ABSTRACT
Purpose
The aim of this study was to develop a Bayesian dose individualisation tool for warfarin. This was incorporated into the freely available software TCIWorks (www.tciworks.info) for use in the clinic.
Methods
All pharmacokinetic and pharmacodynamic (PKPD) models for warfarin in the medical literature were identified and evaluated against two warfarin datasets. The model with the best external validity was used to develop an optimal design for Bayesian parameter control. The performance of this design was evaluated using simulation-estimation techniques. Finally, the model was implemented in TCIWorks.
Results
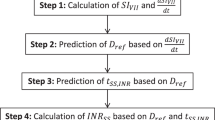
A recently published warfarin KPD model was found to provide the best fit for the two external datasets. Optimal sampling days within the first 14 days of therapy were found to be days 3, 4, 5, 11, 12, 13 and 14. Simulations and parameter estimations suggested that the design will provide stable estimates of warfarin clearance and EC50. A single patient example showed the potential clinical utility of the method in TCIWorks.
Conclusions
A Bayesian dose individualisation tool for warfarin was developed. Future research to assess the predictive performance of the tool in warfarin patients is required.




Similar content being viewed by others
Abbreviations
- BSV:
-
between-subject variability
- CL:
-
clearance
- CYP:
-
cytochrome P450
- diag:
-
diagonal
- EC50:
-
the drug concentration at 1/2 of maximum effect
- FIM:
-
Fisher information matrix
- INR:
-
international normalised ratio
- J:
-
Jacobian matrix
- KPD:
-
kinetic-pharmacodynamic
- MAP:
-
maximum a posterior
- MTT:
-
mean transit time
- PCA:
-
prothrombin complex activity
- PKPD:
-
pharmacokinetic-pharmacodynamic
- PT:
-
prothrombin time
- RSE:
-
relative standard error
- RUV:
-
residual unexplained variability
- SE:
-
standard error
- VKORC1:
-
vitamin K epoxide reductase
- VPC:
-
visual predictive check (external [e] or internal [i])
REFERENCES
The International Warfarin Pharmacogenetics Consortium. Estimation of the warfarin dose with clinical and pharmacogenetic data. N Engl J Med. 2009;360(8):753–64.
Anticoagulants in the Secondary Prevention of Events in Coronary Thrombosis (ASPECT) Research Group. Effect of long-term oral anticoagulant treatment on mortality and cardiovascular morbidity. Lancet. 1994;343(8896):499–503.
The Stroke Prevention in Atrial Fibrillation Investigators. Bleeding during antithrombotic therapy in patients with atrial fibrillation. Arch Intern Med. 1996;156(4):409–16.
Jones M, McEwan P, Morgan CL, Peters JR, Goodfellow J, Currie CJ. Evaluation of the pattern of treatment, level of anticoagulation control, and outcome of treatment with warfarin in patients with non-valvar atrial fibrillation: a record linkage study in a large British population. Heart. 2005;91(4):472–7.
Hylek EM, Evans-Molina C, Shea C, Henault LE, Regan S. Major hemorrhage and tolerability of warfarin in the first year of therapy among elderly patients with atrial fibrillation. Circulation. 2007;115(21):2689–96.
Oake N, Fergusson DA, Forster AJ, van Walraven C. Frequency of adverse events in patients with poor anticoagulation: a meta-analysis. Can Med Assoc J. 2007;176(11):1589–94.
Oake N, Jennings A, Forster AJ, Fergusson D, Doucette S, van Walraven C. Anticoagulation intensity and outcomes among patients prescribed oral anticoagulant therapy: a systematic review and meta-analysis. Can Med Assoc J. 2008;179(3):235–44.
Tan ES, Bonnett TJ, Abdelhafiz AH. Delayed discharges due to initiation of warfarin in atrial fibrillation: a prospective audit. Am J Ger Pharmacol. 2007;5(3):232–5.
The Stroke Prevention in Atrial Fibrillation Investigators. Adjusted-dose warfarin versus low-intensity, fixed-dose warfarin plus aspirin for high-risk patients with atrial fibrillation: Stroke Prevention in Atrial Fibrillation III randomised clinical trial. Lancet. 1996;348(9028):633–8.
van Walraven C, Jennings A, Oake N, Fergusson D, Forster AJ. Effect of study setting on anticoagulation control. Chest. 2006;129(5):1155–66.
