Abstract
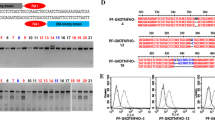
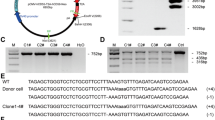
Generation of transgenic pigs for xenotransplantation is one of the most promising technologies for resolving organ shortages. Human heme oxygenase-1 (hHO-1/HMOX1) can protect transplanted organs by its strong anti-oxidative, anti-apoptotic, and anti-inflammatory effects. Soluble human TNFRI-Fc (shTNFRI-Fc) can inhibit the binding of human TNF-α (hTNF-α) to TNF receptors on porcine cells, and thereby, prevent hTNF-α-mediated inflammation and apoptosis. Herein, we successfully generated shTNFRI-Fc-F2A-HA-hHO-1 transgenic (TG) pigs expressing both shTNFRI-Fc and hemagglutinin-tagged-human heme oxygenase-1 (HA-hHO-1) by using an F2A self-cleaving peptide. shTNFRI-Fc and HA-hHO-1 transgenes containing the F2A peptide were constructed under the control of the CAG promoter. Transgene insertion and copy number in the genome of transgenic pigs was confirmed by polymerase chain reaction (PCR) and Southern blot analysis. Expressions of shTNFRI-Fc and HA-hHO-1 in TG pigs were confirmed using PCR, RT-PCR, western blot, ELISA, and immunohistochemistry. shTNFRI-Fc and HA-hHO-1 were expressed in various organs, including the heart, lung, and spleen. ELISA assays detected shTNFRI-Fc in the sera of TG pigs. For functional analysis, fibroblasts isolated from a shTNFRI-Fc-F2A-HA-hHO-1 TG pig (i.e., #14; 1 × 105 cells) were cultured with hTNF-α (20 ng/mL) and cycloheximide (10 μg/mL). The viability of shTNFRI-Fc-F2A-HA-hHO-1 TG pig fibroblasts was significantly higher than that of the wild type (wild type vs. shTNFRI-Fc-F2A-HA-hHO-1 TG at 24 h, 31.6 ± 3.2 vs. 60.4 ± 8.3 %, respectively; p < 0.05). Caspase-3/-7 activity of the shTNFRI-Fc-F2A-HA-hHO-1 TG pig fibroblasts was lower than that of the wild type pig fibroblasts (wild type vs. shTNFRI-Fc-F2A-HA-hHO-1 TG at 12 h, 812,452 ± 113,078 RLU vs. 88,240 ± 10,438 RLU, respectively; p < 0.05). These results show that shTNFRI-Fc and HA-hHO-1 TG pigs generated by the F2A self-cleaving peptide express both shTNFRI-Fc and HA-hHO-1 molecules, which provides protection against oxidative and inflammatory injury. Utilization of the F2A self-cleaving peptide is a promising tool for generating multiple TG pigs for xenotransplantation.





Similar content being viewed by others
References
Abraham NG, Kappas A (2008) Pharmacological and clinical aspects of heme oxygenase. Pharmacol Rev 60(1):79–127
Araki K, Araki M, Miyazaki J, Vassalli P (1995) Site-specific recombination of a transgene in fertilized eggs by transient expression of Cre recombinase. Proc Natl Acad Sci USA 92(1):160–164
Chen G, Qian H, Starzl T, Sun H, Garcia B, Wang X, Wise Y, Liu Y, Xiang Y, Copeman L, Liu W, Jevnikar A, Wall W, Cooper DK, Murase N, Dai Y, Wang W, Xiong Y, White DJ, Zhong R (2005) Acute rejection is associated with antibodies to non-Gal antigens in baboons using Gal-knockout pig kidneys. Nat Med 11(12):1295–1298. doi:10.1038/nm1330
Cho B, Koo OJ, Hwang JI, Kim H, Lee EM, Hurh S, Park SJ, Ro H, Yang J, Surh CD, D’Apice AJ, Lee BC, Ahn C (2011) Generation of soluble human tumor necrosis factor-alpha receptor 1-Fc transgenic pig. Transplantation 92(2):139–147. doi:10.1097/TP.0b013e3182215e7e
Cowan PJ, Aminian A, Barlow H, Brown AA, Chen CG, Fisicaro N, Francis DM, Goodman DJ, Han W, Kurek M, Nottle MB, Pearse MJ, Salvaris E, Shinkel TA, Stainsby GV, Stewart AB, d’Apice AJ (2000) Renal xenografts from triple-transgenic pigs are not hyperacutely rejected but cause coagulopathy in non-immunosuppressed baboons. Transplantation 69(12):2504–2515
