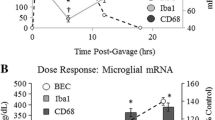
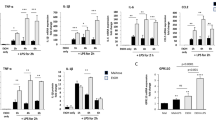
Abstract
We reported that the ethanol-induced innate immune response by activating TLR4 signaling triggers gliosis and neuroinflammation. Ethanol also activates other immune receptors, such as NOD-like-receptors, and specifically NLRP3-inflammasome in astroglial cells, to stimulate caspase-1 cleavage and IL-1β and IL-18 cytokines production. Yet, whether microglia NLRs are also sensitive to the ethanol effects that contribute to neuroinflammation is uncertain. Using cerebral cortexes of the chronic alcohol-fed WT and TLR4−/− mice, we demonstrated that chronic ethanol treatment enhanced TLR4 mediated-NLRP3/Caspase-1 complex activation, and up-regulated pro-inflammatory cytokines and chemokines levels. Ethanol-induced NLRP3-inflammasome activation and mitochondria-ROS generation were also observed in cultured microglial cells. The up-regulation of CD45high/CD11b+ cell populations and matrix metalloproteinase-9 levels was also noted in the cortexes of the ethanol-treated WT mice. Notably, elimination of the TLR4 function abolished most ethanol-induced neuroinflammatory effects. Thus, our results demonstrate that ethanol triggers TLR4-mediated NLRP3-inflammasome activation in glial cells, and suggest that microglia stimulation may compromise the permeability of blood–brain barrier events to contribute to ethanol-induced neuroinflammation and brain damage.







Similar content being viewed by others
References
Becker CE, O’Neill LA (2007) Inflammasomes in inflammatory disorders: the role of TLRs and their interactions with NLRs. Semin Immunopathol 29:239–248
Mariathasan S, Monack DM (2007) Inflammasome adaptors and sensors: intracellular regulators of infection and inflammation. Nat Rev 7:31–40
Franchi L et al (2012) Sensing and reacting to microbes through the inflammasomes. Nat Immunol 13:325–332
Skundric DS et al (2015) Emerging role of IL-16 in cytokine-mediated regulation of multiple sclerosis. Cytokine 75(2):234–248
Akira S (2006) TLR signaling. Curr Top Microbiol Immunol 311:1–16
Kawai T, Akira S (2009) The roles of TLRs, RLRs and NLRs in pathogen recognition. Int Immunol 21:317–337
Mogensen TH (2009) Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev 22:240–273
Haorah J et al (2008) Mechanism of alcohol-induced oxidative stress and neuronal injury. Free Radic Biol Med 45:1542–1550
Fan Y et al (2015) Tight junction disruption of blood-brain barrier in white matter lesions in chronic hypertensive rats. NeuroReport 26:1039–1043
Pfefferbaum A et al (2010) Transcallosal white matter degradation detected with quantitative fiber tracking in alcoholic men and women: selective relations to dissociable functions. Alcohol Clin Exp Res 34:1201–1211
Schulte T et al (2010) White matter fiber degradation attenuates hemispheric asymmetry when integrating visuomotor information. J Neurosci 30:12168–12178
Alfonso-Loeches S et al (2012) Toll-like receptor 4 participates in the myelin disruptions associated with chronic alcohol abuse. Glia 60:948–964
Alfonso-Loeches S et al (2010) Pivotal role of TLR4 receptors in alcohol-induced neuroinflammation and brain damage. J Neurosci 30:8285–8295
Harper C, Matsumoto I (2005) Ethanol and brain damage. Curr Opin Pharmacol 5:73–78
Jiao H et al (2011) Specific role of tight junction proteins claudin-5, occludin, and ZO-1 of the blood-brain barrier in a focal cerebral ischemic insult. J Mol Neurosci: MN 44:130–139
Blanco AM et al (2005) Involvement of TLR4/type I IL-1 receptor signaling in the induction of inflammatory mediators and cell death induced by ethanol in cultured astrocytes. J Immunol 175:6893–6899
Fernandez-Lizarbe S et al (2009) Critical role of TLR4 response in the activation of microglia induced by ethanol. J Immunol 183:4733–4744
Lippai D et al (2013) Alcohol-induced IL-1beta in the brain is mediated by NLRP3/ASC inflammasome activation that amplifies neuroinflammation. J Leukoc Biol 94:171–182
Alfonso-Loeches S et al (2014) Role of mitochondria ROS generation in ethanol-induced NLRP3 inflammasome activation and cell death in astroglial cells. Front Cell Neurosci 8:216
