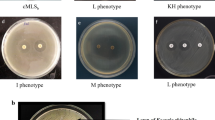
Abstract
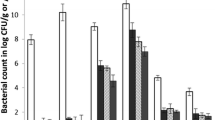
Inappropriate use of aminoglycosides in animal husbandry has led to the selection and emergence of high-level aminoglycoside resistance (HLAR) in lactic acid bacteria (LAB). The objective of this study was to assess the presence of aminoglycoside resistant LAB in farm animals and meat products. Gentamicin resistant LAB (n = 138) were selectively isolated from 50 different meat and farm animal sources. These native isolates of LAB were subsequently characterized for their minimum inhibitory concentration to seven different aminoglycoside antibiotics. HLAR to gentamicin, kanamycin and streptomycin was found to be 38 %, 45 % and 15 %, respectively. Selected cultures of LAB were identified by random amplified polymorphic DNA (RAPD)-PCR and 16S rDNA gene sequencing. Subsequent detection for the presence of nine aminoglycoside modifying genes [aac(6′)Ie-aph(2″)Ia, aph(3′)IIIa, aad6, ant(6)Ia, ant(9)Ia, ant(9)Ib, aph(2″)Ib, aph(2″)Ic and aph(2″)Id] was carried out by PCR. The Enterococcus spp. (n = 64) and Lactobacillus plantarum (n = 6) isolated from farm animals and chicken sausages, respectively, were positive for the bifunctional gene, aac(6′)Ie-aph(2″)Ia in PCR. In addition, Enterococcus sp. (n = 17), Lactobacillus plantarum (n = 3), and Pediococcus lolii (n = 2) possessed the kanamycin resistance gene, aph(3′)IIIa. Other LAB viz. Enterococcus faecalis (n = 2), E. faecium (n = 2) and L. plantarum (n = 1) harbored the streptomycin resistance gene, aad6. The integrase (int) gene, characteristic to Tn 916-Tn 1545 was detected in Enterococcus faecalis CS11+ and Enterococcus cecorum I40a suggesting its involvement in antibiotic resistant gene transfer. Besides, strains of L. plantarum, a species used as probiotic, isolated in this study showed the occurrence of aph(3′)IIIa as well as aac (6′)Ie-aph(2″)Ia genes that could be of concern in human health. The findings of the study highlight the spread and emergence of multi-resistance genes for aminoglycoside antibiotics among beneficial LAB.

Similar content being viewed by others
References
Altschul SF, Maddan TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSIBLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bismuth R, Courvalin P (2010) Aminoglycosides and Gram-positive bacteria: antibiogram, 3rd edn. ESKA, Portland
Boehr DD, Daigle DM, Wright GD (2004) Domain–domain interactions in the aminoglycoside antibiotic resistance enzyme AAC (6′)–APH (2″). Biochemistry 43:9846–9855
Byrne ME, Rouch DA, Skurray RA (1989) Nucleotide sequence analysis of IS256 from the Staphylococcus aureus gentamicin-tobramycin-kanamycin-resistance transposon Tn4001. Gene 81:361–367
Chen YG, Qu TT, Yu YS, Zhou JY, Li LJ (2006) Insertion sequence ISEcp1- like element connected with a novel aph(2″) allele [aph(2″)-Ie] conferring high level gentamicin resistance and a novel streptomycin adenylyltransferase gene in Enterococcus. J Med Microbiol 55:1521–1525
Chow JW, Zervos MJ, Lerner SA, Thal LA, Donabedian SM, Jaworski DD, Tsai S, Shaw KJ, Clewell DB (1997) A novel gentamicin resistance gene in Enterococcus. Antimicrob Agents Chemother 41:511–514
CLSI (2012) Performance standards for antimicrobial susceptibility testing; twenty second informational supplement. In: CLSI document M100-S22. Clinical and Laboratory Standards Institute, Wayne
