Abstract
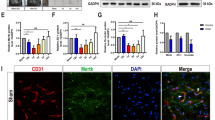
Stromal cell-derived factor-1 alpha (SDF-1a) or CXCL12 is an important cytokine with multiple functions in the brain during development and in adulthood. The inflammatory response initiated by spinal cord injury (SCI) involves the processing of interleukin-1beta (IL-1ß) and IL-18 mediated by caspase-1 which is under the control of an intracellular multiprotein complex termed inflammasome. Using an SCI rat model, we found improved functional long-term recovery which is paralleled by a reduction of apoptosis after intrathecal treatment with SDF-1a. An intriguing aspect is that SDF-1a changed the number of neuroinflammatory cells in the damaged area. We further examined the cellular localization and sequential expression of several inflammasomes during SCI at 6 h, 24 h, 3 days, and 7 days as well as the role of SDF-1a as a regulatory factor for inflammasomes. Using 14-week old male Wistar rats, spinal cord contusion was applied at the thoracic segment 9, and animals were subsequently treated with SDF-1a via intrathecal application through an osmotic pump. SCI temporally increased the expression of the inflammasomes NLRP3, ASC, the inflammatory marker tumor necrosis factor-a (TNF-a), interleukin-1ß (IL-1β) and IL-18. SDF-1a significantly reduced the levels of IL-18, IL-1b, TNF-a, NLRP3, ASC, and caspase-1. Immunofluorescence double-labeling demonstrated that microglia and neurons are major sources of the ASC and NLRP3 respectivley. Our data provide clear evidence that SCI stimulates a complex scenario of inflammasome activation at the injured site and that SDF-1a-mediated neuroprotection presumably depends on the attenuation of the inflammasome complex.







Similar content being viewed by others
References
Thuret S, Moon LD, Gage FH (2006) Therapeutic interventions after spinal cord injury. Nat Rev Neurosci 7(8):628–643
Schwab ME, Bartholdi D (1996) Degeneration and regeneration of axons in the lesioned spinal cord. Physiol Rev 76(2):319–370
Schroder K, Tschopp J (2010) The inflammasomes. Cell 140(6):821–832
de Zoete MR, Palm NW, Zhu S, Flavell RA (2014) Inflammasomes. Cold Spring Harbor perspectives in biology:a016287
Petrilli V, Papin S, Dostert C, Mayor A, Martinon F, Tschopp J (2007) Activation of the NALP3 inflammasome is triggered by low intracellular potassium concentration. Cell Death Differ 14(9):1583–1589
de Rivero Vaccari JP, Lotocki G, Marcillo AE, Dietrich WD, Keane RW (2008) A molecular platform in neurons regulates inflammation after spinal cord injury. J Neurosci 28(13):3404–3414
Liang F, Li C, Gao C, Li Z, Yang J, Liu X, Wang Y (2015) Effects of hyperbaric oxygen therapy on NACHT domain-leucine-rich-repeat-and pyrin domain-containing protein 3 inflammasome expression in rats following spinal cord injury. Mol Medi Rep 11(6):4650–4656
Aiuti A, Webb I, Bleul C, Springer T, Gutierrez-Ramos J (1997) The chemokine SDF-1 is a chemoattractant for human CD34+ hematopoietic progenitor cells and provides a new mechanism to explain the mobilization of CD34+ progenitors to peripheral blood. J Exp Med 185(1):111–120
Cui L, Qu H, Xiao T, Zhao M, Jolkkonen J, Zhao C (2013) Stromal cell-derived factor-1 and its receptor CXCR4 in adult neurogenesis after cerebral ischemia. Restor Neurol Neurosci 31(3):239–251
Shyu W-C, Lin S-Z, Yen P-S, Su C-Y, Chen D-C, Wang H-J, Li H (2008) Stromal cell-derived factor-1α promotes neuroprotection, angiogenesis, and mobilization/homing of bone marrow-derived cells in stroke rats. J Pharmacol Exp Ther 324(2):834–849
Jaerve A, Bosse F, Müller HW (2012) SDF-1/CXCL12: its role in spinal cord injury. Int J Biochem Cell Biol 44(3):452–456
Opatz J, Küry P, Schiwy N, Järve A, Estrada V, Brazda N, Bosse F, Müller HW (2009) SDF-1 stimulates neurite growth on inhibitory CNS myelin. Mol Cell Neurosci 40(2):293–300
