Abstract
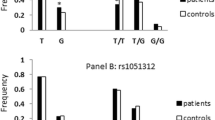
Synaptosomal-associated protein 25 (SNAP25) is an essential component for synaptic vesicle mediated release of neurotransmitters. Deficiencies or abnormal structure or function of SNAP25 protein, possibly arising through genetic variations in the relevant DNA code, has been suggested to play role in the pathology of several neurobehavioural disorders including Attention deficit Hyperactivity Disorder (ADHD) and a number of polymorphisms in the SNAP25 gene has been studied for association with the disorder. In the present investigation, for the first time association between ADHD and six SNAP25 polymorphisms, rs1889189, rs362569, rs362988, rs3746544, rs1051312, and rs8636 was explored in eastern Indian population. Subjects were recruited following the Diagnostic and Statistical Manual for Mental Disorders-IV. Genomic DNA isolated from peripheral blood leukocytes of ADHD probands (n = 150), their parents (n = 272) and ethnically matched controls (n = 100) was used for amplifying target sites. Data obtained were subjected to population- as well as family-based analyses. While case–control analysis revealed lack of any significant difference for alleles, family-based studies revealed a mild over transmission rs3746544 ‘T’ and rs8636 ‘C’ alleles (P = 0.05 and 0.03 respectively). Haplotypes formed between rs362569 “T”, 362988 “G”, rs3746544 “T”, rs1051312 “T” and rs8636 “C” in different combinations showed statistically significant transmission to ADHD probands. Excepting rs3746544 and rs8636, all the tested sites showed very low linkage disequilibrium between them. Data obtained in this preliminary study indicates that rs3746544 ‘T’ allele may have some role in the disease etiology in the studied Indian population.
Similar content being viewed by others
References
Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, Sklar P (2005) Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatr 57:1313–1323
Mehler-Wex C, Rieder P, Gerlach M (2006) Dopaminergic dysbalance in distinct basal ganglia neurocircuits: implications for the pathophysiology of Parkinson’s disease, schizophrenia and attention deficit hyperactivity disorder. Neurotox Res 10:167–179
Kustanovich V, Merriman B, McGough J, McCracken JT, Smalley SL, Nelson SF (2003) Biased paternal transmission of SNAP-25 risk alleles in attention-deficit hyperactivity disorder. Mol Psychiatr 8:309–315
Guan L, Wang B, Chen Y, Yang L, Li J, Qian Q, Wang Z, Faraone SV, Wang Y (2008) A high-density single-nucleotide polymorphism screen of 23 candidate genes in attention deficit hyperactivity disorder: suggesting multiple susceptibility genes among Chinese Han population. Mol Psychiatr 14:546–554
Gizer IA, Ficks C, Waldman ID (2009) Candidate gene studies of ADHD: a meta-analytic review. Hum Genet 126:51–90
Rizo J, Sudhof TC (2002) Snares and Munc18 in synaptic vesicle fusion. Nat Rev Neurosci 3:641–653
Sudhof TC (2004) The synaptic vesicle cycle. Annu Rev Neurosci 27:509–547
Sollner T, Whiteheart SW, Brunner M, Erdjument-Bromage H, Geromanos S, Tempst P, Rothman JE (1993) Snap receptors implicated in vesicle targeting and fusion. Nature 362:318–324
Oyler GA, Higgins GA, Hart RA, Battenberg E, Billingsley M, Bloom FE, Wilson MC (1989) The identification of a novel synaptosomal-associated protein, SNAP-25, differentially expressed by neuronal subpopulations. J Cell Biol 109:3039–3052
Zhao N, Hashida H, Takahashi N, Sakaki Y (1994) Cloning and sequence analysis of the human SNAP25 cDNA. Gene 145:313–314