Culebras E, Martinez JL (1999) Aminoglycoside resistance mediated by the bifunctional enzyme 6′ N aminoglycoside acetyltransferase - 2″-O-aminoglycoside phosphotransferase. Front Biosci 4:1–8
Del Campo R, Tenorio C, Rubio C, Castillo J, Torres C, Gomez-Lus R (2000) Aminoglycoside-modifying enzymes in high level streptomycin and gentamicin resistant Enterococcus spp. in Spain. Int J Antimicrob Agents 15:221–226
Del Campo R, Galán JC, Tenorio C, Ruiz-Garbajosa P, Zarazaga M, Torres C, Baquero F (2005) New aac(6')-I genes in Enterococcus hirae and Enterococcus durans: effect on {beta}-lactam/aminoglycoside synergy. J Antimicrob Chemother 55(6):1053–1055
Devirgiliis C, Barile S, Perozzi G (2011) Antibiotic resistance determinants in the interplay between food and gut microbiota. Genes Nutr 6:275–284
Doherty N, Trzcinski K, Pickerill P, Zawadzki P, Dowson CG (2000) Genetic diversity of the tet(M) gene in tetracycline resistant clonal lineages of Streptococcus pneumoniae. Antimicrob Agents Chemother 44:2979–2984
Donabedian SM, Thal LA, Hershberger E, Perri MB, Chow JW, Bartlett P, Jones R, Joyce K, Rositer S, Gay K, Johnson J, Mackinson C, Debess E, Madden J, Angulo F, Zervos MJ (2003) Molecular characterization of gentamicin-resistant enterococci in the United States: evidence of spread from animals to humans through food. J Clin Microbiol 41:1109–1113
EFSA (2008) Update of the criteria used in the assessment of bacterial resistance to antibiotics of human or veterinary importance. EFSA J 732:1–15
EFSA (2012) Guidance on the assessment of bacterial susceptibility to antimicrobials of human and veterinary importance. EFSA J 10:1–10
Eliopoulos GM, Wennersten C, Zighelboim-Daum S, Reiszner E, Goldmann D, Moellering RC Jr (1988) High level resistance to gentamicin in clinical isolates of Enterococcus faecium. Antimicrob Agents Chemother 32:1528–1532
Ferretti JJ, Gilmore KS, Courvalin P (1986) Nucleotide sequence analysis of the gene specifying the bifunctional 6′- aminoglycoside acetyltransferase 2″ aminoglycoside phosphtransferase enzyme in Streptococcus faecalis and identification and cloning of gene regions specifying the two activities. J Bacteriol 167:631–638
Guardabassi L, Schwarz S, Lloyd DH (2004) Pet animals as reservoirs of antimicrobial-resistant bacteria. J Antimicrob Chemother 54:321–332
Guardabassi L, Jensen LB, Kruse H (2008) Guide to antimicrobial use in animals. Blackwell, UK
Hammerum AM, Lester CH, Heuer OE (2010) Antimicrobial-resistant enterococci in animals and meat: a human health hazard? Foodborne Pathog Dis 7:1137–1146
Hodel-Christian SL, Murray BE (1991) Characterization of the gentamicin resistance transposon Tn 5281 from Enterococcus faecalis and comparison to staphylococcal transposons Tn 4001 and Tn 4031. Antimicrob Agents Chemother 35:1147–1152
Holt JG, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Bergey’s Manual of Determinative Bacteriology, 9th edn. Williams & Wilkins, Baltimore
Hseueh PR, Teng LJ, Chen YC, Yang PC, Ho SW, Luh KT (2000) Recurrent bacteremic peritonitis caused by Enterococcus cecorum in a patient with liver cirrhosis. J Clin Microbiol 38:2450–2452
Jackson CR, Fedorka-Cray PJ, Barrett JB, Ladely SR (2004) Genetic relatedness of high level aminoglycoside resistant enterococci isolated from poultry carcasses. Avian Dis 48:100–107
