Abstract
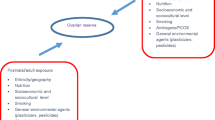
Primary ovarian insufficiency (POI) occurs in about 1% of female population under the age of 40, leading to reproductive problems, an earlier encounter with menopausal symptoms, and complicated diseases. There are three presumable mechanisms involved in the development of POI, namely apoptosis acceleration, follicular maturation blocking and premature follicle activation, through the following studied causes: (i) chromosomal abnormalities or gene mutations: mostly involve X chromosome, such as FMR1 premutation; more and more potentially causal genes have been screened recently; (ii) metabolic disorders such as classic galactosaemia and 17-OH deficiency; (iii) autoimmune mediated ovarian damage: observed alone or with some certain autoimmune disorders and syndromes; but the specificity and sensitivity of antibodies towards ovary are still questionable; (iv) iatrogenic: radiotherapy or chemotherapy used in cancer treatment, as well as pelvic surgery with potential threat to ovaries’ blood supply can directly damage ovarian function; (v) virus infection such as HIV and mumps; (vi) toxins and other environmental/lifestyle factors: cigarette smoking, toxins (e.g., 4-vinylcyclohexene diepoxide), and other environmental factors are associated with the development of POI. The etiology of a majority of POI cases is not identified, and is believed to be multifactorial. Strategies to POI include hormone replacement and infertility treatment. Assisted conception with donated oocytes has been proven to achieve pregnancy in POI women. Embryo cryopreservation, ovarian tissue cryopreservation and oocyte cryopreservation have been used to preserve ovarian reserve in women undergoing cancer treatments.
Article PDF
Similar content being viewed by others
References
Nelson L M. Clinical practice. Primary ovarian insufficiency. N Engl J Med, 2009, 360: 606–614
Cooper A R, Baker V L, Sterling E W, et al. The time is now for a new approach to primary ovarian insufficiency. Fertil Steril, 2011, 95: 1890–1897
van Noord P A, Dubas J S, Dorland M, et al. Age at natural menopause in a population-based screening cohort: the role of menarche, fecundity, and lifestyle factors. Fertil Steril, 1997, 68: 95–102
Coulam C B, Adamson S C, Annegers J F. Incidence of premature ovarian failure. Obstet Gynecol, 1986, 67: 604–606
Skillern A, Rajkovic A. Recent developments in identifying genetic determinants of premature ovarian failure. Sex Dev, 2008, 2: 228–243
Bachelot A, Rouxel A, Massin N, et al. Phenotyping and genetic studies of 357 consecutive patients presenting with premature ovarian failure. Eur J Endocrinol, 2009, 161: 179–187
Rebar R W, Erickson G F, Yen S S. Idiopathic premature ovarian failure: clinical and endocrine characteristics. Fertil Steril, 1982, 37: 35–41
Rebar R W, Connolly H V. Clinical features of young women with hypergonadotropic amenorrhea. Fertil Steril, 1990, 53: 804–810
Nelson L M, Anasti J N, Kimzey L M, et al. Development of luteinized graafian follicles in patients with karyotypically normal spontaneous premature ovarian failure. J Clin Endocrinol Metab, 1994, 79: 1470–1475
Bakalov V K, Anasti J N, Calis K A, et al. Autoimmune oophoritis as a mechanism of follicular dysfunction in women with 46,XX spontaneous premature ovarian failure. Fertil Steril, 2005, 84: 958–965
Santoro N. Update in hyper- and hypogonadotropic amenorrhea. J Clin Endocrinol Metab, 2011, 96: 3281–3288