de Felipe P, Luke GA, Hughes LE, Gani D, Halpin C, Ryan MD (2006) E unum pluribus: multiple proteins from a self-processing polyprotein. Trends Biotechnol 24(2):68–75. doi:10.1016/j.tibtech.2005.12.006
Deng W, Yang D, Zhao B, Ouyang Z, Song J, Fan N, Liu Z, Zhao Y, Wu Q, Nashun B, Tang J, Wu Z, Gu W, Lai L (2011) Use of the 2A peptide for generation of multi-transgenic pigs through a single round of nuclear transfer. PLoS ONE 6(5):e19986. doi:10.1371/journal.pone.0019986
Donnelly ML, Hughes LE, Luke G, Mendoza H, ten Dam E, Gani D, Ryan MD (2001) The ‘cleavage’ activities of foot-and-mouth disease virus 2A site-directed mutants and naturally occurring ‘2A-like’ sequences. J Gen Virol 82(Pt 5):1027–1041
Fisicaro N, Londrigan SL, Brady JL, Salvaris E, Nottle MB, O’Connell PJ, Robson SC, d’Apice AJ, Lew AM, Cowan PJ (2011) Versatile co-expression of graft-protective proteins using 2A-linked cassettes. Xenotransplantation 18(2):121–130. doi:10.1111/j.1399-3089.2011.00631.x
Furler S, Paterna JC, Weibel M, Bueler H (2001) Recombinant AAV vectors containing the foot and mouth disease virus 2A sequence confer efficient bicistronic gene expression in cultured cells and rat substantia nigra neurons. Gene Ther 8(11):864–873. doi:10.1038/sj.gt.3301469
Hurh S, Cho B, You DJ, Kim H, Lee EM, Lee SH, Park SJ, Park HC, Koo OJ, Yang J, Oh KH, Lee BC, Hwang JI, Ahn C (2013) Expression analysis of combinatorial genes using a bi-cistronic T2A expression system in porcine fibroblasts. PLoS ONE 8(7):e70486. doi:10.1371/journal.pone.0070486
Hwang JI, Oh YS, Shin KJ, Kim H, Ryu SH, Suh PG (2005) Molecular cloning and characterization of a novel phospholipase C, PLC-eta. Biochem J 389(Pt 1):181–186. doi:10.1042/bj20041677
Kim D, Kim JY, Koh HS, Lee JP, Kim YT, Kang HJ, Hwang WS, Kim YB, Lee JS, Ahn C (2005) Establishment and characterization of endothelial cell lines from the aorta of miniature pig for the study of xenotransplantation. Cell Biol Int 29(8):638–646. doi:10.1016/j.cellbi.2005.03.016
Kolber-Simonds D, Lai L, Watt SR, Denaro M, Arn S, Augenstein ML, Betthauser J, Carter DB, Greenstein JL, Hao Y, Im GS, Liu Z, Mell GD, Murphy CN, Park KW, Rieke A, Ryan DJ, Sachs DH, Forsberg EJ, Prather RS, Hawley RJ (2004) Production of alpha-1,3-galactosyltransferase null pigs by means of nuclear transfer with fibroblasts bearing loss of heterozygosity mutations. Proc Natl Acad Sci USA 101(19):7335–7340. doi:10.1073/pnas.0307819101
Koo BC, Kwon MS, Choi BR, Kim JH, Cho SK, Sohn SH, Cho EJ, Lee HT, Chang W, Jeon I, Park JK, Park JB, Kim T (2006) Production of germline transgenic chickens expressing enhanced green fluorescent protein using a MoMLV-based retrovirus vector. FASEB J 20(13):2251–2260. doi:10.1096/fj.06-5866com
Le Bas-Bernardet S, Tillou X, Poirier N, Dilek N, Chatelais M, Devalliere J, Charreau B, Minault D, Hervouet J, Renaudin K, Crossan C, Scobie L, Cowan PJ, d’Apice AJ, Galli C, Cozzi E, Soulillou JP, Vanhove B, Blancho G (2011) Xenotransplantation of galactosyl-transferase knockout, CD55, CD59, CD39, and fucosyl-transferase transgenic pig kidneys into baboons. Transplant Proc 43(9):3426–3430. doi:10.1016/j.transproceed.2011.09.024
Maines MD (1988) Heme oxygenase: function, multiplicity, regulatory mechanisms, and clinical applications. FASEB J 2(10):2557–2568
Park SJ, Park HJ, Koo OJ, Choi WJ, Moon JH, Kwon DK, Kang JT, Kim S, Choi JY, Jang G, Lee BC (2012) Oxamflatin improves developmental competence of porcine somatic cell nuclear transfer embryos. Cell Reprogram 14(5):398–406. doi:10.1089/cell 2012.0007
