Abstract
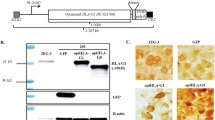
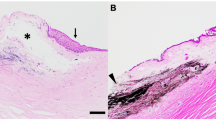
The use of a recombinant adeno-associated viral (rAAV) vector carrying endostatin gene as an anti-angiogenesis strategy to treat corneal neovascularization in a mouse model was evaluated. Subconjunctival injection of recombinant endostatin-AAV was used to examine the inhibition of corneal neovascularization induced by silver nitrate cauterization in mice. The results showed that gene expression in corneal tissue was observed as early as 4 days after gene transfer and stably lasted for over 8 months with minimal immune reaction. Subconjunctival injection of a high-titer rAAV-endostatin successfully inhibited neovascularization. Immunohistchemistry staining of CD 31 and endostatin showed that the treatment significantly inhibits angiogenesis in cornea. We concluded that the rAAV was capable of directly delivering genes to the ocular surface epithelium by way of subconjunctival injection and was able to deliver sustained, high levels of gene expression in vivo to inhibit angiogenesis.
Similar content being viewed by others
References
Chang J.H., Gabison E.E., Kato T., Azar D.T. Corneal neovascularization. Curr. Opin. Ophthalmol. 12: 242–249, 2001
Hill J.C., Maske R. An animal model for corneal graft rejection in high-risk keratoplasty. Transplantation 46: 26–30, 1988
BenEzra D., Griffin B.W., Maftzir G., Sharif N.A., Clark A.F. Topical formulations of novel angiostatic steroids inhibit rabbit corneal neovascularization. Invest. Ophthalmol. Vis. Sci. 38: 1954–1962, 1997
Boneham G.C., Collin H.B. Steroid inhibition of limbal blood and lymphatic vascular cell growth. Curr. Eye Res. 14: 1–10, 1995
Suzuki T., Sano Y., Kinoshita S. Effects of 1alpha,25-dihydroxyvitamin D3 on Langerhans cell migration and corneal neovascularization in mice. Invest. Ophthalmol. Vis. Sci. 41: 154–158, 2000
Dana M.R., Zhu S.N., Yamada J. Topical modulation of interleukin-1 activity in corneal neovascularization. Cornea 17: 403–409, 1998
Benelli U., Ross J.R., Nardi M., Klintworth G.K. Corneal neovascularization induced by xenografts or chemical cautery. Inhibition by cyclosporin A. Invest. Ophthalmol. Vis. Sci. 38: 274–282, 1997
Duenas Z., Torner L., Corbacho A.M., Ochoa A., Gutierrez-Ospina G., Lopez-Barrera F., Barrios F.A., Berger P., Martinez de la Escalera G., Clapp C. Inhibition of rat corneal angiogenesis by 16-kDa prolactin and by endogenous prolactin-like molecules. Invest. Ophthalmol. Vis. Sci. 40: 2498–2505, 1999
Gordon Y.J., Mann R.K., Mah T.S., Gorin M.B. Fluorescein-potentiated argon laser therapy improves symptoms and appearance of corneal neovascularization. Cornea 21: 770–773, 2002
Fossarello M., Peiretti E., Zucca I., Serra A. Photodynamic therapy of corneal neovascularization with verteporfin. Cornea 22: 485–488, 2003
Tsai R.J., Li L.M., Chen J.K. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N. Engl. J. Med. 343: 86–93, 2000
Ma D.H., Tsai R.J., Chu W.K., Kao C.H., Chen J.K. Inhibition of vascular endothelial cell morphogenesis in cultures by limbal epithelial cells. Invest. Ophthalmol. Vis. Sci. 40: 1822–1828, 1999
Ambati B.K., Joussen A.M., Ambati J., Moromizato Y., Guha C., Javaherian K., Gillies S., O’Reilly M.S, Adamis A.P. Angiostatin inhibits and regresses corneal neovascularization. Arch. Ophthalmol. 120: 1063–1068, 2002
Joussen A.M., Beecken W.D., Moromizato Y., Schwartz A., Kirchhof B., Poulaki V. Inhibition of inflammatory corneal angiogenesis by TNP-470. Invest. Ophthalmol. Vis. Sci. 42: 2510–2516, 2001
Kenyon B.M., Browne F., D’Amato R.J. Effects of thalidomide and related metabolites in a mouse corneal model of neovascularization. Exp. Eye Res. 64: 971–978, 1997