Jaerve A, Schiwy N, Schmitz C, Mueller HW (2011) Differential effect of aging on axon sprouting and regenerative growth in spinal cord injury. Exp Neurol 231(2):284–294
Zendedel A, Nobakht M, Bakhtiyari M, Beyer C, Kipp M, Baazm M, Joghataie MT (2012) Stromal cell-derived factor-1 alpha (SDF-1α) improves neural recovery after spinal cord contusion in rats. Brain Res 1473:214–226
Zhang Y-K, Liu J-T, Peng Z-W, Fan H, Yao A-H, Peng C, Liu L, Ju G et al (2013) Different TLR4 expression and microglia/macrophage activation induced by hemorrhage in the rat spinal cord after compressive injury. J Neuroinflammation 10:112
Yaksh TL, Rudy TA (1976) Chronic catheterization of the spinal subarachnoid space. Physiol Behav 17(6):1031–1036
Nyström B, Berglund JE (1988) Spinal cord restitution following compression injuries in rats. Acta Neurol Scand 78(6):467–472
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21
Zendedel A, Habib P, Dang J, Lammerding L, Hoffmann S, Beyer C, Slowik A (2015) Omega-3 polyunsaturated fatty acids ameliorate neuroinflammation and mitigate ischemic stroke damage through interactions with astrocytes and microglia. J Neuroimmunol 278:200–211
Clarner T, Diederichs F, Berger K, Denecke B, Gan L, Van der Valk P, Beyer C, Amor S et al (2012) Myelin debris regulates inflammatory responses in an experimental demyelination animal model and multiple sclerosis lesions. Glia 60(10):1468–1480
Kucia M, Jankowski K, Reca R, Wysoczynski M, Bandura L, Allendorf DJ, Zhang J, Ratajczak J et al (2004) CXCR4–SDF-1 signalling, locomotion, chemotaxis and adhesion. J Mol Histol 35(3):233–245
Gensel JC, Zhang B (2015) Macrophage activation and its role in repair and pathology after spinal cord injury. Brain research
Nagasawa T (2014) CXC chemokine ligand 12 (CXCL12) and its receptor CXCR4. J Mol Med 92(5):433–439
Takeuchi H, Natsume A, Wakabayashi T, Aoshima C, Shimato S, Ito M, Ishii J, Maeda Y et al (2007) Intravenously transplanted human neural stem cells migrate to the injured spinal cord in adult mice in an SDF-1-and HGF-dependent manner. Neurosci Lett 426(2):69–74
Carbajal KS, Schaumburg C, Strieter R, Kane J, Lane TE (2010) Migration of engrafted neural stem cells is mediated by CXCL12 signaling through CXCR4 in a viral model of multiple sclerosis. Proc Natl Acad Sci 107(24):11068–11073
Raman D, Milatovic S-Z, Milatovic D, Splittgerber R, Fan G-H, Richmond A (2011) Chemokines, macrophage inflammatory protein-2 and stromal cell-derived factor-1α, suppress amyloid β-induced neurotoxicity. Toxicol Appl Pharmacol 256(3):300–313
Mortazavi MM, Verma K, Harmon OA, Griessenauer CJ, Adeeb N, Theodore N, Tubbs RS (2015) The microanatomy of spinal cord injury: a review. Clin Anat 28(1):27–36
Skaper S, Facci L, Giusti P (2014) Neuroinflammation, Microglia and Mast Cells in the Pathophysiology of Neurocognitive Disorders: A Review. CNS & neurological disorders drug targets
Zhang B, Gensel J (2014) Is neuroinflammation in the injured spinal cord different than in the brain? Examining intrinsic differences between the brain and spinal cord. Exp Neurol 258:112–120
Allison D, Ditor D (2014) Immune dysfunction and chronic inflammation following spinal cord injury. Spinal cord
Sroga JM, Jones T, Kigerl KA, McGaughy VM, Popovich PG (2003) Rats and mice exhibit distinct inflammatory reactions after spinal cord injury. J Comp Neurol 462(2):223–240
Habib P, Beyer C (2015) Regulation of brain microglia by female gonadal steroids. J Steroid Biochem Mol Biol 146:3–14
Orihuela R, McPherson CA, Harry GJ (2015) Microglial M1/M2 polarization and metabolic states. British journal of pharmacology
Rice T, Larsen J, Rivest S, Yong VW (2007) Characterization of the early neuroinflammation after spinal cord injury in mice. J Neuropathol Exp Neurol 66(3):184–195
Pineau I, Sun L, Bastien D, Lacroix S (2010) Astrocytes initiate inflammation in the injured mouse spinal cord by promoting the entry of neutrophils and inflammatory monocytes in an IL-1 receptor/MyD88-dependent fashion. Brain Behav Immun 24(4):540–553