Hess EJ, Jinnah HA, Kozak CA, Wilson MC (1992) Spontaneous locomotor hyperactivity in a mouse mutant with a deletion including the Snap gene on chromosome 2. J Neurosci 12:2865–2874
Heyser CJ, Wilson MC, Gold LH (1995) Coloboma hyperactive mutant exhibits delayed neurobehavioral developmental milestones. Develop Brain Res 89:264–269
Bruno KJ, Freet CS, Twining RC, Egami K, Grigson PS, Hess EJ (2007) Abnormal latent inhibition and impulsivity in coloboma mice, a model of ADHD. Neurobiol Dis 25:206–216
Raber J, Mehta PP, Kreifeldt M, Parsons LH, Weiss F, Bloom FE, Wilson MC (1997) Coloboma hyperactive mutant mice exhibit regional and transmitter-specific deficits in neurotransmission. J Neurochem 68:176–186
Jones MD, Williams ME, Hess EJ (2001) Abnormal presynaptic catecholamine regulation in a hyperactive SNAP-25-deficient mouse mutant. Pharmacol Biochem Behav 68:669–676
Wilson MC (2000) Coloboma mouse mutant as an animal model of hyperkinesis and attention deficit hyperactivity disorder. Neurosci Biobehav Rev 24:51–57
Maglott DR, Feldblyum TV, Durkin AS, Nierman WC (1996) Radiation hybrid mapping of SNAP, PCSK2, and THBD (human chromosome 20p). Mamm Genome 7:400–401
Barr CL, Feng Y, Wigg K, Bloom S, Roberts W, Malone M, Schachar R, Tannock R, Kennedy JL (2000) Identification of DNA variants in the SNAP-25 gene and linkage study of these polymorphisms and attention-deficit hyperactivity disorder. Mol Psychiatr 5:405–409
Brophy K, Hawi Z, Kirley A, Fitzgerald M, Gill M (2002) Synaptosomal-associated protein 25 (SNAP-25) and attention deficit hyperactivity disorder ADHD: evidence of linkage and association in the Irish population. Mol Psychiatr 7:913–917
Feng Y, Crosbie J, Wigg K, Pathare T, Ickowicz A, Schachar R, Tannock R, Roberts W, Malone M, Swanson J, Kennedy JL, Barr CL (2005) The SNAP25 gene as a susceptibility gene contributing to attention-deficit hyperactivity disorder. Mol Psychiatr 0:998–1005
Gosso MF, de Geus EJC, Polderman TJC, Boomsma DI, Heutink P, Posthuma D (2008) Common variants underlying cognitive ability: further evidence for association between the SNAP-25 gene and cognition using a family-based study in two independent Dutch cohorts. Genes Brain Behav 7:355–364
Lasky-Su J, Neale BM, Franke B, Anney RJ, Zhou K, Maller JB, Vasquez AA, Chen W, Asherson P, Buitelaar J, Banaschewski T, Ebstein R, Gill M, Miranda A, Mulas F, Oades RD, Roeyers H, Rothenberger A, Sergeant J, Sonuga-Barke E, Steinhausen HC, Taylor E, Daly M, Laird N, Lange C, Faraone SV (2008) Genome-wide association scan of quantitative traits for attention deficit hyperactivity disorder identifies novel associations and confirms candidate gene associations. Am J Med Genet 147B:1345–1354
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders: text revised, 4th edn. Washington, DC
Conners CK (1997) Conners’ rating scales–revised multi-health systems. Toronto, Canada
Wechsler D (1991) Wechsler intelligence scale for children: manual, 3rd edn. Psychological Corporation, San Antonio
Raj Bharat (1971) J AIISH norms on SFB with Indian children. J AIISH 2:34–39
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acid Res 16:1215
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–386
Dudbridge F (2003) Pedigree disequilibrium tests for multilocus haplotypes. Genet Epidemiol 25:115–221
Spielman RS, McGinnis RE, Ewens WJ (1993) Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus IDDM. Am J Hum Genet 52:506–516
