Abstract
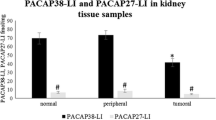
Pituitary adenylate cyclase activating polypeptide (PACAP) is a pleiotropic and multifunctional neuropeptide having important roles in various physiological processes. Recent trends in PACAP research point to the clinical introduction of PACAP or its analogs/fragments possibly in the near future. Recently, we have shown the presence of PACAP in human plasma, milk, placenta, and follicular fluid samples. However, relatively few data are available on PACAP in human tissues from patients with different disorders. The aim of the present study was to determine, by radioimmunoassay, the tissue level of PACAP38-like immunoreactivity (LI) and PACAP27-LI in different primary non-small cell lung cancer, colon tumor samples, and in cardiac muscle samples from patients suffering from ischemic heart disease and valvular disorders. We also labeled the PAC1 receptors in human cardiac cells. All samples showed significantly higher PACAP38-LI compared with PACAP27-LI. We found significantly lower levels of PACAP38-LI and PACAP27-LI in tumoral and peripheral samples compared with normal healthy tissue in both lung and colon cancers. Further investigations are necessary to describe the exact function of PACAP in oncogenesis. We showed that PACAP38-LI and PACAP27-LI are significantly higher in ischemic heart diseases compared with valvular abnormalities, suggesting that PACAP might play a role in ischemic heart disorders.




Similar content being viewed by others
References
Atlasz T, Szabadfi K, Kiss P et al (2010) Pituitary adenylate cyclase activating polypeptide in the retina: focus on the retinoprotective effects. Ann N Y Acad Sci 1200:128–139
Borzsei R, Mark L, Tamas A et al (2009) Presence of pituitary adenylate cyclase activating polypeptide-38 in human plasma and milk. Eur J Endocrinol 160:561–565
Botia B, Basille M, Allais A et al (2007) Neurotrophic effects of PACAP in the cerebellar cortex. Peptides 28:1746–1752
Bourgault S, Chatenet D, Wurtz O et al (2011) Strategies to convert PACAP from a hypophysiotropic neurohormone into a neuroprotective drug. Curr Pharm Des 17:1002–10024
Brubel R, Boronkai A, Reglodi D et al (2010) Changes in the expression of pituitary adenylate cyclase-activating polypeptide in the human placenta during pregnancy and its effects on the survival of JAR choriocarcinoma cells. J Mol Neurosci 42:450–458
Brubel R, Reglodi D, Jambor E et al (2011) Investigation of pituitary adenylate cyclase activating polypeptide in human gynecological and other biological fluids by using MALDI TOF mass spectrometry. J Mass Spectrom 46:189–194
Busto R, Carrero I, Guijarro LG et al (1999) Expression, pharmacological, and functional evidence for PACAP/VIP receptors in human lung. Am J Physiol 277(1 Pt 1):L42–L48
Busto R, Prieto JC, Bodega G, Zapatero J, Carrero I (2000) Immunohistochemical localization and distribution of VIP/PACAP receptors in human lung. Peptides 21:265–269
Busto R, Prieto JC, Bodega G, Zapatero J, Fogue L, Carrero I (2003) VIP and PACAP receptors coupled to adenylyl cyclase in human lung cancer: a study in biopsy specimens. Peptides 24:429–436
Calupca MA, Vizzard MA, Parsons RL (2000) Origin of pituitary adenylate cyclase-activating polypeptide (PACAP)-immunoreactive fibers innervating guinea pig parasympathetic cardiac ganglia. J Comp Neurol 423:26–39
Chang Y, Lawson LJ, Hancock JC, Hoover DB (2005) Pituitary adenylate cyclase-activating polypeptide: localization and differential influence on isolated hearts from rats and guinea pigs. Regul Pept 129:139–146
DeHaven WI, Cuevas J (2002) Heterogeneity of pituitary adenylate cyclase-activating polypeptide and vasoactive intestinal polypeptide receptors in rat intrinsic cardiac neurons. Neurosci Lett 328:45–49
Dejda A, Jolivel V, Bourgault S et al (2008) Inhibitory effect of PACAP on caspase activity in neuronal apoptosis: a better understanding towards therapeutic applications in neurodegenerative diseases. J Mol Neurosci 36:26–37
Delgado M, Ganea D (2001) Vasoactive intestinal peptide and pituitary adenylate cyclase activating polypeptide inhibit expression of Fas ligand in activated T lymphocytes by regulating c-Myc, NF-κB, NF-AT, and early growth factors 2/3. J Immunol 166:1028–1040
Draoui M, Hida T, Jakowlew S, Birrer M, Zia F, Moody TW (1996) PACAP stimulates c-fos mRNAs in small cell lung cancer cells. Life Sci 59:307–313