Hellwig S et al (2013) The brain’s best friend: microglial neurotoxicity revisited. Front Cell Neurosci 7:71
Aguzzi A et al (2013) Microglia: scapegoat, saboteur, or something else? Science 339:156–161
Kettenmann H et al (2013) Microglia: new roles for the synaptic stripper. Neuron 77:10–18
Lehnardt S (2010) Innate immunity and neuroinflammation in the CNS: the role of microglia in Toll-like receptor-mediated neuronal injury. Glia 58:253–263
Hanamsagar R et al (2012) Inflammasome activation and IL-1beta/IL-18 processing are influenced by distinct pathways in microglia. J Neurochem 119:736–748
Heneka MT et al (2014) Innate immune activation in neurodegenerative disease. Nat Rev 14:463–477
Sumi N et al (2010) Lipopolysaccharide-activated microglia induce dysfunction of the blood-brain barrier in rat microvascular endothelial cells co-cultured with microglia. Cell Mol Neurobiol 30:247–253
Jones AW, Sternebring B (1992) Kinetics of ethanol and methanol in alcoholics during detoxification. Alcohol Alcohol 27:641–647
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Franklin KBJ, Paxinos G (1997) The mouse brain in stereotaxic coordinates. Academic Press, San Diego
Mora-Lee S et al (2012) Therapeutic effects of hMAPC and hMSC transplantation after stroke in mice. PLoS ONE 7:e43683
Rodrigo R et al (2010) Hyperammonemia induces neuroinflammation that contributes to cognitive impairment in rats with hepatic encephalopathy. Gastroenterology 139:675–684
Ransohoff RM, Perry VH (2009) Microglial physiology: unique stimuli, specialized responses. Annu Rev Immunol 27:119–145
Nakajima K, Kohsaka S (2001) Microglia: activation and their significance in the central nervous system. J Biochem 130:169–175
Fernandes-Alnemri T, Alnemri ES (2008) Assembly, purification, and assay of the activity of the ASC pyroptosome. Methods Enzymol 442:251–270
Cheeran MC et al (2007) Dysregulated interferon-gamma responses during lethal cytomegalovirus brain infection of IL-10-deficient mice. Virus Res 130:96–102
Ford AL et al (1995) Normal adult ramified microglia separated from other central nervous system macrophages by flow cytometric sorting. Phenotypic differences defined and direct ex vivo antigen presentation to myelin basic protein-reactive CD4+ T cells compared. J Immunol 154:4309–4321
LaFrance-Corey RG, Howe CL (2011) Isolation of brain-infiltrating leukocytes. J Vis Exp. doi:10.3791/2747
Marten NW et al (2000) Role of viral persistence in retaining CD8(+) T cells within the central nervous system. J Virol 74:7903–7910
Zhou H et al (2009) Role of endothelial TLR4 for neutrophil recruitment into central nervous system microvessels in systemic inflammation. J Immunol 183:5244–5250
He J, Crews FT (2008) Increased MCP-1 and microglia in various regions of the human alcoholic brain. Exp Neurol 210:349–358
Barker BR et al (2011) Cross-regulation between the IL-1beta/IL-18 processing inflammasome and other inflammatory cytokines. Curr Opin Immunol 23:591–597
van de Veerdonk FL et al (2011) Inflammasome activation and IL-1beta and IL-18 processing during infection. Trends Immunol 32:110–116
Soltys Z et al (2001) Morphology of reactive microglia in the injured cerebral cortex. Fractal analysis and complementary quantitative methods. J Neurosci Res 63:90–97
Nimmerjahn A et al (2005) Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308:1314–1318
Jin X et al (2012) Temporal changes in cell marker expression and cellular infiltration in a controlled cortical impact model in adult male C57BL/6 mice. PLoS ONE 7:e41892
Marques CP et al (2008) Prolonged microglial cell activation and lymphocyte infiltration following experimental herpes encephalitis. J Immunol 181:6417–6426
Miller AM (2011) Role of IL-33 in inflammation and disease. J Inflamm 8:22
Netea MG et al (2009) Differential requirement for the activation of the inflammasome for processing and release of IL-1beta in monocytes and macrophages. Blood 113:2324–2335
Chai Q et al (2014) Enhancement of blood-brain barrier permeability and reduction of tight junction protein expression are modulated by chemokines/cytokines induced by rabies virus infection. J Virol 88:4698–4710
