Abstract
Purpose
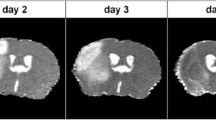
Magnetic resonance imaging (MRI) with targeted contrast agents provides a promising means for diagnosis and treatment monitoring after cerebrovascular injury. Our goal was to demonstrate the feasibility of this approach to detect the neuroinflammatory biomarker intercellular adhesion molecule-1 (ICAM-1) after stroke and to establish a most efficient imaging procedure.
Procedures
We compared two types of ICAM-1-functionalized contrast agent: T 1-shortening gadolinium chelate-containing liposomes and T 2 (*)-shortening micron-sized iron oxide particles (MPIO). Binding efficacy and MRI contrast effects were tested in cell cultures and a mouse stroke model.
Results
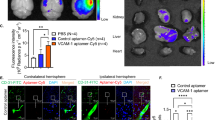
Both ICAM-1-targeted agents bound effectively to activated cerebrovascular cells in vitro, generating significant MRI contrast-enhancing effects. Direct in vivo MRI-based detection after stroke was only achieved with ICAM-1-targeted MPIO, although both contrast agents showed similar target-specific vascular accumulation.
Conclusions
Our study demonstrates the potential of in vivo MRI of post-stroke ICAM-1 upregulation and signifies target-specific MPIO as most suitable contrast agent for molecular MRI of cerebrovascular inflammation.




Similar content being viewed by others
References
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17:796–808
Thakur M, Lentle BC (2005) Report of a summit on molecular imaging. Radiology 236:753–755
Gupta H, Weissleder R (1996) Targeted contrast agents in MR imaging. Magn Reson Imaging Clin N Am 4:171–184
Dijkhuizen RM, Nicolay K (2003) Magnetic resonance imaging in experimental models of brain disorders. J Cereb Blood Flow Metab 23:1383–1402
Baird AE, Warach S (1998) Magnetic resonance imaging of acute stroke. J Cereb Blood Flow Metab 18:583–609
Deddens LH, Van Tilborg GA, Mulder WJ, De Vries HE, Dijkhuizen RM (2012) Imaging neuroinflammation after stroke: current status of cellular and molecular MRI strategies. Cerebrovasc Dis 33:392–402
Muller RN, Roch A, Colet J-M, Ouakssim A, Gillis P (2001) Particulate magnetic contrast agents. In: Merbach AE, Tóth E (eds) The chemistry of contrast agents in medical magnetic resonance imaging. Wiley, New York, pp 417–435
Wijagkanalan W, Kawakami S, Hashida M (2011) Designing dendrimers for drug delivery and imaging: pharmacokinetic considerations. Pharm Res 28:1500–1519
Mulder WJ, Strijkers GJ, Van Tilborg GA, Griffioen AW, Nicolay K (2006) Lipid-based nanoparticles for contrast-enhanced MRI and molecular imaging. NMR Biomed 19:142–164
Laurent S, Boutry S, Mahieu I, Vander Elst L, Muller RN (2009) Iron oxide based MR contrast agents: from chemistry to cell labeling. Curr Med Chem 16:4712–4727
Barber PA, Foniok T, Kirk D et al (2004) MR molecular imaging of early endothelial activation in focal ischemia. Ann Neurol 56:116–120
Jin AY, Tuor UI, Rushforth D et al (2009) Magnetic resonance molecular imaging of post-stroke neuroinflammation with a P-selectin targeted iron oxide nanoparticle. Contrast Media Mol Imaging 4:305–311
Van Kasteren SI, Campbell SJ, Serres S, Anthony DC, Sibson NR, Davis BG (2009) Glyconanoparticles allow pre-symptomatic in vivo imaging of brain disease. Proc Natl Acad Sci U S A 106:18–23
Hoyte LC, Brooks KJ, Nagel S et al (2010) Molecular magnetic resonance imaging of acute vascular cell adhesion molecule-1 expression in a mouse model of cerebral ischemia. J Cereb Blood Flow Metab 30:1178–1187