Rose AJ, Ozonoff A, Berlowitz DR, Henault LE, Hylek EM. Warfarin dose management affects INR control. J Thromb Haemost. 2009;7(1):94–101.
Kalra L, Yu G, Perez I, Lakhani A, Donaldson N. Prospective cohort study to determine if trial efficacy of anticoagulation for stroke prevention in atrial fibrillation translates into clinical effectiveness. BMJ. 2000;320(7244):1236–9.
Gage BF, Eby C, Johnson JA, Deych E, Rieder MJ, Ridker PM, et al. Use of pharmacogenetic and clinical factors to predict the therapeutic dose of warfarin. Clin Pharmacol Ther. 2008;84(3):326–31.
Wadelius M, Pirmohamed M. Pharmacogenetics of warfarin: current status and future challenges. Pharmacogenom J. 2007;7(2):99–111.
McMillin GA, Melis R, Wilson A, Strong MB, Wanner NA, Vinik RG, et al. Gene-based warfarin dosing compared with standard of care practices in an orthopedic surgery population: a prospective, parallel cohort study. Ther Drug Monit. 2010;32(3):338–45.
Caraco Y, Blotnick S, Muszkat M. CYP2C9 genotype-guided warfarin prescribing enhances the efficacy and safety of anticoagulation: a prospective randomized controlled study. Clin Pharmacol Ther. 2007;83(3):460–70.
Wadelius M, Chen LY, Lindh JD, Eriksson N, Ghori MJR, Bumpstead S, et al. The largest prospective warfarin-treated cohort supports genetic forecasting. Blood. 2009;113(4):784–92.
Sconce EA, Khan TI, Wynne HA, Avery P, Monkhouse L, King BP, et al. The impact of CYP2C9 and VKORC1 genetic polymorphism and patient characteristics upon warfarin dose requirements: proposal for a new dosing regimen. Blood. 2005;106(7):2329–33.
Lenzini P, Wadelius M, Kimmel S, Anderson JL, Jorgensen AL, Pirmohamed M, et al. Integration of genetic, clinical, and inr data to refine warfarin dosing. Clin Pharmacol Ther. 2010;87(5):572–8.
Anderson JL, Horne BD, Stevens SM, Grove AS, Barton S, Nicholas ZP, et al. Randomized trial of genotype-guided versus standard warfarin dosing in patients initiating oral anticoagulation. Circulation. 2007;116(22):2563–70.
Sheiner LB, Beal S, Rosenberg B, Marathe VV. Forecasting individual pharmacokinetics. Clin Pharmacol Ther. 1979;26(3):294–305.
Sheiner LB, Beal SL. Bayesian individualization of pharmacokinetics: simple implementation and comparison with non-Bayesian methods. J Pharm Sci. 1982;71(12):1344–8.
Duffull SB, Chadaud S, Nony P, Laveille C, Girard P, Aarons L. A pharmacokinetic simulation model for ivabradine in healthy volunteers. Eur J Pharm Sci. 2000;10(4):285–94.
O’Reilly RA, Aggeler PM, Leong LS. Studies on the coumadin anticoagulant drugs: the pharmacodynamics of warfarin in man. J Clin Invest. 1963;42:1542–51.
O’Reilly RA, Aggeler PM. Studies on coumarin anticoagulant drugs. Initiation of warfarin therapy without a loading dose. Circulation. 1968;38(1):169–77.
Mohammed Abdul M, Jiang X, Williams K, Day R, Roufogalis B, Liauw W, et al. Pharmacodynamic interaction of warfarin with cranberry but not with garlic in healthy subjects. Br J Pharmacol. 2008;154:1691–700.
Jiang XM, Blair EYL, McLachlan AJ. Investigation of the effects of herbal medicines on warfarin response in healthy subjects: a population pharmacokinetic-pharmacodynamic modeling approach. J Clin Pharmacol. 2006;46(11):1370–8.
Hamberg AK, Wadelius M, Lindh JD, Dahl ML, Padrini R, Deloukas P, et al. A pharmacometric model describing the relationship between warfarin dose and INR response with respect to variations in CYP2C9, VKORC1, and age. Clin Pharmacol Ther. 2010;87(6):727–34.
Jacqmin P, Snoeck E, van Schaick E, Gieschke R, Pillai P, Steimer J, et al. Modellling response time profile in the absence of drug concentrations: definition and performance evaluation of the K-PD model. J Pharmacokinet Pharmacodyn. 2007;34(1):57–85.