Taoka Y, Okajima K, Uchiba M, Murakami K, Kushimoto S, Johno M, Naruo M, Okabe H et al (1997) Role of neutrophils in spinal cord injury in the rat. Neuroscience 79(4):1177–1182
Gaudet AD, Popovich PG (2014) Extracellular matrix regulation of inflammation in the healthy and injured spinal cord. Exp Neurol 258:24–34
Heiman A, Pallottie A, Heary RF, Elkabes S (2014) Toll-like receptors in central nervous system injury and disease: a focus on the spinal cord. Brain Behav Immun 42:232–245
Walsh JG, Muruve DA, Power C (2014) Inflammasomes in the CNS. Nat Rev Neurosci 15(2):84–97
Kigerl KA, de Rivero Vaccari JP, Dietrich WD, Popovich PG, Keane RW (2014) Pattern recognition receptors and central nervous system repair. Exp Neurol 258:5–16
Hanamsagar R, Hanke ML, Kielian T (2012) Toll-like receptor (TLR) and inflammasome actions in the central nervous system. Trends Immunol 33(7):333–342
Kigerl KA, Popovich PG (2009) Toll-like receptors in spinal cord injury. In: Toll-like Receptors: Roles in Infection and Neuropathology. Springer, pp 121–136
Impellizzeri D, Mazzon E, Paterniti I, Esposito E, Cuzzocrea S (2012) Effect of fasudil, a selective inhibitor of Rho kinase activity, in the secondary injury associated with the experimental model of spinal cord trauma. J Pharmacol Exp Ther 343(1):21–33
Lu M, Grove EA, Miller RJ (2002) Abnormal development of the hippocampal dentate gyrus in mice lacking the CXCR4 chemokine receptor. Proc Natl Acad Sci 99(10):7090–7095
Li M, Ransohoff RM (2008) Multiple roles of chemokine CXCL12 in the central nervous system: a migration from immunology to neurobiology. Prog Neurobiol 84(2):116–131
Ambrosini E, Remoli ME, Giacomini E, Rosicarelli B, Serafini B, Lande R, Aloisi F, Coccia EM (2005) Astrocytes produce dendritic cell-attracting chemokines in vitro and in multiple sclerosis lesions. J Neuropathol Exp Neurol 64(8):706–715
Karin N (2010) The multiple faces of CXCL12 (SDF-1α) in the regulation of immunity during health and disease. J Leukoc Biol 88(3):463–473
Engelhardt B, Ransohoff RM (2012) Capture, crawl, cross: the T cell code to breach the blood–brain barriers. Trends Immunol 33(12):579–589
Momcilović M, Mostarica-Stojković M, Miljković D (2012) CXCL12 in control of neuroinflammation. Immunol Res 52(1–2):53–63
Colamussi ML, Secchiero P, Gonelli A, Marchisio M, Zauli G, Capitani S (2001) Stromal derived factor-1α (SDF-1α) induces CD4+ T cell apoptosis via the functional up-regulation of the Fas (CD95)/Fas ligand (CD95L) pathway. J Leukoc Biol 69(2):263–270
Meiron M, Zohar Y, Anunu R, Wildbaum G, Karin N (2008) CXCL12 (SDF-1α) suppresses ongoing experimental autoimmune encephalomyelitis by selecting antigen-specific regulatory T cells. J Exp Med 205(11):2643–2655
Patel JR, McCandless EE, Dorsey D, Klein RS (2010) CXCR4 promotes differentiation of oligodendrocyte progenitors and remyelination. Proc Natl Acad Sci 107(24):11062–11067
Acknowledgments
This work was supported by an internal START grant from the Medical Clinic (UKA) of the RWTH Aachen University (A. Zendedel).
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Statement
All animals used in this study were housed under specific pathogen-free conditions and with access to standard rodent diet and tap water ad libitum. All experiments were conducted in conformance with the NIH Guidelines for the Care and Use of Laboratory Animals.
Author information
Authors and Affiliations
Corresponding author
Additional information
Adib Zendedel and Sonja Johann contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zendedel, A., Johann, S., Mehrabi, S. et al. Activation and Regulation of NLRP3 Inflammasome by Intrathecal Application of SDF-1a in a Spinal Cord Injury Model. Mol Neurobiol 53, 3063–3075 (2016). https://doi.org/10.1007/s12035-015-9203-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9203-5