Dvorakova MC (2005) Cardioprotective role of the VIP signaling system. Drug News Perspect 18:387–391
Gasz B, Racz B, Roth E et al (2006a) PACAP inhibits oxidative stress-induced activation of MAP kinase dependent apoptotic pathway in cultured cardiomyocytes. Ann N Y Acad Sci 1070:293–297
Gasz B, Racz B, Roth E et al (2006b) Pituitary adenylate cyclase activating polypeptide protects cardiomyocytes against oxidative stress-induced apoptosis. Peptides 27:87–94
Godlewski J, Lakomy IM (2010) Changes in vasoactive intestinal peptide, pituitary adenylate cyclase-activating polypeptide and neuropeptide Y-ergic structures of the enteric nervous system in the carcinoma of the human large intestine. Folia Histochem Cytobiol 48:208–216
Hannibal J, Ekblad E, Mulder H, Sundler F, Fahrenkrug J (1998) Pituitary adenylate cyclase activating polypeptide (PACAP) in the gastrointestinal tract of the rat: distribution and effects of capsaicin or denervation. Cell Tissue Res 291:65–79
Hardwick JC, Tompkins JD, Locknar SA, Merriam LA, Young BA, Parsons RL (2006) Calcium influx through channels other than voltage-dependent calcium channels is critical to the pituitary adenylate cyclase-activating polypeptide-induced increase in excitability in guinea pig cardiac neurons. Ann N Y Acad Sci 1070:317–321
Hirose M, Leatmanoratn Z, Laurita KR, Carlson MD (2001) Effects of pituitary adenylate cyclase-activating polypeptide on canine atrial electrophysiology. Am J Physiol Heart Circ Physiol 281:H1667–H1674
Jakab B, Reglodi D, Jozsa R et al (2004) Distribution of PACAP-38 in the central nervous system of various species determined by a novel radioimmunoassay. J Biochem Biophys Methods 61:189–198
Koppan M, Varnagy A, Reglodi D et al (2012) Correlation between oocyte number and follicular fluid concentration of pituitary adenylate cyclase-activating polypeptide (PACAP) in women after superovulation treatment. J Mol Neurosci (in press) doi:10.1007/s12031-012-9743-3
Lauff JM, Modlin IM, Tang LH (1999) Biological relevance of pituitary adenylate cyclase-activating polypeptide (PACAP) in the gastrointestinal tract. Regul Pept 84:1–12
Le SV, Yamaguchi DJ, McArdle CA, Tachiki K, Pisegna JR, Germano P (2002) PAC1 and PACAP expression, signaling, and effect on the growth of HCT8, human colonic tumor cells. Regul Pept 109:115–125
Levy A, Gal R, Granoth R, Dreznik Z, Fridkin M, Gozes I (2002) In vitro and in vivo treatment of colon cancer by VIP antagonists. Regul Pept 109:127–133
Luts A, Uddman R, Alm P, Basterra J, Sundler F (1993) Peptide-containing nerve fibres in human airways: distribution and coexistence pattern. Int Arch Allergy Immunol 101:52–60
Miampamba M, Germano PM, Arli S et al (2002) Expression of pituitary adenylate cyclase-activating polypeptide and PACAP type 1 receptor in the rat gastric and colonic myenteric neurons. Regul Pept 105:145–154
Moody TW, Zia F, Makheja A (1993) Pituitary adenylate cyclase activating polypeptide receptors are present on small cell lung cancer cells. Peptides 14:241–246
Moody TW, Walters J, Casibang M, Zia F, Gozes Y (2000) VPAC1 receptors and lung cancer. Ann N Y Acad Sci 921:26–32
Moody TW, Leyton J, Casibang M, Pisegna J, Jensen RT (2002) PACAP-27 tyrosine phosphorylates mitogen activated protein kinase and increases VEGF mRNAs in human lung cancer cells. Regul Pept 109:135–140
Moody TW, Osefo N, Nuche-Berenguer B, Ridnour L, Wink D, Jensen RT (2012) Pituitary adenylate cyclase activating polypeptide causes tyrosine phosphorylation on the EGF receptor in lung cancer cells. J Pharmacol Exp Ther 341:873–881
Mori H, Nakamachi T, Ohtaki H et al (2010) Cardioprotective effect of endogenous pituitary adenylate cyclase-activating polypeptide on doxorubicin-induced cardiomyopathy in mice. Circ J 74:1183–1190
Naruse S, Suzuki T, Ozaki T, Nokihara K (1993) Vasodilator effect of pituitary adenylate cyclase activating polypeptide (PACAP) on femoral blood flow in dogs. Peptides 14:505–510
Nemetz N, Abad C, Lawson G et al (2008) Induction of colitis and rapid development of colorectal tumors in mice deficient in the neuropeptide PACAP. Int J Cancer 122:1803–1809
Otto C, Hein L, Brede M et al (2004) Pulmonary hypertension and right heart failure in pituitary adenylate cyclase-activating polypeptide type I receptor-deficient mice. Circulation 110:3245–3251
