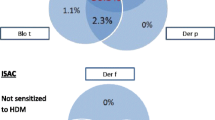
Abstract
Background
ImmunoCAP ISAC 112, is a commercially available molecular allergy IgE multiplex test. Data on the comparison of this rather novel test with extract-based as well as molecular ImmunoCAP singleplex IgE tests is missing.
Objective
To perform a comparison between the ISAC multiplex IgE assay and the ImmunoCAP singleplex test results.
Methods
Serum samples of 101 adults with grass pollen allergy were analysed for sIgE to 112 allergenic molecules represented on the ISAC test as well as to common atopy-related extract-based allergy tests with the ImmunoCAP System (house dust mite [d1], cat [e1], dog [e5], cow’s milk [f2], hen’s egg [f1], hazelnut [f17], celery [f85], Alternaria alternate [m6], as well as pollen from birch [t3], hazel [t4], mugwort [w6], and ragweed [w1]). Subsequently statistical analysis was performed with the Spearman rank correlation test and the Clopper-Pearson method in order to compare the ISAC multiplex results with the sIgE singleplex results.
Results
The positive percent agreements (PPA) and negative percent agreement (NPA) of corresponding allergens between the ISAC sIgE test and the extract-based singleplex ImmunoCAP results at cutoff 0.1 kUA/l varied between 60–100 % for PPA and 78–97 % for NPA.
Conclusion
When taking into account corresponding allergens molecular testing with the ISAC multiplex test correlates well with ImmunoCAP singleplex results.

Similar content being viewed by others
Abbreviations
- CCD:
-
Cross-reactive carbohydrate determinant
- CI:
-
Confidence interval
- CV:
-
Coefficient of variation
- IgE:
-
Immunglobulin E
- ISAC:
-
Immuno solid-phase allergen chip
- ISU:
-
ISAC standardized unit
- ISU-E:
-
ImmunoCAP specific IgE
- kUA:
-
Kilounits (corresponding allergens)
- LoD:
-
Limit of detection
- MA:
-
Molecular allergy diagnostics
- NPA:
-
Negative percent agreement
- PPA:
-
Positive percent agreement
- SD:
-
Standard deviation
- sIgE:
-
Specific Immunglobulin E
- SIT:
-
Specific Immunotherapy
References
Canonica GW, Ansotegui IJ, Pawankar R, Schmid-Grendelmeier P, van Hage M, Baena-Cagnani CE, Melioli G, Nunes C, Passalacqua G, Rosenwasser L, Sampson H, Sastre J, Bousquet J, Zuberbier T. A WAO - ARIA - GA(2)LEN consensus document on molecular-based allergy diagnostics. World Allergy Organ J 2013;6:17
Szecsi PB, Stender S. Comparison of immunoglobulin E measurements on IMMULITE and ImmunoCAP in samples consisting of allergen-specific mouse-human chimeric monoclonal antibodies towards allergen extracts and four recombinant allergens. Int Arch Allergy Immunol 2013;162:131–4
Wood RA, Segall N, Ahlstedt S, Williams PB. Accuracy of IgE antibody laboratory results. Ann Allergy Asthma Immunol 2007 Jul;99:34–41
Cabrera-Freitag P, Goikoetxea MJ, Beorlegui C, Gamboa P, Gastaminza G, Fernandez-Benitez M, Ferrer M, Blanca M, Sanz ML. Can component-based microarray replace fluorescent enzimoimmunoassay in the diagnosis of grass and cypress pollen allergy? Clin Exp Allergy 2011 Oct;41:1440–6
Gadisseur R, Chapelle JP, Cavalier E. A new tool in the field of in-vitro diagnosis of allergy: preliminary results in the comparison of ImmunoCAP(c) 250 with the ImmunoCAP(c) ISAC. Clin Chem Lab Med 2011 Feb;49:277–80
Lizaso MT, Garcia BE, Tabar AI, Lasa E, Echechipia S, Alvarez MJ, Anda M, Gomez B. Comparison of conventional and component-resolved diagnostics by two different methods (Advia-Centaur/Microarray-ISAC) in pollen allergy. Ann Allergy Asthma Immunol 2011 Jul;107:35–41
Melioli G, Bonifazi F, Bonini S, Maggi E, Mussap M, Passalacqua G, Rossi ER, Vacca A, Canonica GW. The ImmunoCAP ISAC molecular allergology approach in adult multi-sensitized Italian patients with respiratory symptoms. Clin Biochem 2011 Aug;44:1005–11
Ott H, Baron JM, Heise R, Ocklenburg C, Stanzel S, Merk HF, Niggemann B, Beyer K. Clinical usefulness of microarray-based IgE detection in children with suspected food allergy. Allergy 2008 Nov;63:1521–8