Wellons M. Cardiovascular disease and primary ovarian insufficiency. Semin Reprod Med, 2011, 29: 328–341
Practice Committee of American Society for Reproductive Medicine. Current evaluation of amenorrhea. Fertil Steril, 2008, 90: S219–225
Younis J S. Ovarian aging and implications for fertility female health. Minerva Endocrinol, 2012, 37: 41–57
Goswami D, Conway G S. Premature ovarian failure. Hum Reprod Update, 2005, 11: 391–410
Morita Y, Tilly J L. Oocyte apoptosis: like sand through an hourglass. Dev Biol, 1999, 213: 1–17
Sullivan S D, Castrillon D H. Insights into primary ovarian insufficiency through genetically engineered mouse models. Semin Reprod Med, 2011, 29: 283–298
Adhikari D, Zheng W, Shen Y, et al. Tsc/mTORC1 signaling in oocytes governs the quiescence and activation of primordial follicles. Hum Mol Genet, 2010, 19: 397–410
Johnson J, Canning J, Kaneko T, et al. Germline stem cells and follicular renewal in the postnatal mammalian ovary. Nature, 2004, 428: 145–150
van Kasteren Y M, Hundscheid R D, Smits A P, et al. Familial idiopathic premature ovarian failure: an overrated and underestimated genetic disease? Hum Reprod, 1999, 14: 2455–2459
Davis C J, Davison R M, Payne N N, et al. Female sex preponderance for idiopathic familial premature ovarian failure suggests an X chromosome defect: opinion. Hum Reprod, 2000, 15: 2418–2422
Hunter J E, Epstein M P, Tinker SW, et al. Fragile X-associated primary ovarian insufficiency: evidence for additional genetic contributions to severity. Genet Epidemiol, 2008, 32: 553–559
Wittenberger M D, Hagerman R J, Sherman S L, et al. The FMR1 premutation and reproduction. Fertil Steril, 2007, 87: 456–465
Murabito J M, Yang Q, Fox C, et al. Heritability of age at natural menopause in the Framingham Heart Study. J Clin Endocrinol Metab, 2005, 90: 3427–3430
Simpson J L. Genetic and phenotypic heterogeneity in ovarian failure: overview of selected candidate genes. Ann N Y Acad Sci, 2008, 1135: 146–154
Dixit H, Rao L, Padmalatha V, et al. Genes governing premature ovarian failure. Reprod Biomed Online, 2010, 20: 724–740
Zinn A R. The X chromosome and the ovary. J Soc Gynecol Investig, 2001, 8: S34–36
Hens L, Devroey P, Van Waesberghe L, et al. Chromosome studies and fertility treatment in women with ovarian failure. Clin Genet, 1989, 36: 81–91
Kawano Y, Narahara H, Matsui N, et al. Premature ovarian failure associated with a Robertsonian translocation. Acta Obstet Gynecol Scand, 1998, 77: 467–469
Burton K A, Van Ee C C, Purcell K, et al. Autosomal translocation associated with premature ovarian failure. J Med Genet, 2000, 37: E2
Gleicher N, Weghofer A, Kim A, et al. The impact in older women of ovarian FMR1 genotypes and sub-genotypes on ovarian reserve. PLoS ONE, 2012, 7: e33638
Allen E G, Sullivan A K, Marcus M, et al. Examination of reproductive aging milestones among women who carry the FMR1 premutation. Hum Reprod, 2007, 22: 2142–2152
Murray A, Ennis S, Morton N. No evidence for parent of origin influencing premature ovarian failure in fragile X premutation carriers. Am J Hum Genet, 2000, 67: 253–254; author reply 256–258
Sullivan A K, Marcus M, Epstein M P, et al. Association of FMR1 repeat size with ovarian dysfunction. Hum Reprod, 2005, 20: 402–412
Sullivan S D, Welt C, Sherman S. FMR1 and the continuum of primary ovarian insufficiency. Semin Reprod Med, 2011, 29: 299–307
Allen E G, He W, Yadav-Shah M, et al. A study of the distributional characteristics of FMR1 transcript levels in 238 individuals. Hum Genet, 2004, 114: 439–447