Cursiefen C., Rummelt C., Kuchle M. Immunohistochemical localization of vascular endothelial growth factor, transforming growth factor alpha, and transforming growth factor beta1 in human corneas with neovascularization. Cornea 19: 526–533, 2000
Amano S., Rohan R., Kuroki M., Tolentino M., Adamis A.P. Requirement for vascular endothelial growth factor in wound- and inflammation-related corneal neovascularization. Invest. Ophthalmol. Vis. Sci. 39: 18–22, 1998
Folkman J. New perspectives in clinical oncology from angiogenesis research. Eur. J. Cancer 32A: 2534–2539, 1996
Ma H.I., Lin S.Z., Chiang Y.H., Li J., Chen S.L., Tsao Y.P., Xiao X. Intratumoral gene therapy of malignant brain tumor in a rat model with angiostatin delivered by adeno-associated viral (AAV) vector. Gene Ther. 9: 2–11, 2002
Liekens S., De Clercq E., Neyts J. Angiogenesis: regulators and clinical applications. Biochem. Pharmacol. 61: 253–270, 2001
O’Reilly M.S., Boehm T., Shing Y., Fukai N., Vasios G., Lane W.S., Flynn E.B., Birkhead J.R., Olsen B.R., Folkman J. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88: 277–285, 1997
Hansma A.H., Broxterman H.J., van der Horst I., Yuana Y., Boven E., Giaccone G., Pinedo H.M., Hoekman K. Recombinant human endostatin administered as a 28 day continuous intravenous infusion, followed by daily subcutaneous injections: a phase 1 and pharmacokinetic study in patients with advanced cancer. Ann. Oncol. 16: 1695–1701, 2005
Liang Z.H., Wu P.H., Li L., Xue G., Zeng Y.X., Huang W.L. Inhibition of tumor growth in xenografted nude mice with adenovirus-mediated endostatin gene comparison with recombinant endostatin protein. Chin. Med. J. 117: 1809–1814, 2004
Luo X., Stater J.M., Gridley D.S. Enhancement of radiation effects by pXLG-mEndo in a lung carcinoma model. Int. J. Rad. Oncol. Biol. Phys. 63: 553–564, 2005
Wu Y., Yang L., Hu B., Liu J.Y., Su K.M., Luo Y., Dino Z.Y., Niu T., Li Q., Xie X.J., Wen Y.J., Tian L., Kan B., Mao Y.Q., Wei Y.Q. Synergistic anti-tumor effect of recombinant human endostatin adenovirus combined with gemcitabine. Anti-Cancer Drugs 16: 551–557, 2005
Uesato M., Gunji Y., Tomonaga T., Miyazaki S., Shiratori T., Matsubara H., Kouzu T., Shimada H., Nomura F., Ochiai T. Synergistic antitumor effect of antiangiogenic factor genes on colon 26 produced by low-voltage electroporation. Cancer Gene Ther 11: 625–632, 2004
Kurdow R., Boehle A.S., Ruhnke M., Mendoza R., Boenicke L., Sipos B., Schniewind B., Dohrmann P., Kalthoff H.H. Retroviral endostatin gene transfer inhibits growth of human lung cancer in a murine orthotopic xenotransplant model. Langenbecks Arch. Surg. 388: 401–405, 2003
Kurosaka D., Yoshida K., Yasuda J., Yokoyama T., Kingetsu I., Yamaguchi N., Joh K., Matsushima M., Saito S., Yamada A. Inhibition of arthritis by systemic administration of endostatin in passive murine collagen induced arthritis. Ann. Rheum. Dis. 62: 677–679, 2003
Jung S.P., Siegrist B., Hornick C.A., Wang Y.Z., Wade M.R., Anthony C.T., Woltering E.A. Effect of human recombinant Endostatin protein on human angiogenesis. Angiogenesis 5: 111–118, 2002
Thomas J.P., Arzoomanian R.Z., Alberti D., Marnocha R., Lee F., Friedl A., Tstsch K., Dresen A., Geiger P., Pluda J., Fogler W., Schiller J.H., Wilding G. Phase I pharmacokinetic and pharmacodynamic study of recombinant human endostatin in patients with advanced solid tumors. J. Clin. Oncol. 21: 223–231, 2003
Ren B., Hoti N., Rabasseda X., Wang Y.Z., Wu M. The antiangiogenic and therapeutic implications of endostatin. Methods Find Exp. Clin. Pharmacol. 25: 215–224, 2003
Xiao X., Li J., Samulski R.J. Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vector. J. Virol. 70: 8098–8108, 1996
Wang B., Li J., Xiao X. Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model. Proc. Natl. Acad. Sci. USA 97: 13714–13719, 2000