Bekku Y et al (2009) Brevican distinctively assembles extracellular components at the large diameter nodes of Ranvier in the CNS. J Neurochem 108:1266–1276
Yamaguchi Y (1996) Brevican: a major proteoglycan in adult brain. Perspect Dev Neurobiol 3:307–317
Valles SL et al (2004) Chronic ethanol treatment enhances inflammatory mediators and cell death in the brain and in astrocytes. Brain Pathol 14:365–371
Netea MG et al (2010) IL-1beta processing in host defense: beyond the inflammasomes. PLoS Pathog 6:e1000661
Zou J, Crews FT (2012) Inflammasome-IL-1beta signaling mediates ethanol inhibition of hippocampal neurogenesis. Front Neurosci 6:77
Martinon F (2010) Signaling by ROS drives inflammasome activation. Eur J Immunol 40:616–619
Montoliu C et al (1995) Ethanol increases cytochrome P4502E1 and induces oxidative stress in astrocytes. J Neurochem 65:2561–2570
Qin L, Crews FT (2012) Chronic ethanol increases systemic TLR3 agonist-induced neuroinflammation and neurodegeneration. J Neuroinflamm 9:130
Shaftel SS et al (2007) Chronic interleukin-1beta expression in mouse brain leads to leukocyte infiltration and neutrophil-independent blood brain barrier permeability without overt neurodegeneration. J Neurosci 27:9301–9309
Lee EJ et al (2014) Comparison of the effects of matrix metalloproteinase inhibitors on TNF-alpha release from activated microglia and TNF-alpha converting enzyme activity. Biomol Ther 22:414–419
Lee SH et al (2010) Effect of arachidonic acid on hypoxia-induced IL-6 production in mouse ES cells: involvement of MAPKs, NF-kappaB, and HIF-1alpha. J Cell Physiol 222:574–585
Haorah J et al (2007) Alcohol-induced blood-brain barrier dysfunction is mediated via inositol 1,4,5-triphosphate receptor (IP3R)-gated intracellular calcium release. J Neurochem 100:324–336
Nakamura H et al (2000) Brevican is degraded by matrix metalloproteinases and aggrecanase-1 (ADAMTS4) at different sites. J Biol Chem 275:38885–38890
Anderson JM (2001) Molecular structure of tight junctions and their role in epithelial transport. News Physiol Sci 16:126–130
Takeshita Y, Ransohoff RM (2012) Inflammatory cell trafficking across the blood-brain barrier: chemokine regulation and in vitro models. Immunol Rev 248:228–239
Kielian T et al (2001) CXC chemokine receptor-2 ligands are required for neutrophil-mediated host defense in experimental brain abscesses. J Immunol 166:4634–4643
Pascual M et al (2015) Cytokines and chemokines as biomarkers of ethanol-induced neuroinflammation and anxiety-related behavior: role of TLR4 and TLR2. Neuropharmacology 89:352–359
Qin L et al (2008) Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment. J Neuroinflamm 5:10
Achur RN et al (2010) Circulating cytokines as biomarkers of alcohol abuse and alcoholism. J Neuroimmune Pharmacol 5:83–91
Acknowledgments
We would like to thank Alberto Hernandez and Alicia Martinez from the Confocal Microscopy and Flow Cytometry Services at Prince Felipe Research Center, respectively. We are thankful to Dr. S. Akira who provided us with the TLR4−/− knockout mice. This work has been supported by grants from the Spanish Ministry of Science and Innovation (SAF 2009-07503, SAF2012-33747), ERAB (EA 13 08), the Carlos III Institute and FEDER funds (RTA-Network, RD2012/0028/0007 G0/005), PNSD (2014I010), GV-Consellería de Educación (ACOMP2014, 2015) and PROMETEO/2014.
Author contributions
SAL designed, performed and analyzed the experimental work results. JUP participated in the immunohistochemistry and RT-PCR analyses. MJM helped with the RT-PCR analyses and western blot. UGP performed experiments and analyses of microglial activation. CG designed the experiments and corrected the final version of the manuscript. All the authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial/competing interests.
Additional information
Special Issue: 40th year of the Neurochemical Research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alfonso-Loeches, S., Ureña-Peralta, J., Morillo-Bargues, M.J. et al. Ethanol-Induced TLR4/NLRP3 Neuroinflammatory Response in Microglial Cells Promotes Leukocyte Infiltration Across the BBB. Neurochem Res 41, 193–209 (2016). https://doi.org/10.1007/s11064-015-1760-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-015-1760-5