Duffull SB, Retout S, Mentre F. The use of simulated annealing for finding optimal population designs. Comput Methods Programs Biomed. 2002;69(1):25–35.
Holford NH. Target concentration intervention: beyond Y2K. Br J Clin Pharmacol. 2001;52 Suppl 1:55S–9.
Sheiner LB. Computer-aided long-term anticoagulation therapy. Comput Biomed Res. 1969;2(6):507–18.
Nagashima R, O’Reilly RA, Levy G. Kinetics of pharmacologic effects in man: the anticoagulant action of warfarin. Clin Pharmacol Ther. 1969;10(1):22–35.
Theofanous TG, Barile RG. Multiple-dose kinetics of oral anticoagulants: methods of analysis and optimized dosing. J Pharm Sci. 1973;62(2):261–6.
Wiegman H, Vossepoel AM. A computer program for long term anticoagulation control. Comput Programs Biomed. 1977;7(2):71–84.
Wingard Jr LB, O’Reilly RA, Levy G. Pharmacokinetics of warfarin enantiomers: a search for intrasubject correlations. Clin Pharmacol Ther. 1978;23(2):212–7.
Svec JM, Coleman RW, Mungall DR, Ludden TM. Bayesian pharmacokinetic/pharmacodynamic forecasting of prothrombin response to warfarin therapy: preliminary evaluation. Ther Drug Monit. 1985;7(2):174–80.
Murray B, Coleman R, McWaters D, Ludden T, Mungall D. Pharmacodynamics of warfarin at steady state. Ther Drug Monit. 1987;9(1):1–5.
Powers WF, Abbrecht PH, Covell DG. Systems and microcomputer approach to anticoagulant therapy. IEEE Trans Biomed Eng. 1980;27(9):520–3.
Vadher B, Patterson DLH, Leaning M. Prediction of the international normalized ratio and maintenance dose during the initiation of warfarin therapy. Br J Clin Pharmacol. 1999;48(1):63–70.
Abbrecht PH, O’Leary TJ, Behrendt DM. Evaluation of a computer-assisted method for individualized anticoagulation: retrospective and prospective studies with a pharmacodynamic model. Clin Pharmacol Ther. 1982;32(1):129–36.
Chan E, O’Reilly R, Rowland M. Stereochemical aspects of warfarin in man: a combined pharmacokinetic-pharmacodynamic model. Br J Clin Pharmacol. 1984;19:545P.
Holford NH. Rational warfarin dosing: a pharmacokinetic-pharmacodynamic analysis. Clin Pharmacol Ther [abstract]. 1986;39:199.
Yuen E, Gueorguieva I, Wise S, Soon D, Aarons L. Ethnic differences in the population pharmacokinetics and pharmacodynamics of warfarin. J Pharmacokinet Pharmacodyn. 2010;37(1):3–24.
Hamberg AK, Dahl ML, Barban M, Scordo MG, Wadelius M, Pengo V, et al. A PK-PD model for predicting the impact of age, CYP2C9, and VKORC1 genotype on individualization of warfarin therapy. Clin Pharmacol Ther. 2007;81(4):529–38.
Pitsiu M, Parker EM, Aarons L, Rowland M. A Bayesian method based on clotting factor activity for the prediction of maintenance warfarin dosage regimens. Ther Drug Monit. 2003;25(1):36–40.
Manotti C, Moia M, Palareti G, Pengo V, Ria L, Dettori AG. Effect of computer-aided management on the quality of treatment in anticoagulated patients: a prospective, randomized, multicenter trial of APROAT (Automated PRogram for Oral Anticoagulant Treatment). Haematologica. 2001;86(10):1060–70.
Poller L, Shiach CR, MacCallum PK, Johansen AM, Munster AM, Magalhaes A, et al. Multicentre randomised study of computerised anticoagulant dosage. Lancet. 1998;352(9139):1505–9.
Ryan PJ, Gilbert M, Rose PE. Computer control of anticoagulant dose for therapeutic management. Br Med J. 1989;299(6709):1207–9.
Roberts GW, Helboe T, Nielsen CBM, Gallus AS, Jensen I, Cosh DG, et al. Assessment of an age-adjusted warfarin initiation protocol. Ann Pharmacother. 2003;37(6):799–803.
Lee J, Lin T. Warfdocs for web version 1.0 Copyright © 2006 Regents of the University of California; 2006 [cited 2010 July 5 2010]; [website]]. Available from: warfdocs.ucdavis.edu.