Jackson CR, Fedorka-Cray PJ, Davies JA, Barrett JB, Frye JG (2009) Prevalence, species distribution and antimicrobial resistance of enterococci isolated from dogs and cats in the United States. J Appl Microbiol 107:1269–1278
Jackson CR, Lombard JE, Dargatz DA, Fedorka-Cray PJ (2010) Prevalence, species distribution and antimicrobial resistance of enterococci isolated from US dairy cattle. Lett Appl Microbiol 52:41–48
Jamshidi S, Jamshidi S (2011) NTSYSpc 2.02e implementation in molecular biodata analysis (clustering, screening, and individual selection). 2011 International Conference on Environmental and Computer Science. IPCBEE, vol.19. IACSIT, Singapore, pp 165–169
Kao SJ, You IL, Clewell DB, Donabedian SM, Zervos MJ, Petrin J, Shaw KJ, Chow JW (2000) Detection of the high-level aminoglycoside resistance gene aph(2″)-Ib in Enterococcus faecium. Antimicrob Agents Chemother 44:2876–2879
Kimprasit T, Sukontasing S, Amavisit P (2013) In vitro selection of potential lactic acid bacteria isolated from ducks and geese in Thailand. Kasetsart J (Nat Sci) 47:261–270
Klibi N, Aouini R, Borgo F, Said LB, Ferrario C, Dziri R, Boudabous A, Torres C, Slama KB (2014) Antibiotic resistance and virulence of faecal enterococci isolated from food-producing animals in Tunisia. Ann Microbiol. doi:10.1007/s13213-014-0908-x
Kuhn I, Iverson A, Burman LG, Ollsson-Liljequist B, Franklin A, Finn M, Aarestrup F, Seyfarth AM, Blanch AR, Vilanova X, Taylor H, Caplin J, Moreno MA, Dominguez L, Herrero IA, Möllby R (2003) Comparison of enterococcal populations in animals, humans, and the environment—a European study. Int J Food Microbiol 88:133–145
Kuhn I, Iversen A, Finn M, Greko C, Burman LG, Blanch AR, Vilanova X, Manero A, Taylor H, Caplin J, Dominguez L, Herrero IA, Moreno MA, Mollby R (2005) Occurrence and relatedness of vancomycin-resistant enterococci in animals, humans, and the environment in different European regions. Appl Environ Microbiol 71:5383–5390
Lopes MFS, Ribeiro T, Martins MP, Tenreiro R, Crespo MTB (2003) Gentamicin resistance in dairy and clinical enterococcal isolates and in reference strains. J Antimicrob Chemother 52:214–219
Mathur S, Singh R (2005) Antibiotic resistance in food lactic acid bacteria—a review. Int J Food Microbiol 105:281–295
Mora D, Fortina MG, Parini C, Daffonchio D, Manachini PL (2000) Genomic subpopulations within the species Pediococcus acidilactici detected by multilocus typing analysis: relationships between pediocin AcH/PA-1 producing and non-producing strains. Microbiology 46:2027–2038
Murray BE (1990) The life and times of the Enterococcus. Clin Microbiol Rev 3:46–65
Nielsen KM, Bohn T, Townsend JP (2014) Detecting rare gene transfer events in bacterial populations. Front Microbiol 4:1–12
Ouoba LII, Lei V, Jensen LB (2008) Resistance of potential probiotic lactic acid bacteria and bifidobacteria of African and European origin to antimicrobials: determination and transferability of the resistance genes to other bacteria. Int J Food Microbiol 121:217–224
Poeta P, Costa D, Saenz Y, Klibi N, Ruiz-Larrea F, Rodrigues J, Torres C (2005) Characterization of antibiotic resistance genes and virulence factors in faecal enterococci of wild animals in Portugal. J Vet Med B Infect Dis Vet Public Health 52:396–402
Popova M, Molimard P, Courau S, Crociani J, Dufour C, Le Vacon F, Carton T (2012) Beneficial effects of probiotics in upper respiratory tract infections and their mechanical actions to antagonize pathogens. J Appl Microbiol 113:1305–1318
