Abstract
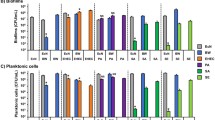
In this study, we examined the biofilm forming ability, the mRNA expression of curli genes and the morphologies of curli fimbriae and biofilms in clinical isolates of Enterobacter cloacae. The csgBA operon was found in 11 (78.6%) of the 14 isolates. The ability of E. cloacae isolates to form biofilms was significantly correlated with the mRNA expression level of the csgA and csgD genes. The curli protein fimbriae appeared as tangled fibers and the curli-proficient strain formed mature biofilms. Our data suggest that the expression of the curli fimbriae play an important role in biofilm formation in E. cloacae.
Similar content being viewed by others
References
Austin, J.W., Sanders, G., Kay, W., and Collinson, S. 1998. Thin aggregative fimbriae enhance Salmonella enteritidis biofilm formation. FEMS Microbiol. Lett. 162, 295–301.
Ben Nasr, A., Olsen, A., Sjobring, U., Muller-Esterl, W., and Bjorck, L. 1996. Assembly of human contact phase proteins and release of bradykinin at the surface of curli-expressing Escherichia coli. Mol. Microbiol. 20, 927–935.
Brombacher, E., Baratto, A., Dorel, C., and Landini, P. 2006. Gene expression regulation by the curli activator CsgD protein: Modulation of cellulose biosynthesis and control of negative determinants for microbial adhesion. J. Bact. 188, 2027–2037.
Choi, Y.H., Lee, H.W., Kim, S.M., Lee, J.C., Lee, Y.C., Seol, S.Y., Cho, D.T., and Kim, J. 2011. Biofilm forming ability and production of curli and cellulose in clinical isolates of Enterobacteriaceae. Kor. J. Microbiol. 47, 335–341.
Costerton, J.W., Stewart, P.S., and Greenberg, E.P. 1999. Bacterial biofilms: a common cause of persistent infections. Science 284, 1318–1322.
Crump, J.A. and Collignon, P.J. 2000. Intravascular catheter-associated infections. Eur. J. Clin. Microbiol. Infect. Dis. 19, 1–8.
Donlan, R.M. and Costerton, J.W. 2002. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin. Microbiol Rev. 15, 167–193.
Fernandez-Baca, V., Ballesteros, F., Hervas, J.A., Villalon, P., Dominquez, M.A., Benedi, V.J., and Alberti, S. 2001. Molecular epidemiological typing of Enterobacter cloacae isolates from a neonatal intensive care unit: 3-year prospective study. J. Hosp. Infect. 49, 173–182.
Hammar, M., Arnqvist, A., Bian, Z., Olsen, A., and Normark, S. 1995. Expression of two csg operons is required for production of fibronectin- and Congo red-binding curli polymers in Escherichia coli K-12. Mol. Microbiol. 18, 661–670.
Hammar, M., Bian, Z., and Normark, S. 1996. Nucleator-dependent intercellular assembly of adhesive curli organelles in Escherichia coli. Proc. Natl. Acad. Sci. USA 93, 6562–6566.
Harbarth, S., Sudre, P., Dharan, S., Cadenas, M., and Pittet, D. 1999. Outbreak of Enterobacter cloacae related to understaffing, overcrowding, and poor hygiene practices. Infect. Control Hosp. Epidemiol. 20, 598–603.
Heilmann, C., Hussain, M., Peters, G., and Gotz, F. 1997. Evidence for autolysin-mediated primary attachment of Staphylococcus epidermidis to a polystyrene surface. Mol. Microbiol. 29, 1013–1024.
Jonas, K., Tomenius, H., Kader, A., Normark, S., Romling, U., Belova, L.M., and Melefors, O. 2007. Roles of curli, cellulose and BapA in Salmonella biofilm morphology studied by atomic force microscopy. BMC Microbiol. 7, 70.
Kikuchi, T., Mizunoe, Y., Takade, A., Naito, S., and Yoshida, S. 2005. Curli fibers are required for development of biofilm architecture in Escherichia coli K-12 and enhance bacterial adherence to human uroepithelial cells. Mol. Immun. 49, 875–884.
Loferer, H., Hammar, M., and Normark, S. 1997. Availability of the fibre subunit CsgA and the nucleator protein CsgB during assembly of fibronectin-binding curli is limited by the intracellular concentration of the novel lipoprotein CsgG. Mol. Microbiol. 26, 11–23.
Martinez, A., Torello, S., and Kolter, R. 1999. Sliding motility in mycobacteria. J. Bacteriol. 181, 7331–7338.
Olsen, A., Arnqvist, A., Hammar, M., and Normark, S. 1993. Environmental regulation of curli production in Escherichia coli. Infect. Agents Dis. 2, 272–274.
Raad, I. 1998. Intravascular-catheter-related infections. Lancet 351, 893–898.
Romling, U., Bian, Z., Hammar, M., Sierralta, W., and Normark, S. 1998. Curli fibers are highly conserved between Salmonella typhimurium and Escherichia coli with respect to operon structure and regulation. J. Bacteriol. 180, 722–731.
Sjobring, U., Pohl, G., and Olsen, A. 1994. Plasminogen, absorbed by Escherichia coli expressing curli or by Salmonella enteritidis expressing thin aggregative fimbriae, can be activated by simultaneously captured tissue-type plasminogen activator (t-PA). Mol. Microbiol. 14, 443–452.
Vidal, O., Longin, R., Prigent-Combaret, C., Dorel, C., Hooreman, M., and Lejeune, P. 1998. Isolation of an Escherichia coli K-12 mutant strain able to form biofilms on inert surfaces: involvement of a new ompR allele that increases curli expression. J. Bacteriol. 180, 2442–2449.
Vinh, D.C. and Embil, J.M. 2005. Device-related infections: a review. J. Long Term Eff. Med. Implants 15, 467–488.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, SM., Lee, HW., Choi, YW. et al. Involvement of curli fimbriae in the biofilm formation of Enterobacter cloacae . J Microbiol. 50, 175–178 (2012). https://doi.org/10.1007/s12275-012-2044-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2044-2