Abstract
Purpose
REM (rapid eye movement) sleep behavior disorder (RBD) is characterized by increased muscle tone and violent limb movements and is a usual symptom of the early stages of Parkinson’s disease (PD). PD patients with RBD represent faster motor and cognitive dysfunction progression. However, there are limited studies on possible structural brain changes underpinning this disorder.
Methods
Diffusion magnetic resonance imaging (DMRI) was used to assess whether microstructural abnormalities in the brain of 23 RBD positive PD are detectable compared to 31 RBD negative PD. DMRI scans were analyzed without a prior hypothesis. Diffusion MRI connectometry was used to carry out group analysis between age and gender matched PD patients with and without RBD. Diffusion MRI connectometry is based on spin distribution function (SDF) which quantifies the density of diffusing water and is more sensitive to psychological differences between groups.
Results
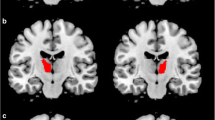
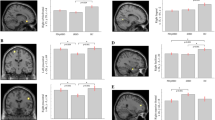
Patients with RBD positive showed microstructural white matter changes in the left and right cingulum, inferior front occipital fasciculus (IFOF), bilateral corticospinal tracts (CST), and middle cerebellar peduncles (MCPs), compared to patients without RBD.
Conclusions
White matter alterations in the cingulum, IFOF regions, and corpus callosum might explain faster cognitive deterioration in PD patients with RBD, in terms of visual recognition and visuospatial dysfunction and executive function. Disturbed brain structural tissue markers in CST in PD + RBD patients, could be justified in the light of faster motor progression in these patients.

Similar content being viewed by others
References
Munhoz RP et al (2015) Non-motor signs in Parkinson’s disease: a review. Arq Neuropsiquiatr 73(5):454–462
Postuma RB et al (2015) Risk factors for neurodegeneration in idiopathic rapid eye movement sleep behavior disorder: a multicenter study. Ann Neurol 77(5):830–839
Slow EJ, Postuma RB, Lang AE (2014) Implications of nocturnal symptoms towards the early diagnosis of Parkinson’s disease. J Neural Transm (Vienna) 121(Suppl 1):S49–S57
Postuma RB et al (2009) Markers of neurodegeneration in idiopathic rapid eye movement sleep behaviour disorder and Parkinson’s disease. Brain 132(Pt 12):3298–3307
Lim JS et al (2016) Neural substrates of rapid eye movement sleep behavior disorder in Parkinson’s disease. Parkinsonism Relat Disord 23:31–36
Zhang JR et al (2016) Rapid eye movement sleep behavior disorder symptoms correlate with domains of cognitive impairment in Parkinson’s disease. Chin Med J 129(4):379–385
Scherfler C et al (2011) White and gray matter abnormalities in idiopathic rapid eye movement sleep behavior disorder: a diffusion-tensor imaging and voxel-based morphometry study. Ann Neurol 69(2):400–407
Mitterling T, Högl B (2013) Magnetic resonance imaging in rapid eye movement sleep behavior disorder: diffusion tensor imaging and voxel-based morphometry. Sleep and Biological Rhythms 11(S1):52–55
Miyamoto M et al (2012) Preclinical substantia nigra dysfunction in rapid eye movement sleep behaviour disorder. Sleep Med 13(1):102–106
The Parkinson Progression Marker Initiative (PPMI) (2011) Prog Neurobiol 95(4):629–635. doi:10.1016/j.pneurobio.2011.09.005
Ford AH et al (2015) Rapid eye movement sleep behavior disorder in Parkinson’s disease: magnetic resonance imaging study. Mov Disord 28(6):832–836
Boucetta S et al (2016) Structural brain alterations associated with rapid eye movement sleep behavior disorder in Parkinson’s disease. Sci Rep 6:26782
Reinoso-Suarez F et al (2001) Brain structures and mechanisms involved in the generation of REM sleep. Sleep Med Rev 5(1):63–77