Petersen B, Ramackers W, Lucas-Hahn A, Lemme E, Hassel P, Queisser AL, Herrmann D, Barg-Kues B, Carnwath JW, Klose J, Tiede A, Friedrich L, Baars W, Schwinzer R, Winkler M, Niemann H (2011) Transgenic expression of human heme oxygenase-1 in pigs confers resistance against xenograft rejection during ex vivo perfusion of porcine kidneys. Xenotransplantation 18(6):355–368. doi:10.1111/j.1399-3089.2011.00674.x
Phelps CJ, Koike C, Vaught TD, Boone J, Wells KD, Chen SH, Ball S, Specht SM, Polejaeva IA, Monahan JA, Jobst PM, Sharma SB, Lamborn AE, Garst AS, Moore M, Demetris AJ, Rudert WA, Bottino R, Bertera S, Trucco M, Starzl TE, Dai Y, Ayares DL (2003) Production of alpha 1,3-galactosyltransferase-deficient pigs. Science (New York, NY) 299(5605):411–414. doi:10.1126/science.1078942
Provost E, Rhee J, Leach SD (2007) Viral 2A peptides allow expression of multiple proteins from a single ORF in transgenic zebrafish embryos. Genesis (New York, NY: 2000) 45(10):625–629. doi:10.1002/dvg.20338
Robson SC, Cooper DK, d’Apice AJ (2000) Disordered regulation of coagulation and platelet activation in xenotransplantation. Xenotransplantation 7(3):166–176
Sato K, Balla J, Otterbein L, Smith RN, Brouard S, Lin Y, Csizmadia E, Sevigny J, Robson SC, Vercellotti G, Choi AM, Bach FH, Soares MP (2001) Carbon monoxide generated by heme oxygenase-1 suppresses the rejection of mouse-to-rat cardiac transplants. J Immunol (Baltimore, Md: 1950) 166(6):4185–4194
Szymczak AL, Vignali DA (2005) Development of 2A peptide-based strategies in the design of multicistronic vectors. Expert Opin Biol Ther 5(5):627–638. doi:10.1517/14712598.5.5.627
Szymczak AL, Workman CJ, Wang Y, Vignali KM, Dilioglou S, Vanin EF, Vignali DA (2004) Correction of multi-gene deficiency in vivo using a single ‘self-cleaving’ 2A peptide-based retroviral vector. Nat Biotechnol 22(5):589–594. doi:10.1038/nbt957
Tabata T, de Perrot M, Keshavjee S, Liu M, Downey GP, Waddell TK (2003) Accommodation after lung xenografting from hamster to rat. Transplantation 75(5):607–612. doi:10.1097/01.tp.0000053353.03389.1c
Takahagi Y, Fujimura T, Miyagawa S, Nagashima H, Shigehisa T, Shirakura R, Murakami H (2005) Production of alpha 1,3-galactosyltransferase gene knockout pigs expressing both human decay-accelerating factor and N-acetylglucosaminyltransferase III. Mol Reprod Dev 71(3):331–338. doi:10.1002/mrd.20305
Tian Y, Li W, Wang L, Liu C, Lin J, Zhang X, Zhang N, He S, Huang J, Jia B, Liu M (2013) Expression of 2A peptide mediated tri-fluorescent protein genes were regulated by epigenetics in transgenic sheep. Biochem Biophys Res Commun 434(3):681–687. doi:10.1016/j.bbrc.2013.04.009
Trichas G, Begbie J, Srinivas S (2008) Use of the viral 2A peptide for bicistronic expression in transgenic mice. BMC Biol 6:40. doi:10.1186/1741-7007-6-40
Yeom HJ, Koo OJ, Yang J, Cho B, Hwang JI, Park SJ, Hurh S, Kim H, Lee EM, Ro H, Kang JT, Kim SJ, Won JK, O’Connell PJ, Surh CD, Lee BC, Ahn C (2012) Generation and characterization of human heme oxygenase-1 transgenic pigs. PLoS ONE 7(10):e46646. doi:10.1371/journal.pone.0046646
Acknowledgments
This study was supported by MKE (#10033805, #1033838, #1033839), RDA (#PJ009802), BK 21 for Veterinary Science, and Hanwha L&C. We thank Dr. Young Seek Lee for his valuable contribution and discussions. We also thank Miss Asong Kim and Nari Lee for their excellent administrative support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Sol Ji Park and Bumrae Cho have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Park, S.J., Cho, B., Koo, O.J. et al. Production and characterization of soluble human TNFRI-Fc and human HO-1(HMOX1) transgenic pigs by using the F2A peptide. Transgenic Res 23, 407–419 (2014). https://doi.org/10.1007/s11248-013-9780-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-013-9780-x