Abstract
Purpose
Calculating the absorbed dose is important for the determination of risk and therapeutic benefit of internal radiation therapy. The aim of this study was to perform image-based absorbed dose calculation for critical organs during the first cycle of [177Lu]DKFZ-PSMA-617 therapy in a small cohort of patients with metastatic prostate cancer.
Procedures
Nine patients with a history of prostate cancer documented by histopathology and radiologic evidence of metastatic diseases underwent radioligand therapy with [177Lu]DKFZ-PSMA-617. Conjugated planar whole-body scintigraphies acquired at 0.5, 24, 48, 72, and 168 h post-injection were analyzed by regions of interest, and time-activity curves were generated for various organs. Cumulated activities and residence times were calculated by bi-exponential fit of the time-activity curves. Mean absorbed doses were finally estimated using OLINDA/EXM1.1™. Additionally, the uncertainty when omitting the last measurement (168 h p.i.) was studied.
Results
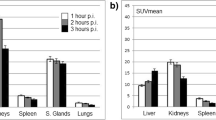
The following mean absorbed doses were calculated: 2.82 mGy/MBq for the lacrimal glands, 0.72 mGy/MBq for the salivary glands, 0.53 mGy/MBq for the kidneys, and 0.42 mGy/MBq for the nasal mucous membrane. Omitting the last measurement resulted in a mean deviation of 10 to 25 % for absorbed dose values as compared to the ones received by analyzing all measurements.
Conclusion
Absorbed organ doses of [177Lu]DKFZ-PSMA-617 therapy are not likely to be critical for kidneys, salivary glands, and the nasal mucous membrane. The lacrimal glands may represent the dose-limiting organs. Whole-body scintigraphy appears sufficient for dose estimation, but late measurements are mandatory, if accurate dose calculation is required.


Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136:E359–E386
Zechmann CM, Afashar-Oromieh A, Armor T et al (2014) Radiation dosimetry and first therapy results with a (124)I/ (131)I-labeled small molecule (MIP-1095) targeting PSMA for prostate cancer therapy. Eur J Nucl Med Mol Imaging 41:1280–1292
Bacich DJ, Pinto JT, Tong WP, Heston WD (2001) Cloning expression, genomic localization, and enzymatic activities of the mouse homolog of prostate-specific membrane antigen/NAALADase/folate hydrolase. Mamm Genome 12:117–123
Perner S, Hofer MD, Kim R et al (2007) Prostate-specific membrane antigen expression as a predictor of prostate cancer. Hum Pathol 38:696–701
Bradford TJ, Tomlins SA, Wang X, Chinnaiyan AM (2006) Molecular markers of prostate cancer. Urol Oncol 24:538–551
Bander NH, Milowsky MI, Nanus DM et al (2005) Phase I trial of 177lutetium-labeled J591, a monoclonal antibody to prostate-specific membrane antigen, in patients with androgen-independent prostate cancer. J Clin Oncol 23:4591–4601
Foss CA, Mease RC, Fan H et al (2005) Radiolabeled small-molecule ligands for prostate-specific membrane antigen: in vivo imaging in experimental models of prostate cancer. Clin Cancer Res 11:4022–4028
Humblet V, Lapidus R, Williams LR et al (2005) High-affinity near-infrared fluorescent small-molecule contrast agents for in vivo imaging of prostate-specific membrane antigen. Mol Imaging 4:448–462
Pomper MG, Musachio JL, Zhang J et al (2002) 11C-MCG: synthesis, uptake selectivity, and primate PET of a probe for glutamate carboxypeptidase II (NAALADase). Mol Imaging 1:96–101
Eder M, Schafer M, Bauder-Wust U et al (2012) 68Ga-complex lipophilicity and targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem 23:688–697
Afshar-Oromieh A, Haberkorn U, Hadaschik B et al (2013) PET/MRI with a 68Ga-PSMA ligand for the detection of prostate cancer. Eur J Nucl Med Mol Imaging 40:1629–1630
Eiber M, Nekolla SG, Maurer T et al (2014) Ga-PSMA PET/MR with multimodality image analysis for primary prostate cancer. Abdom Imaging 40:1769–1771
Tasch J, Gong M, Sadelain M, Heston WD (2001) A unique folate hydrolase, prostate-specific membrane antigen (PSMA): a target for immunotherapy? Crit Rev Immunol 21:249–261
Kratochwil C, Giesel FL, Eder M et al (2015) [177Lu]Lutetium-labelled PSMA ligand-induced remission in a patient with metastatic prostate cancer. Eur J Nucl Med Mol Imaging 42:987–988
Bé MM, Chisté V, Dulie C, et al (2004) Table of radionuclides (Vol. 2-A = 151 to 242). Bureau international des poids et mesures, Pavillon de Breteuil, Sèvres