Li J., Dressman D., Tsao Y.P., Sakamoto A., Hoffman E.P., Xiao X. rAAV vector-mediated sarcogylcan gene transfer in a hamster model for limb girdle muscular dystrophy. Gene Ther. 6: 74–82, 1999
McCarty D.M., Monahan P.E., Samulski R.J. Self-complementary recombinant adeno-associated virus (scAAV) vectors promote efficient transduction independently of DNA synthesis. Gene Ther. 8: 1248–1254, 2001
Kaplitt M.G., Leone P., Samulski R.J., Xiao X., Pfaff D.W., O’Malley K.L., During M.J. Long-term gene expression and phenotypic correction using adeno-associated virus vectors in the mammalian brain. Nat. Genet. 8: 148–154, 1994
Duan D., Sharma P., Yang J., Yue Y., Dudus L., Zhang Y., Fisher K.J., Engelhardt J.F. Circular intermediates of recombinant adeno-associated virus have defined structural characteristics responsible for long-term episomal persistence in muscle tissue. J. Virol. 72: 8568–8577, 1998
Flotte T.R., Afione S.A., Zeitlin P.L. Adeno-associated virus vector gene expression occurs in nondividing cells in the absence of vector DNA integration. Am. J. Respir. Cell Mol. Biol. 11: 517–521, 1994
Xiao X., Li J., Samulski R.J. Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus. J. Virol. 72: 2224–2232, 1998
Ma H.I., Guo P., Li J., Lin S.Z., Chiang Y.H., Xiao X., Cheng S.Y. Suppression of intracranial human glioma growth after intramuscular administration of an adeno-associated viral vector expressing angiostatin. Cancer Res. 62: 756–763, 2002
Tsai M.L., Chen S.L., Chou P.I., Wen L.Y., Tsai R.J.F., Tsao Y.P. Inducible adenoassociated virus vector-delivered transgene expression in corneal endothelium. Invest. Ophthalmol. Vis. Sci. 43: 751–757, 2002
Tsubota K., Inoue H., Ando K., Ono M., Yoshino K., Saito I. Adenovirus-mediated gene transfer to the ocular surface epithelium. Exp. Eye Res. 67: 531–538, 1998
Lai C.M., Brankov M., Zaknich T., Lai Y.K., Shen W.Y., Constable I.J., Kovesdi I., Rakoczy P.E. Inhibition of angiogenesis by adenovirus-mediated sFlt-1 expression in a rat model of corneal neovascularization. Hum. Gene Ther. 12: 1299–1310, 2001
Abdollahi A., Hlatky L., Huber P.E. Endostatin: the logic of antiangiogenic therapy. Drug Resist Updat 8: 59–74, 2005
Auricchio A., Behling K.C., Maguire A.M., O’Connor E.M., Bennett J., Wilson J.M., Tolentino M.J. Inhibition of retinal neovascularization by intraocular viral-mediated delivery of anti-angiogenic agents. Mol. Ther. 6: 490–494, 2002
Wang Z., Zhu T., Rehman K.K., Bertera S., Zhang J., Chen C., Papworth G., Watkins S., Trucco M., Robbins P.D., Li J., Xiao X. Widespread and stable pancreatic gene transfer by adeno-associated virus vectors via different routes. Diabetes 55: 875–884, 2006
Berger A.C., Feldman A.L., Gnant M.F., Kruger E.A., Sim B.K., Hewitt S., Figg W.D., Alexander H.R., Libutti S.K. The angiogenesis inhibitor, endostatin, does not affect murine cutaneous wound healing. J. Surg. Res. 91: 26–31, 2000
Wang Z., Zhu T., Qiao C., Zhou L., Wang B., Zhang J., Chen C., Li J., Xiao X. Adeno-associated virus serotype 8 efficiently delivers genes to muscle and heart. Nat. Biotech. 23: 321–328, 2003
Acknowledgements
The authors acknowledge Dr. Shi-Yuan Cheng for kindly providing endostatin antibody for Immunohistochemistry stain. The authors thank Jian Zhang for the expert technical assistance. The authors thank Nicole for reviewing the paper. This work was supported in part by NIH grants 93-2314-B-182A-041.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lai, LJ., Xiao, X. & Wu, J.H. Inhibition of corneal neovascularization with endostatin delivered by adeno-associated viral (AAV) vector in a mouse corneal injury model. J Biomed Sci 14, 313–322 (2007). https://doi.org/10.1007/s11373-007-9153-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-007-9153-7