Yeh FC, Badre D, Verstynen T (2016) Connectometry: a statistical approach harnessing the analytical potential of the local connectome. Neuroimage. doi:10.1016/j.neuroimage.2015.10.053
Yeh FC et al (2013) Deterministic diffusion fiber tracking improved by quantitative anisotropy. PLoS One 8(11):e80713
Tatura R, Kraus T, Giese A, Arzberger T, Buchholz M, Hoglinger G, Muller U (2016) Parkinson’s disease: SNCA-, PARK2-, and LRRK2- targeting microRNAs elevated in cingulate gyrus. Parkinsonism Relat Disord. doi:10.1016/j.parkreldis.2016.09.028
Zheng Z et al (2014) DTI correlates of distinct cognitive impairments in Parkinson’s disease. Hum Brain Mapp 35(4):1325–1333
Yoo K et al (2015) Neural substrates of motor and non-motor symptoms in Parkinson’s disease: a resting FMRI study. PLoS One 10(4):e0125455
Kamagata K et al (2012) White matter alteration of the cingulum in Parkinson disease with and without dementia: evaluation by diffusion tensor tract-specific analysis. AJNR Am J Neuroradiol 33(5):890–895
Pena MCS et al (2008) Vsuospatial cognitive tests for the evaluation of patients with Parkinson’s disease. Dement Neuropsychol 2(3):201–205
Canu, E., et al., 2015 Brain structural and functional connectivity in Parkinson’s disease with freezing of gait. Hum Brain Mapp
Chen B et al (2015) Changes in anatomical and functional connectivity of Parkinson’s disease patients according to cognitive status. Eur J Radiol 84(7):1318–1324
Almairac F et al (2015) The left inferior fronto-occipital fasciculus subserves language semantics: a multilevel lesion study. Brain Struct Funct 220(4):1983–1995
Luo C et al (2016) Cortical thinning in drug-naive Parkinson’s disease patients with depression. J Neurol 263(10):2114–2119
Torterolo P et al (2015) Melanin-concentrating hormone (MCH): role in REM sleep and depression. Front Neurosci 9:475
Marelli, S., et al., 2016 National validation and proposed revision of REM sleep behavior disorder screening questionnaire (RBDSQ). J Neurol
Acknowledgments
This work was funded by grants from the Michael J Fox Foundation for Parkinson’s Research, the W Garfield Weston Foundation, and the Alzheimer’s Association, the Canadian Institutes for Health Research, and the Natural Sciences and Engineering Research Council of Canada. We thank Christian Beckmann and Simon Eickhoff for their advice on data analysis. Data used in this article were obtained from the Parkinson Progression Markers Initiative (PPMI) database (www.ppmi-info.org/data). For up-to-date information on the study, visit www.ppmi-info.org. PPMI is sponsored and partially funded by the Michael J Fox Foundation for Parkinson Research and funding partners, including AbbVie, Avid Radiopharmaceuticals, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline (GSK), Eli Lilly and Company, Lundbeck, Merck, Meso Scale Discovery (MSD), Pfizer, Piramal Imaging, Roche, Servier, and UCB (www.ppmi-info.org/fundingpartners).
Contribution
M.H.A., F. R and M.A. contributed to the conception and design of the study; M. A, F. R, M.D., A. P, and M.H.A. contributed to data collection and analysis; and M.A., M.D., A. P, F. R, and M.H.A. contributed to writing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this research.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed here, including human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Mina Ansari and Farzaneh Rahmani have equal contribution in this article.
Rights and permissions
About this article
Cite this article
Ansari, M., Rahmani, F., Dolatshahi, M. et al. Brain pathway differences between Parkinson’s disease patients with and without REM sleep behavior disorder. Sleep Breath 21, 155–161 (2017). https://doi.org/10.1007/s11325-016-1435-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-016-1435-8