Lenth RV (2007) Statistical power calculations. J Anim Sci 85:E24–E29
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc B 57:289–300
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265
Yuan Yu H, Chiou JJ, Tseng WH, Liu CH, Liu CK, Lin YJ, Wang HH, Yao A, Chen YT, Hsu CN (2006) FASTSNP: an always up-to-date and extendable service for SNP function analysis and prioritization. Nucleic Acids Res 34:W635–W641
Lee PH, Shatkay H (2007) F-SNP: computationally predicted functional SNPs for disease association studies. Nucleic Acids Res 36:D821–D824
Mill J, Richards S, Knight J, Curran S, Taylor E, Asherson P (2004) Haplotype analysis of SNAP-25 suggests a role in the aetiology of ADHD. Mol Psychiatr 9:801–810
Forero DA, Arboleda GH, Vasquez R, Arboleda H (2009) Candidate genes involved in neural plasticity and the risk for ADHD: a meta-analysis of 8 common variants. J Psychiatr Neurosci 34:361–366
Söderqvist S, McNab F, Peyrard-Janvid M, Matsson H, Humphreys K, Kere J, Klingberg T (2010) The SNAP25 Gene is linked to working memory capacity and maturation of the posterior cingulate cortex during childhood. Biol Psychiatr 68:1120–1125
Kim JW, Waldman ID, Faraone SV, Biederman J, Doyle AE, Purcell S, Arbeitman L, Fagerness J, Sklar P, Smoller JW (2007) Investigation of Parent-of-origin effects in ADHD candidate genes. Am J Med Genet b144B:776–780
Kim JW, Biederman J, Arbeitman L, Fagerness J, Doyle AE, Petty C, Perlis RH, Purcell S, Smoller JW, Faraone SV, Sklar P (2007) Investigation of variation in SNAP-25 and ADHD and relationship to co-morbid major depressive disorder. Am J Med Genet a144B:781–790
Renner TJ, Walitza S, Dempfle A, Eckert L, Romanos M, Gerlach M, Schafer H, Warnke A, Lesch KP, Jacob C (2008) Allelic variants of SNAP25 in a family-based sample of ADHD. J Neural Transm 115:317–321
Das M, Das Bhowmik A, Bhaduri N, Sarkar K, Ghosh P, Sinha S, Ray A, Chatterjee A, Mukhopadhyay K (2011) Role of gene-gene/gene-environment interaction in the etiology of eastern Indian ADHD probands. Prog Neuropsychopharmacol Biol Psychiatr 35:577–587
Carroll LS, Kendall K, O’Donovan ML, Owen MJ, Williams NM (2009) Evidence that putative ADHD low risk alleles at SNAP25 may increase the risk of Schizophrenia. Am J Med Genet 150:893–899
Gosso MF, de Geus EJC, van Belzen MJ, Polderman TJC, Heutink P, Boomstra DI (2006) The SNAP-25 gene is associated with cognitive ability: evidence from a family-based study in two independent Dutch cohorts. Mol Psychiatr 11:878–888
Ardlie KG, Kruglyak L, Seielstad M (2002) Patterns of linkage disequilibrium in the human genome. Nat Rev Genet 3:299–309
Chadwick R, McGinnis W (1987) Temporal and spatial distribution of transcripts from the Deformed gene of Drosophila. EMBO J 63:779–789
McGinnis N, Kuziora MA, McGinnis W (1990) Human Hox-42 and Drosophila Deformed encode similar regulatory specificities in Drosophila embryos and larvae. Cell 63:969–976
Acknowledgments
Authors would like to thank all the families who participated in this research. KS and NB are recipients of the Council of Scientific and Industrial Research Fellowship. Authors are also thankful to the Lady Tata Memorial Trust for Research Scholarship provided to KS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarkar, K., Bhaduri, N., Ghosh, P. et al. Role of SNAP25 Explored in Eastern Indian Attention Deficit Hyperactivity Disorder Probands. Neurochem Res 37, 349–357 (2012). https://doi.org/10.1007/s11064-011-0618-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0618-8