Bretherick K L, Fluker M R, Robinson W P. FMR1 repeat sizes in the gray zone and high end of the normal range are associated with premature ovarian failure. Hum Genet, 2005, 117: 376–382
Bretherick K L, Hanna C W, Currie L M, et al. Estrogen receptor alpha gene polymorphisms are associated with idiopathic premature ovarian failure. Fertil Steril, 2008, 89: 318–324
Vujovic S. Aetiology of premature ovarian failure. Menopause Int, 2009, 15: 72–75
Otsuka F, McTavish K J, Shimasaki S. Integral role of GDF-9 and BMP-15 in ovarian function. Mol Reprod Dev, 2011, 78: 9–21
McNatty K P, Juengel J L, Reader K L, et al. Bone morphogenetic protein 15 and growth differentiation factor 9 co-operate to regulate granulosa cell function in ruminants. Reproduction, 2005, 129: 481–487
Dube J L, Wang P, Elvin J, et al. The bone morphogenetic protein 15 gene is X-linked and expressed in oocytes. Mol Endocrinol, 1998, 12: 1809–1817
Aaltonen J, Laitinen M P, Vuojolainen K, et al. Human growth differentiation factor 9 (GDF-9) and its novel homolog GDF-9B are expressed in oocytes during early folliculogenesis. J Clin Endocrinol Metab, 1999, 84: 2744–2750
Di Pasquale E, Beck-Peccoz P, Persani L. Hypergonadotropic ovarian failure associated with an inherited mutation of human bone morphogenetic protein-15 (BMP15) gene. Am J Hum Genet, 2004, 75: 106–111
Di Pasquale E, Rossetti R, Marozzi A, et al. Identification of new variants of human BMP15 gene in a large cohort of women with premature ovarian failure. J Clin Endocrinol Metab, 2006, 91: 1976–1979
Dixit H, Rao L K, Padmalatha V V, et al. Missense mutations in the BMP15 gene are associated with ovarian failure. Hum Genet, 2006, 119: 408–415
Laissue P, Christin-Maitre S, Touraine P, et al. Mutations and sequence variants in GDF9 and BMP15 in patients with premature ovarian failure. Eur J Endocrinol, 2006, 154: 739–744
Zhao Z Z, Painter J N, Palmer J S, et al. Variation in bone morphogenetic protein 15 is not associated with spontaneous human dizygotic twinning. Hum Reprod, 2008, 23: 2372–2379
Inagaki K, Shimasaki S. Impaired production of BMP-15 and GDF-9 mature proteins derived from proproteins WITH mutations in the proregion. Mol Cell Endocrinol, 2010, 328: 1–7
Spicer L J, Aad P Y, Allen D T, et al. Growth differentiation factor 9 (GDF9) stimulates proliferation and inhibits steroidogenesis by bovine theca cells: influence of follicle size on responses to GDF9. Biol Reprod, 2008, 78: 243–253
Orisaka M, Tajima K, Tsang B K, et al. Oocyte-granulosa-theca cell interactions during preantral follicular development. J Ovarian Res, 2009, 2: 9
Dong J, Albertini D F, Nishimori K, et al. Growth differentiation factor-9 is required during early ovarian folliculogenesis. Nature, 1996, 383: 531–535
Huang Q, Cheung A P, Zhang Y, et al. Effects of growth differentiation factor 9 on cell cycle regulators and ERK42/44 in human granulosa cell proliferation. Am J Physiol Endocrinol Metab, 2009, 296: E1344–1353
Galloway S M, McNatty K P, Cambridge L M, et al. Mutations in an oocyte-derived growth factor gene (BMP15) cause increased ovulation rate and infertility in a dosage-sensitive manner. Nat Genet, 2000, 25: 279–283
Hanrahan J P, Gregan S M, Mulsant P, et al. Mutations in the genes for oocyte-derived growth factors GDF9 and BMP15 are associated with both increased ovulation rate and sterility in Cambridge and Belclare sheep (Ovis aries). Biol Reprod, 2004, 70: 900–909