Ramirez MS, Tolmasky ME (2010) Aminoglycoside modifying enzymes. Drug Resist Updat 13:151–171
Ramos S, Igrejas G, Capelo-Martinez JL, Poeta P (2012) Antibiotic resistance and mechanisms implicated in fecal enterococci recovered from pigs, cattle and sheep in a Portuguese slaughterhouse. Ann Microbiol 62:1485–1494
Schillinger U, Yousif NMK, Sesar L, Franz CMAP (2003) Use of group specific and RAPD PCR analyses for rapid differentiation of Lactobacillus strains from probiotic yoghurts. Curr Microbiol 47:453–456
Shao Y, Zhang W, Guo H, Pan L, Zhang H, Sun T (2015) Comparitive studies on antibiotic resistance in Lactobacillus casei and Lactobacillus plantarum. Food Control 50:250–258
Shwarz S, Kehrenberg C, Walsh TR (2000) Use of antimicrobial agents in veterinary medicine and food animal production. Int J Antimicrob Agents 17:431–437
Silva N, Igrejas G, Gonçalves A, Poeta P (2012) Commensal gut bacteria: distribution of Enterococcus species and prevalence of E. coli phylogenetic groups in animals and humans in Portugal. Ann Microbiol 62:449–459
Simjee S, Gill MJ (1997) Gene transfer, gentamicin resistance and enterococci. J Hosp Infect 36:249–259
Stalker MJ, Brash ML, Weisz A, Ouckama RM, Slavic D (2010) Arthritis and osteomyelitis associated with Enterococcus cecorum infection in broiler and broiler breeder chickens in Ontario, Canada. J Vet Diagn Investig 22:643–645
Tenorio C, Zarazaga M, Martinez C, Torres C (2001) Bifunctional 6′-N-aminoglycoside acetyltransferase-2″-O-aminoglycoside phosphotransferase in Lactobacillus and Pedicoccus isolates of animal origin. J Clin Microbiol 39:824–825
The United States Pharmacopeial Convention, Inc (2008) The United States pharmacopeia 31/the national formulary 26, Supp. 1, 8-1-08, online. United State Pharmacopeial Convention, Rockville
Thumu SCR, Halami PM (2012) Presence of erythromycin and tetracycline resistance genes in lactic acid bacteria from fermented foods of Indian origin. Antonie Van Leeuwenhoek 102:541–551
Thumu SCR, Halami PM (2013) Phenotypic expression, molecular characterization and transferability of erythromycin resistance genes in Enterococcus spp. isolated from naturally fermented food. J Appl Microbiol 116:689–699
Tsai SF, Zervos MJ, Clewell DB, Donabedian SM, Sahm DF, Chow JW (1998) A new high-level gentamicin resistance gene, aph(2″)-Id, in Enterococcus spp. Antimicrob Agents Chemother 42:1229–1232
Yates CM, Pearce MC, Woolhouse ME, Amyes SG (2004) High frequency transfer and horizontal spread of apramycin resistance in calf faecal E. coli. J Antimicrob Chemother 54:534–537
Acknowledgments
The authors would like to thank Prof. Ram Rajasekharan, Director, CSIR-CFTRI, Mysore, for use of facilities. J.G. acknowledges the UGC for the grant of MAN fellowship. We also thank Indian Council of Medical Research, New Delhi for financial assistance under the scheme of antimicrobial drug resistance. The authors wish to sincerely acknowledge Dr M.C. Varadaraj for critical comments on the manuscript.
Conflict of interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jaimee, G., Halami, P.M. High level aminoglycoside resistance in Enterococcus, Pediococcus and Lactobacillus species from farm animals and commercial meat products. Ann Microbiol 66, 101–110 (2016). https://doi.org/10.1007/s13213-015-1086-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-015-1086-1