Sastre J, Landivar ME, Ruiz-Garcia M, Andregnette-Rosigno MV, Mahillo I. How molecular diagnosis can change allergen-specific immunotherapy prescription in a complex pollen area. Allergy 2012 May;67:709–11
Twaroch TE, Focke M, Fleischmann K, Balic N, Lupinek C, Blatt K, Ferrara R, Mari A, Ebner C, Valent P, Spitzauer S, Swoboda I, Valenta R. Carrier-bound Alt a 1 peptides without allergenic activity for vaccination against Alternaria alternata allergy. Clin Exp Allergy 2012 Jun;42:966–75
Wang J, Godbold JH, Sampson HA. Correlation of serum allergy (IgE) tests performed by different assay systems. J Allergy Clin Immunol 2008 May;121:1219–24
Ebo DG, Hagendorens MM, De Knop KJ, Verweij MM, Bridts CH, De Clerck LS, Stevens WJ. Component-resolved diagnosis from latex allergy by microarray. Clin Exp Allergy 2010 Feb;40:348–58
Martinez-Aranguren R, Lizaso MT, Goikoetxea MJ, Garcia BE, Cabrera-Freitag P, Trellez O, Sanz ML. Is the determination of specific IgE against components using ISAC 112 a reproducible technique? PLoS One 2014;9:e88394
Darsow U, Brockow K, Pfab F, Jakob T, Petersson CJ, Borres MP, Ring J, Behrendt H, Huss-Marp J. Heterogeneity of molecular sensitization profiles in grass pollen allergy implications for immunotherapy? Clin Exp Allergy 2014 Mar 6
Huss-Marp J, Darsow U, Brockow K, Pfab F, Weichenmeier I, Schober W, Petersson CJ, Borres MP, Ring J, Behrendt H. Can immunoglobulin E-measurement replace challenge tests in allergic rhinoconjunctivits to grass pollen? Clin Exp Allergy 2011 Aug;41:1116–24
Ferrer M, Sanz ML, Sastre J, Bartra J, del Cuvillo A, Montoro J, Jauregui I, Davila I, Mullol J, Valero A. Molecular diagnosis in allergology: application of the microarray technique. J Investig Allergol Clin Immunol 2009;19 Suppl 1:19–24
Renault NK, Mirotti L, Alcocer MJ. Biotechnologies in new high-throughput food allergy tests: why we need them. Biotechnol Lett 2007 Mar;29:333–9
Sanz ML, Blazquez AB, Garcia BE. Microarray of allergenic component-based diagnosis in food allergy. Curr Opin Allergy Clin Immunol 2011 Jun;11:204–9
Ahlgrim C, Gutermuth J, Ænell A, Borres MP, Schäffner I, Darsow U et. al Comparison of molecular multiplex and singleplex analysis of IgE to grass pollen allergens in untreated German grass pollen allergic patients. J Invest All Clin Immunol 2015; Vol 25 (in press)
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions (as proposed by International Committee of Medical Journal Editors [ICMJE])
J. Huss-Marp: design of the study, data collection, statistical analysis, preparation and revision of the manuscript; J. Gutermuth: data collection and revision of the manuscript; I. Schäffner: preparation and revision of the manuscript; U. Darsow: data collection and revision of the manuscript; F. Pfab: data collection and revision of the manuscript; K. Brockow: data collection and revision of the manuscript; J. Ring: design of the study and revision of the manuscript; H. Behrendt: design of the study and revision of the manuscript; T. Jakob: data collection and revision of the manuscript; C. Ahlgrim: statistical analysis, preparation and revision of the manuscript
Cite this as: Huss-Marp J, Gutermuth J, Schäffner I, Darsow U, Pfab F, Brockow K, Ring J, Behrendt H, Jakob T, Ahlgrim C. Comparison of molecular and extract-based allergy diagnostics with multiplex and singleplex analysis. Allergo J Int 2015;24:46–53 DOI: 10.1007/s40629-015-0045-5
Funding
This work was supported by a research grant from Thermo Fisher Scientific, Phadia GmbH, Freiburg
Conflict of interest
Schäffner I and Huss-Marp J were employees of Thermo Fisher Scientific. Jakob T has received honoraria for consulting and for serving on the speakers buro of Thermo Fisher Scientific and has received research funding from Thermo Fisher Sceintific. The other authors state that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Huss-Marp, J., Gutermuth, J., Schäffner, I. et al. Comparison of molecular and extract-based allergy diagnostics with multiplex and singleplex analysis. Allergo J Int 24, 46–53 (2015). https://doi.org/10.1007/s40629-015-0046-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40629-015-0046-4