Nicol L, Bishop S C, Pong-Wong R, et al. Homozygosity for a single base-pair mutation in the oocyte-specific GDF9 gene results in sterility in Thoka sheep. Reproduction, 2009, 138: 921–933
Montgomery G W, Zhao Z Z, Marsh A J, et al. A deletion mutation in GDF9 in sisters with spontaneous DZ twins. Twin Res, 2004, 7: 548–555
Palmer J S, Zhao Z Z, Hoekstra C, et al. Novel variants in growth differentiation factor 9 in mothers of dizygotic twins. J Clin Endocrinol Metab, 2006, 91: 4713–4716
Pisarska M D, Kuo F T, Bentsi-Barnes I K, et al. LATS1 phosphorylates forkhead L2 and regulates its transcriptional activity. Am J Physiol Endocrinol Metab, 2010, 299: E101–109
Matthews C H, Borgato S, Beck-Peccoz P, et al. Primary amenorrhoea and infertility due to a mutation in the beta-subunit of follicle-stimulating hormone. Nat Genet, 1993, 5: 83–86
Aittomaki K, Lucena J L, Pakarinen P, et al. Mutation in the follicle-stimulating hormone receptor gene causes hereditary hypergonadotropic ovarian failure. Cell, 1995, 82: 959–968
Liu J Y, Gromoll J, Cedars M I, et al. Identification of allelic variants in the follicle-stimulating hormone receptor genes of females with or without hypergonadotropic amenorrhea. Fertil Steril, 1998, 70: 326–331
Takahashi K, Ozaki T, Okada M, et al. Increased prevalence of luteinizing hormone beta-subunit variant in patients with premature ovarian failure. Fertil Steril, 1999, 71: 96–101
Lofrano-Porto A, Barra G B, Giacomini L A, et al. Luteinizing hormone beta mutation and hypogonadism in men and women. N Engl J Med, 2007, 357: 897–904
Latronico A C, Chai Y, Arnhold I J, et al. A homozygous microdeletion in helix 7 of the luteinizing hormone receptor associated with familial testicular and ovarian resistance is due to both decreased cell surface expression and impaired effector activation by the cell surface receptor. Mol Endocrinol, 1998, 12: 442–450
Chand A L, Harrison C A, Shelling A N. Inhibin and premature ovarian failure. Hum Reprod Update, 2010, 16: 39–50
Prakash G J, Ravi Kanth V V, Shelling A N, et al. Mutational analysis of inhibin alpha gene revealed three novel variations in Indian women with premature ovarian failure. Fertil Steril, 2010, 94: 90–98
MacNaughton J, Banah M, McCloud P, et al. Age related changes in follicle stimulating hormone, luteinizing hormone, oestradiol and immunoreactive inhibin in women of reproductive age. Clin Endocrinol (Oxf), 1992, 36: 339–345
Petraglia F, Hartmann B, Luisi S, et al. Low levels of serum inhibin A and inhibin B in women with hypergonadotropic amenorrhea and evidence of high levels of activin A in women with hypothalamic amenorrhea. Fertil Steril, 1998, 70: 907–912
Soules M R, Battaglia D E, Klein N A. Inhibin and reproductive aging in women. Maturitas, 1998, 30: 193–204
Waggoner D D, Buist N R, Donnell G N. Long-term prognosis in galactosaemia: results of a survey of 350 cases. J Inherit Metab Dis, 1990, 13: 802–818
Schuh-Huerta S M, Johnson N A, Rosen M P, et al. Genetic variants and environmental factors associated with hormonal markers of ovarian reserve in Caucasian and African American women. Hum Reprod, 2012, 27: 594–608
Christin-Maitre S, Pasquier M, Donadille B, et al. Premature ovarian failure. Ann Endocrinol (Paris), 2006, 67: 557–566
Cordts E B, Christofolini D M, Dos Santos A A, et al. Genetic aspects of premature ovarian failure: a literature review. Arch Gynecol Obstet, 2011, 283: 635–643