Hindorf C, Glatting G, Chiesa C, Lindén O et al (2010) EANM dosimetry committee guidelines for bone marrow and whole-body dosimetry. Eur J Med Mol Imgaging 37:1238–1250
Stabin MG, Sparks RB, Crowe E (2005) OLINDA/EXM: the second-generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 46:1023–1027
Snyder WS, Fischer HL Jr, Ford MR, Warner GG (1969) Estimate of absorbed fractions for monoenergetic photon sources uniformly distributed in various organs of a heterogeneous phantom. MIRD pamphlet no. 5. J Nucl Med 10:7–52
Loevinger R, Budinger TF, Watson EE (1988) MIRD primer for absorbed dose calculations. Society of Nuclear Medicine, New York
Snyder WS, Cook MJ, Nasset ES, et al (1979) Report of the task group on reference man. ICRP Publication 23
Bukhari AA, Basheer NA, Joharjy HI (2014) Age, gender and interracial variability of normal lacrimal gland volume using MRI. Ophthal Plast Reconstr Surg 30:388–391
Bingham CM, Castro A, Realini T et al (2013) Calculated CT volumes of lacrimal glands in normal Caucasian orbits. Ophthal Plast Reconstr Surg 29:157–159
Garkavij M, Nickel M, Sjögreen-Gleisner K et al (2010) 177Lu-[DOTA0, Tyr3] Octreotate therapy in patients with disseminated neuroendocrine tumors: analysis of dosimetry with impact on future therapeutic strategy. Cancer 116:1084–1092
Sandström M, Garske U, Granberg D et al (2010) Individualized dosimetry in patients undergoing therapy with 177Lu-DOTA-D-Phe1-Tyr3-octreotate. Eur J Nucl Med Mol Imaging 37:212–225
Delker A, Fendler WP, Kratochwil C et al (2016) Dosimetry for 177Lu-DKFZ-PSMA-617: a new radiopharmaceutical for the treatment of metastatic prostate cancer. Eur J Nucl Med Mol Imaging 43:42–51
Weineisen M, Schottelius M, Simecek J et al (2015) 68Ga- and 177Lu-labeled PSMA I&T: optimization of a PSMA-targeted theranostic concept and first proof-of-concept human studies. J Nucl Med 56:1169–1176
Pandit-Taskar N, O’Donoghue JA, Durack JC et al (2015) A phase I/II study for analytic validation of 89Zr-J591 immunoPET as a molecular imaging agent for metastatic prostate cancer. Clin Cancer Res 21:5277–5285
Banerjee SR, Foss CA, Castanares M et al (2008) Synthesis and evaluation of technetium-99m- and rhenium-labeled inhibitors of the prostate-specific membrane antigen (PSMA). J Med Chem 51:4504–4517
Maurer T, Weirich G, Schottelius M et al (2015) Prostate-specific membrane antigen-radioguided surgery for metastatic lymph nodes in prostate cancer. Eur Urol 68:530–534
Benesova M, Schafer M, Bauder-Wust U et al (2015) Preclinical evaluation of a tailormade DOTA-conjugated PSMA inhibitor with optimized linker moiety for imaging and endoradiotherapy of prostate cancer. J Nucl Med 56:914–920
Ahmadzadehfar H, Rahbar K, Kurpig S et al (2015) Early side effects and first results of radioligand therapy with (177)Lu-DKFZ-617 PSMA of castrate-resistant metastatic prostate cancer: a two-centre study. EJNMMI Res. doi:10.1186/s13550-015-0114-2
Pfestroff A, Luster M, Jilg CA et al (2015) Current status and future perspectives of PSMA-targeted therapy in Europe: opportunity knocks. Eur J Nucl Med Mol Imaging 42:1971–1975
Parsons JT, Bova FJ, Mendenhall WM et al (1996) Response of the normal eye to high dose radiotherapy. Oncology 10:837–847
Buchali A, Schröder C, Sidow D, Blank E (1991) Influence of the radiation dose to salivary glands on xerostomia in patients with head and neck carcinomas. J Cancer Ther 4:188–194
Emami B, Lyman J, Brown A et al (1991) Tolerance of normal tissue to therapeutic irradiation. Int J Radiat Oncol Biol Phys 21:109–122
Yin G, Xiong G, Zhao C, Chen Y (2010) Damage of nasal mucociliary movement after intensity-modulated radiation therapy of nasopharyngeal carcinoma. Chin J Cancer 29:824–829
Larson M, Bernhardt P, Svensson JB et al (2012) Estimation of absorbed dose to the kidneys in patients with 177Lu-octreotate: comparison between methods based on planar scintigraphy. EJNMMI Res. doi:10.1186/2191-219X-2-49
Siegel JA, Thomas SR, Stubbs JB et al (1999) MIRD pamphlet no. 16: techniques for quantitative radiopharmaceutical biodistribution data acquisition and analysis for use in human radiation dose estimates. J Nucl Med 40:37S–61S
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hohberg, M., Eschner, W., Schmidt, M. et al. Lacrimal Glands May Represent Organs at Risk for Radionuclide Therapy of Prostate Cancer with [177Lu]DKFZ-PSMA-617. Mol Imaging Biol 18, 437–445 (2016). https://doi.org/10.1007/s11307-016-0942-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-016-0942-0