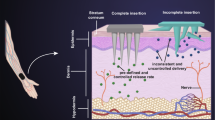
Abstract
Although vitamin D3 (VD3), which is the main form of vitamin D, can be produced in human skin under the sunlight, vitamin D deficiency emerged as a major public health problem worldwide. Mainly, oral supplements or vitamin D-fortified foods are distributed to help supplementation of vitamin D. However, those oral methods are limitedly supplied in the Middle East countries, and oral absorption has low efficiency due to many barriers and various changes of conditions along the route. Then, it is recommended to take them every day in order to maintain the adequate serum level of vitamin D. Alternatively, transdermal delivery system could provide a convenient way to get sustained supplement of vitamin D by its advantages like avoiding first-pass effect of the liver and providing release for long periods of time. In this study, we introduced transdermal delivery system for sustained vitamin D release using coating microneedles that easily pierce the skin layer with enough mechanical strength and allow the localization of drugs within the dermal region. According to the experimental results, poly (lactic-co-glycolic acid) (PLGA) successfully encapsulated VD3 as a nanoparticle form with appropriate properties for transdermal delivery such as size distribution, skin compatibility, and effective release of encapsulated compound. Finally, PVD3 layers coated on solid microneedles were completely dissolved into intradermal region in porcine skin model and revealed better performance for VD3 release into plasma compared to ointment base transdermal method.









Similar content being viewed by others
References
Nykjaer A, Dragun D, Walther D, Vorum H, Jacobsen C, Herz J, et al. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell. 1999;96(4):507–15. https://doi.org/10.1016/S0092-8674(00)80655-8.
Adams JS, Hewison M. Update in vitamin D. J Clin Endocrinol Metab. 2010;95(2):471–8. https://doi.org/10.1210/jc.2009-1773.
Lehmann B, Meurer M. Vitamin D metabolism. Dermatol Ther. 2010;23(1):2–12. https://doi.org/10.1111/j.1529-8019.2009.01286.x.
Luo Y, Teng Z, Wang Q. Development of zein nanoparticles coated with carboxymethyl chitosan for encapsulation and controlled release of vitamin D3. J Agric Food Chem. 2012;60(3):836–43. https://doi.org/10.1021/jf204194z.
Al Anouti F, Thomas J, Abdel-Wareth L, Rajah J, Grant WB, Haq A. Vitamin D deficiency and sun avoidance among university students at Abu Dhabi, United Arab Emirates. Dermato-endocrinology. 2011;3(4):235–9. https://doi.org/10.4161/derm.3.4.16881.
Heaney RP. Vitamin D in health and disease. Clin J Am Soc Nephrol. 2008;3(5):1535–41. https://doi.org/10.2215/CJN.01160308.
Alsaqr A, Rasoully M, Musteata FM. Investigating transdermal delivery of vitamin D3. AAPS PharmSciTech. 2015;16(4):963–72. https://doi.org/10.1208/s12249-015-0291-3.
N’Da DD. Prodrug strategies for enhancing the percutaneous absorption of drugs. Molecules. 2014;19(12):20780–807. https://doi.org/10.3390/molecules191220780.
Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol. 2008;26(11):1261–8. https://doi.org/10.1038/nbt.1504.
Haham M, Ish-Shalom S, Nodelman M, Duek I, Segal E, Kustanovich M, et al. Stability and bioavailability of vitamin D nanoencapsulated in casein micelles. Food Funct. 2012;3(7):737–44. https://doi.org/10.1039/c2fo10249h.
Abbasi A, Emam-Djomeh Z, Mousavi MA, Davoodi D. Stability of vitamin D(3) encapsulated in nanoparticles of whey protein isolate. Food Chem. 2014;143:379–83. https://doi.org/10.1016/j.foodchem.2013.08.018.
Manosroi A, Chankhampan C, Manosroi W, Manosroi J. Transdermal absorption enhancement of papain loaded in elastic niosomes incorporated in gel for scar treatment. Eur J Pharm Sci. 2013;48(3):474–83. https://doi.org/10.1016/j.ejps.2012.12.010.
Baek SH, Shin JH, Kim YC. Drug-coated microneedles for rapid and painless local anesthesia. Biomed Microdevices. 2017;19(1):2. https://doi.org/10.1007/s10544-016-0144-1.
Donnelly RF, Morrow DI, Fay F, Scott CJ, Abdelghany S, Singh RR, et al. Microneedle-mediated intradermal nanoparticle delivery: potential for enhanced local administration of hydrophobic pre-formed photosensitisers. Photodiagn Photodyn Ther. 2010;7(4):222–31. https://doi.org/10.1016/j.pdpdt.2010.09.001.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64(14):1547–68. https://doi.org/10.1016/j.addr.2012.04.005.
Prausnitz MR. Microneedles for transdermal drug delivery. Adv Drug Deliv Rev. 2004;56(5):581–7. https://doi.org/10.1016/j.addr.2003.10.023.
Lee SG, Jeong JH, Lee KM, Jeong KH, Yang H, Kim M, et al. Nanostructured lipid carrier-loaded hyaluronic acid microneedles for controlled dermal delivery of a lipophilic molecule. Int J Nanomedicine. 2014;9:289–99. https://doi.org/10.2147/IJN.S54529.
Ramadan E, Borg T, Abdelghani G, Saleh NM. Transdermal microneedle-mediated delivery of polymeric lamivudine-loaded nanoparticles. J Pharm Technol Drug Res. 2016;5(1):1. https://doi.org/10.7243/2050-120X-5-1.