Breckwoldt MO, Chen JW, Stangenberg L et al (2008) Tracking the inflammatory response in stroke in vivo by sensing the enzyme myeloperoxidase. Proc Natl Acad Sci U S A 105:18584–18589
Zhang RL, Chopp M, Zaloga C et al (1995) The temporal profiles of ICAM-1 protein and mRNA expression after transient MCA occlusion in the rat. Brain Res 682:182–188
Shyu KG, Chang H, Lin CC (1997) Serum levels of intercellular adhesion molecule-1 and E-selectin in patients with acute ischaemic stroke. J Neurol 244:90–93
Everts M, Koning GA, Kok RJ et al (2003) In vitro cellular handling and in vivo targeting of E-selectin-directed immunoconjugates and immunoliposomes used for drug delivery to inflamed endothelium. Pharm Res 20:64–72
Koning GA, Morselt HW, Velinova MJ et al (1999) Selective transfer of a lipophilic prodrug of 5-fluorodeoxyuridine from immunoliposomes to colon cancer cells. Biochim Biophys Acta 1420:153–167
Rouser G, Fkeischer S, Yamamoto A (1970) Two dimensional then layer chromatographic separation of polar lipids and determination of phospholipids by phosphorus analysis of spots. Lipids 5:494–496
Wagner EF, Risau W (1994) Oncogenes in the study of endothelial cell growth and differentiation. Semin Cancer Biol 5:137–145
Perls M (1867) Nachweis von Eisenoxyd in gewissen Pigmenten. Virchows Archiv 39:42–48
Oude Engberink RD, Van der Pol SM, Dopp EA, De Vries HE, Blezer EL (2007) Comparison of SPIO and USPIO for in vitro labeling of human monocytes: MR detection and cell function. Radiology 243:467–474
Hata R, Mies G, Wiessner C et al (1998) A reproducible model of middle cerebral artery occlusion in mice: hemodynamic, biochemical, and magnetic resonance imaging. J Cereb Blood Flow Metab 18:367–375
Bouts MJ, Tiebosch IA, Zwartbol R, Hoogveld E, Wu O, Dijkhuizen RM (2011) Early prediction of salvageable tissue with multiparametric MRI-based algorithms after experimental ischemic stroke [abstract]. Proc Intl Soc Mag Reson Med 19:2141P
Zhu Y, Ling Y, Zhong J, Liu X, Wei K, Huang S (2012) Magnetic resonance imaging of radiation-induced brain injury using targeted microparticles of iron oxide. Acta Radiol 53:812–819
Serres S, Mardiguian S, Campbell SJ, et al (2011) VCAM-1-targeted magnetic resonance imaging reveals subclinical disease in a mouse model of multiple sclerosis. FASEB J 25:4415–4422
Montagne A, Gauberti M, Macrez R et al (2012) Ultra-sensitive molecular MRI of cerebrovascular cell activation enables early detection of chronic central nervous system disorders. NeuroImage 63:760–770
Yang Y, Yanasak N, Schumacher A, Hu TC (2010) Temporal and noninvasive monitoring of inflammatory-cell infiltration to myocardial infarction sites using micrometer-sized iron oxide particles. Magn Reson Med 63:33–40
Ye Q, Wu YL, Foley LM et al (2008) Longitudinal tracking of recipient macrophages in a rat chronic cardiac allograft rejection model with noninvasive magnetic resonance imaging using micrometer-sized paramagnetic iron oxide particles. Circulation 118:149–156
Vandeputte C, Thomas D, Dresselaers T et al (2011) Characterization of the inflammatory response in a photothrombotic stroke model by MRI: implications for stem cell transplantation. Mol Imaging Biol 13:663–671
Mulder WJ, Strijkers GJ, Habets JW et al (2005) MR molecular imaging and fluorescence microscopy for identification of activated tumor endothelium using a bimodal lipidic nanoparticle. FASEB J 19:2008–2010