Hamberg AK, Wadelius M, Pirmohamed M, Jonsson EN. Internal and external evaluation of a pharmacometric model for warfarin using prediction corrected visual predicitive check (PC-VPC). Population Approach Group Europe. Nineteenth meeting June 8–11 2010. http://www.page-meeting.org/pdf_assets/2122-Poster_PAGE2010_published.pdf; Berlin, Germany2010.
Pitsiu M, Parker EM, Aarons L, Rowland M. Population pharmacokinetics and pharmacodynamics of warfarin in healthy-young adults. Eur J Pharm Sci. 1993;1(3):151–7.
Horsti J, Uppa H, Vilpo JA. Poor agreement among prothrombin time international normalized ratio methods: comparison of seven commercial reagents. Clin Chem. 2005;51(3):553–60.
ACKNOWLEDGMENTS
The authors wish to thank Professor Andrew McLachlan, Faculty of Pharmacy, University of Sydney, Australia, for access to the warfarin data set for external model evaluation. We thank Anna-Karin Hamberg, Faculty of Pharmacy, Uppsala University, Uppsala, Sweden, for assistance with the published KPD warfarin model. At the time of writing, Dan Wright was the recipient of a University of Otago Postgraduate Scholarship.
Author information
Authors and Affiliations
Corresponding author
APPENDIX 1: THE OTAGO WARFARIN PKPD MODEL
APPENDIX 1: THE OTAGO WARFARIN PKPD MODEL
Methods
PKPD Modelling
A warfarin PKPD model was developed using data from the O’Reilly dataset. Population PK analysis was carried out with NONMEM VI using the first-order conditional estimation with interaction (FOCEI). One- and two-compartment pharmacokinetic models with first-order absorption were fitted to the warfarin concentration data. An absorption lag time (tlag) was also considered to help describe the apparent absorption delay of warfarin. Total body weight, sex and age were considered as covariates. These were retained in the model if inclusion decreased the objective function value by 3.84 or more (χ 2, p ≤ 0.05, d.f. = 1). Model discrimination and covariate inclusion were also assessed using graphical goodness-of-fit analysis. The between-subject variability was assumed to be log-normally distributed, with a mean of zero and a variance of ω2. Residual error was modelled using a combined additive and proportional error model.
Once the best PK model had been identified, a simultaneous PKPD model was developed. Graphical inspection of a PCA versus time plot overlayed on a concentration versus time plot suggested a considerable delay between warfarin response and dose. Therefore, only delayed effects models, such as effect compartment and inhibitory turnover (Imax) models, were considered. Candidate PKPD models were evaluated by comparison of the objective function values and by visual inspection of visual predictive checks (VPCs).
Internal and External Model Evaluation
The model was evaluated by simulating 1000 patients under the model and plotting the 10th, 50th, and 90th percentiles of the simulated PCA values. This was compared to the same percentiles from the O’Reilly dataset (VPCi) and the University of Sydney (VPCe).
Results
PKPD Modelling (The Otago Model)
Parameter and error model estimates for the final PKPD model are presented in Table III. A one-compartment pharmacokinetic model with first-order absorption and tlag provided the best fit for the data. Visual inspection of covariate plots suggested a relationship between CL, V and weight. A model for weight, standardised to 70 kg, was applied to clearance and volume. An allometric scaling function was also applied to clearance. The final model for clearance is given by
and for volume by
An inhibitory turnover model provided the best fit for the pharmacodynamic (PCA) data (see Eq. 3).
where Ratein is the zero-order production rate for PCA, Kout is PCA elimination rate constant, Imax is the maximum inhibition of PCA, Cp is the plasma concentration (of warfarin), and EC50 is warfarin plasma concentration at 1/2 Imax.
Internal and External Evaluation of the Otago Model
An internal visual predictive check (VPCi) for warfarin response compared to the O’Reilly dataset is presented in the main body of the text (Fig. 1a). The plot suggests good model performance. An external evaluation (VPCe) of the model against the Sydney dataset is presented in the main body of the text (Fig. 2a). The model appears to under-predict anticoagulant response somewhat. This is discussed further in the main body of the paper.
Rights and permissions
About this article
Cite this article
Wright, D.F.B., Duffull, S.B. Development of a Bayesian Forecasting Method for Warfarin Dose Individualisation. Pharm Res 28, 1100–1111 (2011). https://doi.org/10.1007/s11095-011-0369-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-011-0369-x