inflammation and interstitial changes were also seen. Clinical samples with positive culture results were obtained from sputum or secretions obtained by bronchoscopy, venous blood, percutaneous pulmonary puncture, bone marrow, or skin lesions. Between 1993 and 2003, only four cases of PSM, all connected with AIDS, were diagnosed, while 11 cases of PSM, with or without concurrent AIDS, were diagnosed between 2003 and 2012. Amphotericin B was used to control the disease in some cases. In conclusion, the occurrence of PSM, especially in patients without concurrent AIDS, has increased. The early culture of Penicillium marneffei from clinical samples is critical for correct diagnosis of PSM, and amphotericin B is recommended as the first choice for treatment.









Similar content being viewed by others
References
Chariyalertsak S, Sirisanthana T, Supparatpinyo K, Nelson KE. Seasonal variation of disseminated Penicillium marneffei infections in northern Thailand: a clue to the reservoir? J Infect Dis. 1996;173:1490–3.
Li J, Zhao Y, Jin C. Analysis of antiretroviral treatment policy and strategy for AIDS. Chin J AIDS STD. 2013;19:144–7.
Zhou X, Wang L, Ming S. An analysis of AIDS projects funded by National Natural Science Foundation of China from 1999 to 2011. Chin J AIDS STD. 2013;19:56–60.
San Segundo D, Brunet M, Ballesteros MA, Millan O, Muro M, Castro MJ, et al. Prospective study of biomarkers of immune response in lung transplant recipients. Transplant Proc. 2012;44:2666–8.
Hu Y, Zhang J, Li X, Yang Y, Zhang Y, Ma J, et al. Penicillium marneffei infection: an emerging disease in mainland China. Mycopathologia. 2013;175:57–67.
Cao C, Liang L, Wang W, Luo H, Huang S, Liu D, et al. Common reservoirs for Penicillium marneffei infection in humans and rodents, China. Emerg Infect Dis. 2011;17:209–14.
Ogawa Y, Watanabe D, Sako H, Bando H, Yajima K, Taniguchi T, et al. A diagnosed, cured case of an HIV-infected Japanese subject developing disseminated penicilliosis after Thailand travel. Kansenshogaku Zasshi. 2010;84:740–3.
Jung JY, Jo GH, Kim HS, Park MY, Shin JH, Chin BS, et al. Disseminated penicilliosis in a Korean human immunodeficiency virus infected patient from Laos. J Korean Med Sci. 2012;27:697–700.
Hart J, Dyer JR, Clark BM, McLellan DG, Perera S, Ferrari P. Travel-related disseminated Penicillium marneffei infection in a renal transplant patient. Transpl Infect Dis. 2012;14:434–9.
Piehl MR, Kaplan RL, Haber MH. Disseminated penicilliosis in a patient with acquired immunodeficiency syndrome. Arch Pathol Lab Med. 1988;112:1262–4.
Supparatpinyo K, Khamwan C, Baosoung V, Nelson KE, Sirisanthana T. Disseminated Penicillium marneffei infection in southeast Asia. Lancet. 1994;344:110–3.
Shen YZ, Wang ZY, Lu HZ. Penicillium marneffei chylous ascites in acquired immune deficiency syndrome: a case report. World J Gastroenterol. 2012;18:5312–4.
Luo DQ, Chen MC, Liu JH, Li Z, Li HT. Disseminated Penicillium marneffei infection in an SLE patient: a case report and literature review. Mycopathologia. 2011;171:191–6.
Rokiah I, Ng KP, Soo-Hoo TS. Penicillium marneffei infection in an AIDS patient—a first case report from Malaysia. Med J Malays. 1995;50:101–4.
Tong AC, Wong M, Smith NJ. Penicillium marneffei infection presenting as oral ulcerations in a patient infected with human immunodeficiency virus. J Oral Maxillofac Surg. 2001;59:953–6.
Le T, Chi NH, Cuc NTK, Sieu TPM, Shikuma CM, Farrar J, et al. AIDS-associated Penicillium marneffei infection of the central nervous system. Clin Infect Dis. 2010;51:1458–62.
Lin CY, Sun HY, Chen MY, Hsieh SM, Sheng WH, Lo YC, et al. Aetiology of cavitary lung lesions in patients with HIV infection. HIV Med. 2009;10:191–8.
Rongrungruang Y, Levitz SM. Interactions of Penicillium marneffei with human leukocytes in vitro. Infect Immun. 1999;67:4732–6.
Huang LF, Tang XP, Cai WP, Chen XJ, Lei CL, Li LH, et al. An analysis of opportunistic infection in 762 in patients with human immunodeficiency virus infection in Guangdong areas. Zhonghua Nei Ke Za Zhi. 2010;49:653–6.
Le T, Wolbers M, Chi NH, Quang VM, Chinh NT, Lan NP, et al. Epidemiology, seasonality, and predictors of outcome of AIDS-associated Penicillium marneffei infection in Ho Chi Minh City, Viet Nam. Clin Infect Dis. 2011;52:945–52.
Hamilton AJ, Jeavons L, Youngchim S, Vanittanakom N. Recognition of fibronectin by Penicillium marneffei conidia via sialic acid-dependent process and its relationship to the interaction between conidia and laminin. Infect Immun. 1999;67:5200–5.
Boyce KJ, Andrianopoulos A. Morphogenetic circuitry regulating growth and development in the dimorphic pathogen Penicillium marneffei. Eukaryot Cell. 2013;12:154–60.
Chong YB, Tan LP, Robinson S, Lim SK, Ng KP, Keng TC, et al. Penicilliosis in lupus patients presenting with unresolved fever: a report of 2 cases and literature review. Trop Biomed. 2012;29:270–6.
Liu Y, Xing H, Xiao YY, Fu LB, Yang Z, Ma L. Disseminated Penicilliosis marneffei in a child: case report. J Clin Dermatol. 2012;41:420–2.
Sudjaritruk T, Sirisanthana T, Sirisanthana V. Immune reconstitution inflammatory syndrome from Penicillium marneffei in an HIV-infected child: a case report and review of literature. BMC Infect Dis. 2012;12:28.
Lee PP, Chan KW, Lee TL, Ho MH, Chen XY, Li CH, et al. Penicilliosis in children without HIV infection—are they immunodeficient? Clin Infect Dis. 2012;54:8–19.
Galimberti R, Torre AC, Baztán MC, Rodriquez-Chiapetta F. Emerging systemic fungal infections. Clin Dermatol. 2012;30:633–50.
Larsson M, Nguyen LH, Wertheim HF, Dao TT, Taylor W, Horby P, et al. Clinical characteristics and outcome of Penicillium marneffei infection among HIV-infected patients in northern Vietnam. AIDS Res Ther. 2012;9:24.
Wong SY, Wong KF. Penicillium marneffei infection in AIDS. Pathol Res Int. 2011;2011:10.
Acknowledgments
Social development project of science and technology department of Guangdong province. Serial number, 2010B31600034.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, F., Bi, X., Zou, X. et al. Retrospective Analysis of 15 Cases of Penicilliosis marneffei in a Southern China Hospital. Mycopathologia 177, 271–279 (2014). https://doi.org/10.1007/s11046-014-9737-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-014-9737-5