Forges T, Monnier-Barbarino P, Leheup B, et al. Pathophysiology of impaired ovarian function in galactosaemia. Hum Reprod Update, 2006, 12: 573–584
Kaufman F R, Kogut M D, Donnell G N, et al. Hypergonadotropic hypogonadism in female patients with galactosemia. N Engl J Med, 1981, 304: 994–998
Rubio-Gozalbo M E, Gubbels C S, Bakker J A, et al. Gonadal function in male and female patients with classic galactosemia. Hum Reprod Update, 2010, 16: 177–188
Levy H L. Reproductive effects of maternal metabolic disorders: implications for pediatrics and obstetrics. Turk J Pediatr, 1996, 38: 335–344
Gubbels C S, Kuppens S M, Bakker J A, et al. Pregnancy in classic galactosemia despite undetectable anti-Mullerian hormone. Fertil Steril, 2009, 91: 1293–1296
Sanders R D, Spencer J B, Epstein M P, et al. Biomarkers of ovarian function in girls and women with classic galactosemia. Fertil Steril, 2009, 92: 344–351
Gubbels C S, Thomas C M, Wodzig W K, et al. FSH isoform pattern in classic galactosemia. J Inherit Metab Dis, 2011, 34: 387–390
Hoek A, Schoemaker J, Drexhage H A. Premature ovarian failure and ovarian autoimmunity. Endocr Rev, 1997, 18: 107–134
Smith J A, Vitale S, Reed G F, et al. Dry eye signs and symptoms in women with premature ovarian failure. Arch Ophthalmol, 2004, 122: 151–156
Husebye E S, Lovas K. Immunology of Addison’s disease and premature ovarian failure. Endocrinol Metab Clin North Am, 2009, 38: 389–405
Cheng M H, Nelson L M. Mechanisms and models of immune tolerance breakdown in the ovary. Semin Reprod Med, 2011, 29: 308–316
La Marca A, Brozzetti A, Sighinolfi G, et al. Primary ovarian insufficiency: autoimmune causes. Curr Opin Obstet Gynecol, 2010, 22: 277–282
Luborsky J. Ovarian autoimmune disease and ovarian autoantibodies. J Womens Health Gend Based Med, 2002, 11: 585–599
Dragojevic-Dikic S, Marisavljevic D, Mitrovic A, et al. An immunological insight into premature ovarian failure (POF). Autoimmun Rev, 2010, 9: 771–774
Donnez J, Dolmans M M, Pirard C, et al. Allograft of ovarian cortex between two genetically non-identical sisters: case report. Hum Reprod, 2007, 22: 2653–2659
Howard-Anderson J, Ganz P A, Bower J E, et al. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst, 2012, 104: 386–405
Fleischer R T, Vollenhoven B J, Weston G C. The effects of chemotherapy and radiotherapy on fertility in premenopausal women. Obstet Gynecol Surv, 2011, 66: 248–254
Clowse M E, Behera M A, Anders C K, et al. Ovarian preservation by GnRH agonists during chemotherapy: a meta-analysis. J Womens Health (Larchmt), 2009, 18: 311–319
Meirow D, Biederman H, Anderson R A, et al. Toxicity of chemotherapy and radiation on female reproduction. Clin Obstet Gynecol, 2010, 53: 727–739
Nicosia S V, Matus-Ridley M, Meadows A T. Gonadal effects of cancer therapy in girls. Cancer, 1985, 55: 2364–2372
Meirow D, Dor J, Kaufman B, et al. Cortical fibrosis and blood-vessels damage in human ovaries exposed to chemotherapy. Potential mechanisms of ovarian injury. Hum Reprod, 2007, 22: 1626–1633
Read M D, Edey K A, Hapeshi J, et al. The age of ovarian failure following premenopausal hysterectomy with ovarian conservation. Menopause Int, 2010, 16: 56–59
Amato P, Roberts A C. Transient ovarian failure: a complication of uterine artery embolization. Fertil Steril, 2001, 75: 438–439
Api M. Is ovarian reserve diminished after laparoscopic ovarian drilling? Gynecol Endocrinol, 2009, 25: 159–165