Rao JP, Geckeler KE. Polymer nanoparticles: preparation techniques and size-control parameters. Prog Polym Sci. 2011;36(7):887–913. https://doi.org/10.1016/j.progpolymsci.2011.01.001.
Panyam J, Dali MM, Sahoo SK, Ma W, Chakravarthi SS, Amidon GL, et al. Polymer degradation and in vitro release of a model protein from poly(D,L-lactide-co-glycolide) nano- and microparticles. J Control Release. 2003;92(1–2):173–87. https://doi.org/10.1016/S0168-3659(03)00328-6.
Kim SJ, Shin JH, Noh JY, Song CS, Kim YC. Development of the novel coating formulations for skin vaccination using stainless steel microneedle. Drug Deliv Transl Res. 2016;6(5):486–97. https://doi.org/10.1007/s13346-016-0321-z.
Kim YC, Quan FS, Compans RW, Kang SM, Prausnitz MR. Formulation and coating of microneedles with inactivated influenza virus to improve vaccine stability and immunogenicity. J Control Release. 2010;142(2):187–95. https://doi.org/10.1016/j.jconrel.2009.10.013.
Gill HS, Prausnitz MR. Coated microneedles for transdermal delivery. J Control Release. 2007;117(2):227–37. https://doi.org/10.1016/j.jconrel.2006.10.017.
Seok H, Noh JY, Lee DY, Kim SJ, Song CS, Kim YC. Effective humoral immune response from a H1N1 DNA vaccine delivered to the skin by microneedles coated with PLGA-based cationic nanoparticles. J Control Release. 2017; https://doi.org/10.1016/j.jconrel.2017.04.027.
Caldorera-Moore M, Guimard N, Shi L, Roy K. Designer nanoparticles: incorporating size, shape and triggered release into nanoscale drug carriers. Expert Opin Drug Deliv. 2010;7(4):479–95. https://doi.org/10.1517/17425240903579971.
Cegnar M, Kos J, Kristl J. Cystatin incorporated in poly(lactide-co-glycolide) nanoparticles: development and fundamental studies on preservation of its activity. Eur J Pharm Sci. 2004;22(5):357–64. https://doi.org/10.1016/j.ejps.2004.04.003.
Coulman SA, Anstey A, Gateley C, Morrissey A, McLoughlin P, Allender C, et al. Microneedle mediated delivery of nanoparticles into human skin. Int J Pharm. 2009;366(1–2):190–200. https://doi.org/10.1016/j.ijpharm.2008.08.040.
Desai ND, Singh PP, Amin PD, Jain SP. Stability-indicating LC method for assay of cholecalciferol. Chromatographia. 2009;69(3–4):385–8. https://doi.org/10.1365/s10337-008-0914-x.
Budhian A, Siegel SJ, Winey KI. Production of haloperidol-loaded PLGA nanoparticles for extended controlled drug release of haloperidol. J Microencapsul. 2005;22(7):773–85. https://doi.org/10.1080/02652040500273753.
Yang YY, Chia HH, Chung TS. Effect of preparation temperature on the characteristics and release profiles of PLGA microspheres containing protein fabricated by double-emulsion solvent extraction/evaporation method. J Control Release. 2000;69(1):81–96. https://doi.org/10.1016/S0168-3659(00)00291-1.
Makadia HK, Siegel SJ. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers. 2011;3(3):1377–97. https://doi.org/10.3390/polym3031377.
Hossein-nezhad A, Holick MF, editors. Vitamin D for health: a global perspective. Mayo Clinic Proceedings. Elsevier; 2013.
Li X, Kong X, Zhang Z, Nan K, Li L, Wang X, et al. Cytotoxicity and biocompatibility evaluation of N,O-carboxymethyl chitosan/oxidized alginate hydrogel for drug delivery application. Int J Biol Macromol. 2012;50(5):1299–305. https://doi.org/10.1016/j.ijbiomac.2012.03.008.
Archana D, Singh BK, Dutta J, Dutta P. In vivo evaluation of chitosan–PVP–titanium dioxide nanocomposite as wound dressing material. Carbohydr Polym. 2013;95(1):530–9. https://doi.org/10.1016/j.carbpol.2013.03.034.
Kim YC, Song JM, Lipatov AS, Choi SO, Lee JW, Donis RO, et al. Increased immunogenicity of avian influenza DNA vaccine delivered to the skin using a microneedle patch. Eur J Pharm Biopharm. 2012;81(2):239–47. https://doi.org/10.1016/j.ejpb.2012.03.010.
Ammar H, Ghorab M, El-Nahhas S, Kamel R. Evaluation of chemical penetration enhancers for transdermal delivery of aspirin. Asian J Pharm Sci. 2007;2:96–105.
Godin B, Touitou E. Transdermal skin delivery: predictions for humans from in vivo, ex vivo and animal models. Adv Drug Deliv Rev. 2007;59(11):1152–61. https://doi.org/10.1016/j.addr.2007.07.004.
Acknowledgements
This work was supported by Matching Fund Project of KAIST-Kustar Institute (Project No. N11160038) and by the Ministry of Science and ICT (Project No. NRF-2014M3A9E4064580).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kim, HG., Gater, D.L. & Kim, YC. Development of transdermal vitamin D3 (VD3) delivery system using combinations of PLGA nanoparticles and microneedles. Drug Deliv. and Transl. Res. 8, 281–290 (2018). https://doi.org/10.1007/s13346-017-0460-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-017-0460-x