Parsons RL, Rossignol TM, Calupca MA, Hardwick JC, Brass KM (2000) PACAP peptides modulate guinea pig cardiac neuron membrane excitability and neuropeptide expression. Ann N Y Acad Sci 921:202–210
Portbury AL, McConalogue K, Furness JB, Young HM (1995) Distribution of pituitary adenylyl cyclase activating peptide (PACAP) immunoreactivity in neurons of the guinea-pig digestive tract and their projections in the ileum and colon. Cell Tissue Res 279:385–392
Racz B, Gasz B, Borsiczky B et al (2007) Protective effects of pituitary adenylate cyclase activating polypeptide in endothelial cells against oxidative stress-induced apoptosis. Gen Comp Endocrinol 153:115–123
Racz B, Gasz B, Gallyas F Jr et al (2008) PKA-Bad-14-3-3 and Akt-Bad-14-3-3 signaling pathways are involved in the protective effects of PACAP against ischemia/reperfusion-induced cardiomyocyte apoptosis. Regul Pept 145:105–115
Racz B, Reglodi D, Horvath G et al (2010) Protective effect of PACAP against doxorubicin-induced cell death in cardiomyocyte culture. J Mol Neurosci 42:419–427
Reglodi D, Gyarmati J, Ertl T et al (2010) Alterations of pituitary adenylate cyclase-activating polypeptide-like immunoreactivity in the human plasma during pregnancy and after birth. J Endocrinol Invest 33:443–445
Reglodi D, Kiss P, Lubics A, Tamas A (2011) Review on the protective effects of PACAP in models of neurodegenerative diseases in vitro and in vivo. Curr Pharm Des 17:962–972
Reubi JC (2000) In vitro evaluation of VIP/PACAP receptors in healthy and diseased human tissues. Clinical implications. Ann N Y Acad Sci 921:1–25
Reubi JC, Laderach U, Waser B, Gebbers JO, Robberecht P, Laissue JA (2000) Vasoactive intestinal peptide/pituitary adenylate cyclase-activating peptide receptor subtypes in human tumors and their tissues of origin. Cancer Res 60:3105–3112
Roth E, Weber G, Kiss P et al (2009) Effects of PACAP and preconditioning against ischemia/reperfusion-induced cardiomyocyte apoptosis in vitro. Ann N Y Acad Sci 1163:512–516
Sano H, Miyata A, Horio T, Nishikimi T, Matsuo H, Kangawa K (2002) The effect of pituitary adenylate cyclase activating polypeptide on cultured rat cardiocytes as a cardioprotective factor. Regul Pept 109:107–113
Schoenfeld LK, Souder JA, Hardwick JC (2000) Pituitary adenylate cyclase-activating polypeptide innervation of the mudpuppy cardiac ganglion. Brain Res 882:180–190
Seaborn T, Masmoudi-Kouli O, Fournier A, Vaudry H, Vaudry D (2011) Protective effects of pituitary adenylate cyclase-activating polypeptide (PACAP) against apoptosis. Curr Pharm Des 17:204–214
Somogyvari-Vigh A, Reglodi D (2004) Pituitary adenylate cyclase activating polypeptide: a potential neuroprotective peptide. Curr Pharm Des 10:2861–2889
Sundler F, Ekblad E, Absood A, Hakanson R, Koves K, Arimura A (1992) Pituitary adenylate cyclase activating peptide: a novel vasoactive intestinal peptide-like neuropeptide in the gut. Neuroscience 46:439–454
Szakaly P, Kiss P, Lubics A et al (2008) Effects of PACAP on survival and renal morphology in rats subjected to renal ischemia–reperfusion. J Mol Neurosci 36:89–96
Ushiyama M, Sugawara H, Inoue K, Kangawa K, Yamada K, Miyata A (2006) Characterization of the PAC1 variants expressed in the mouse heart. Ann N Y Acad Sci 1070:586–590
Vaudry D, Falluel-Morel A, Bourgault S et al (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61:283–357
Wei Y, Mojsov S (1996) Multiple human receptors for pituitary adenylyl cyclase-activating polypeptide and vasoactive intestinal peptide are expressed in a tissue-specific manner. Ann N Y Acad Sci 805:624–627
Zia F, Fagarasan M, Bitar K et al (1995) Pituitary adenylate cyclase activating peptide receptors regulate the growth of non-small cell lung cancer cells. Cancer Res 55:4886–4891
Acknowledgments
This study was supported by the following grants: OTKA (K72592, 75965, CNK78480), TAMOP (4.2.1.B-10/2/KONV-2010-002, 4.2.2.B-10/1-2010-0029), Bolyai Scholarship, Richter Foundation, PTE AOK Research Grant KA-4039/10-26, PTE-MTA “Lendulet” Program. The authors also thank all the volunteers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Szanto, Z., Sarszegi, Z., Reglodi, D. et al. PACAP Immunoreactivity in Human Malignant Tumor Samples and Cardiac Diseases. J Mol Neurosci 48, 667–673 (2012). https://doi.org/10.1007/s12031-012-9815-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9815-4