Prinz W, Taubert H D. Mumps in pubescent females and its effect on later reproductive function. Gynaecologia, 1969, 167: 23–27
Morrison J C, Givens J R, Wiser W L, et al. Mumps oophoritis: a cause of premature menopause. Fertil Steril, 1975, 26: 655–659
Cejtin H E, Kalinowski A, Bacchetti P, et al. Effects of human immunodeficiency virus on protracted amenorrhea and ovarian dysfunction. Obstet Gynecol, 2006, 108: 1423–1431
Ohl J, Partisani M, Demangeat C, et al. Alterations of ovarian reserve tests in Human Immunodeficiency Virus (HIV)-infected women. Gynecol Obstet Fertil, 2010, 38: 313–317
Hoyer P B, Devine P J, Hu X, et al. Ovarian toxicity of 4-vinylcyclohexene diepoxide: a mechanistic model. Toxicol Pathol, 2001, 29: 91–99
Christin-Maitre S, Ronci-Chaix N, Bouchard P. Ovary genes and molecular pathology. J Soc Biol, 2002, 196: 207–216
De Miguel M P, Cheng L, Holland E C, et al. Dissection of the c-Kit signaling pathway in mouse primordial germ cells by retroviral-mediated gene transfer. Proc Natl Acad Sci USA, 2002, 99: 10458–10463
Kappeler C J, Hoyer P B. 4-vinylcyclohexene diepoxide: a model chemical for ovotoxicity. Syst Biol Reprod Med, 2012, 58: 57–62
Di Prospero F, Luzi S, Iacopini Z. Cigarette smoking damages women’s reproductive life. Reprod Biomed Online, 2004, 8: 246–247
Chang S H, Kim C S, Lee K S, et al. Premenopausal factors influencing premature ovarian failure and early menopause. Maturitas, 2007, 58: 19–30
Kinney A, Kline J, Kelly A, et al. Smoking, alcohol and caffeine in relation to ovarian age during the reproductive years. Hum Reprod, 2007, 22: 1175–1185
Nardo L G, Christodoulou D, Gould D, et al. Anti-Mullerian hormone levels and antral follicle count in women enrolled in in vitro fertilization cycles: relationship to lifestyle factors, chronological age and reproductive history. Gynecol Endocrinol, 2007, 23: 486–493
Matikainen T, Perez G I, Jurisicova A, et al. Aromatic hydrocarbon receptor-driven Bax gene expression is required for premature ovarian failure caused by biohazardous environmental chemicals. Nat Genet, 2001, 28: 355–360
Sharara F I, Seifer D B, Flaws J A. Environmental toxicants and female reproduction. Fertil Steril, 1998, 70: 613–622
Klein P, Serje A, Pezzullo J C. Premature ovarian failure in women with epilepsy. Epilepsia, 2001, 42: 1584–1589
Testa G, Chiaffarino F, Vegetti W, et al. Case-control study on risk factors for premature ovarian failure. Gynecol Obstet Invest, 2001, 51: 40–43
Proetto Menopausa Italia Study Group, et al. Premature ovarian failure: frequency and risk factors among women attending a network of menopause clinics in Italy. BJOG, 2003, 110: 59–63
Henzl M R, Loomba P K. Transdermal delivery of sex steroids for hormone replacement therapy and contraception. A review of principles and practice. J Reprod Med, 2003, 48: 525–540
Jones S C. Subcutaneous estrogen replacement therapy. J Reprod Med, 2004, 49: 139–142
Nelson H D. Commonly used types of postmenopausal estrogen for treatment of hot flashes: scientific review. JAMA, 2004, 291: 1610–1620
Popat V B, Vanderhoof V H, Calis K A, et al. Normalization of serum luteinizing hormone levels in women with 46,XX spontaneous primary ovarian insufficiency. Fertil Steril, 2008, 89: 429–433
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Jin, M., Yu, Y. & Huang, H. An update on primary ovarian insufficiency. Sci. China Life Sci. 55, 677–686 (2012). https://doi.org/10.1007/s11427-012-4355-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-012-4355-2