Sipkins DA, Gijbels K, Tropper FD, Bednarski M, Li KC, Steinman L (2000) ICAM-1 expression in autoimmune encephalitis visualized using magnetic resonance imaging. J Neuroimmunol 104:1–9
Drummond DC, Meyer O, Hong K, Kirpotin DB, Papahadjopoulos D (1999) Optimizing liposomes for delivery of chemotherapeutic agents to solid tumors. Pharmacol Rev 51:691–743
Woodle MC, Matthay KK, Newman MS et al (1992) Versatility in lipid compositions showing prolonged circulation with sterically stabilized liposomes. Biochim Biophys Acta 1105:193–200
Van Tilborg GA, Mulder WJ, Van der Schaft DW et al (2008) Improved magnetic resonance molecular imaging of tumor angiogenesis by avidin-induced clearance of nonbound bimodal liposomes. Neoplasia 10:1459–1469
Paulis LE, Jacobs I, Van den Akker NM, et al. (2011) In vivo molecular MRI of ICAM-1 expression in murine cardiac ischemia/reperfusion using a liposomal nanoparticle [abstract]. Proc Intl Soc Mag Reson Med 19:1660P
Paulis LE, Jacobs I, van de Akker N et al (2012) Targeting of ICAM-1 on vascular endothelium under static and shear stress conditions using a liposomal Gd-based MRI contrast agent. J Nanobiotechnology 10:25
Abra RM, Hunt CA (1981) Liposome disposition in vivo. III. Dose and vesicle-size effects. Biochim Biophys Acta 666:493–503
Van Tilborg GA, Strijkers GJ, Pouget EM et al (2008) Kinetics of avidin-induced clearance of biotinylated bimodal liposomes for improved MR molecular imaging. Magn Reson Med 60:1444–1456
McAteer MA, Sibson NR, von Zur MC et al (2007) In vivo magnetic resonance imaging of acute brain inflammation using microparticles of iron oxide. Nat Med 13:1253–1258
Kok MB, Hak S, Mulder WJ, Van der Schaft DW, Strijkers GJ, Nicolay K (2009) Cellular compartmentalization of internalized paramagnetic liposomes strongly influences both T1 and T2 relaxivity. Magn Reson Med 61:1022–1032
Mastrobattista E, Storm G, van Bloois L et al (1999) Cellular uptake of liposomes targeted to intercellular adhesion molecule-1 (ICAM-1) on bronchial epithelial cells. Biochim Biophys Acta 1419:353–363
Van de Stolpe A, Van der Saag PT (1996) Intercellular adhesion molecule-1. J Mol Med (Berl) 74:13–33
Zhou W, Liesz A, Bauer H et al (2012) Postischemic brain infiltration of leukocyte subpopulations differs among murine permanent and transient focal cerebral ischemia models. Brain Pathol 23:34–44
Schilling M, Besselmann M, Leonhard C, Mueller M, Ringelstein EB, Kiefer R (2003) Microglial activation precedes and predominates over macrophage infiltration in transient focal cerebral ischemia: a study in green fluorescent protein transgenic bone marrow chimeric mice. Exp Neurol 183:25–33
Nkansah MK, Thakral D, Shapiro EM (2011) Magnetic poly(lactide-co-glycolide) and cellulose particles for MRI-based cell tracking. Magn Reson Med 65:1776–1785
Acknowledgments
We gratefully acknowledge funding from the Netherlands Organization for Scientific Research (NWO; VIDI 917.76.347) and the European Union’s Seventh Framework Programme (FP7/2007–2013) under grant agreements no. 201024 and no. 202213 (European Stroke Network).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deddens, L.H., van Tilborg, G.A.F., van der Toorn, A. et al. MRI of ICAM-1 Upregulation After Stroke: the Importance of Choosing the Appropriate Target-Specific Particulate Contrast Agent. Mol Imaging Biol 15, 411–422 (2013). https://doi.org/10.1007/s11307-013-0617-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-013-0617-z